Advanced Manufacturing of Benflumetol Intermediates: A Technical Breakdown for Global Supply Chains
The pharmaceutical industry's relentless pursuit of efficient antimalarial therapies has placed significant focus on the optimization of Benflumetol synthesis pathways. Patent CN101747210A introduces a transformative methodology for preparing alpha-(di-n-butylaminomethyl)-2,7-dichloro-4-fluorenemethanol and its corresponding hydrochloride salt, a critical intermediate in the production of this life-saving medication. This technical disclosure addresses long-standing inefficiencies in traditional manufacturing by leveraging a direct acylation strategy coupled with an innovative solvent recycling protocol. For global supply chain leaders and R&D directors, understanding the nuances of this process is essential for securing a reliable benflumetol intermediate supplier capable of delivering high-purity materials at sustainable cost points. The patent outlines a robust chemical sequence that begins with readily available 2,7-dichlorofluorene, bypassing the cumbersome oxidation states required in legacy routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key antimalarial precursor has been plagued by operational complexity and economic inefficiency. Traditional methods, such as those documented in earlier literature including Acta Pharmaceutica Sinica, typically initiate the synthesis from fluorene-4-carboxylic acid. This starting material necessitates a multi-step transformation involving reduction and subsequent functionalization, which inherently extends the production cycle time. Furthermore, the post-treatment procedures in these conventional routes are notoriously labor-intensive, often requiring extensive ether extractions, rigorous drying with anhydrous magnesium sulfate, and multiple solvent removal steps. These unit operations not only increase the consumption of expensive organic solvents but also introduce potential points of failure regarding product loss and purity degradation. The reliance on such intricate workup procedures renders the traditional approach less favorable for large-scale industrial application, where throughput and cost-per-kilogram are paramount metrics for success.
The Novel Approach
In stark contrast, the methodology disclosed in CN101747210A presents a streamlined alternative that fundamentally restructures the synthetic logic. By utilizing 2,7-dichlorofluorene as the primary raw material, the process eliminates the need for pre-functionalized carboxylic acid derivatives, thereby shortening the overall synthetic timeline. The core innovation lies in the direct Friedel-Crafts acylation followed by a controlled reduction and amination sequence. Crucially, this approach integrates a solvent recovery system directly into the reaction workflow. Instead of discarding the ethanol mother liquor generated during the reduction phase, the process concentrates and recycles approximately 50% to 60% of this solvent for use in the subsequent amination step. This circular economy within the reactor vessel drastically reduces the demand for fresh dehydrated alcohol and minimizes waste generation. The result is a manufacturing protocol that is not only chemically elegant but also economically superior, offering substantial cost reduction in pharmaceutical intermediates manufacturing without compromising on product quality or yield.
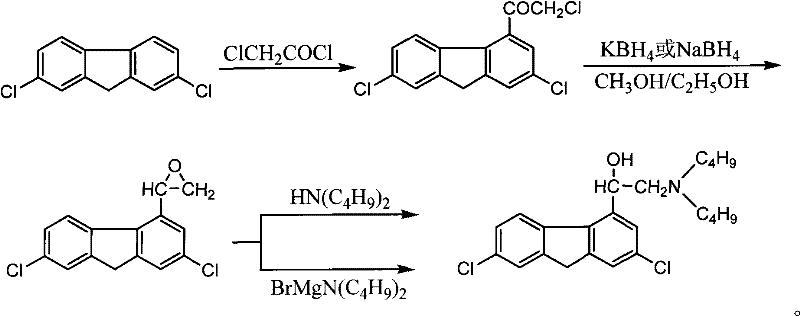
Mechanistic Insights into Friedel-Crafts Acylation and Borohydride Reduction
The chemical foundation of this improved process rests on two pivotal transformations: the electrophilic aromatic substitution and the subsequent hydride reduction. The initial step involves the reaction of 2,7-dichlorofluorene with chloroacetyl chloride in the presence of anhydrous aluminum trichloride (AlCl3) acting as a Lewis acid catalyst. This Friedel-Crafts acylation occurs selectively at the 4-position of the fluorene ring system, driven by the electronic activation provided by the aromatic structure and the steric environment created by the chlorine substituents. The reaction is typically conducted in chlorinated solvents such as methylene dichloride, trichloromethane, or 1,2-ethylene dichloride at low temperatures ranging from 0°C to 5°C to control exothermicity and prevent polyacylation. Following the acylation, the resulting 2,7-dichloro-4-chloroacetyl fluorene undergoes a critical reduction phase. Unlike rapid reductions that might lead to incomplete conversion or side reactions, this patent specifies an extended reaction window of 10 to 24 hours using potassium borohydride (KBH4) or sodium borohydride (NaBH4) in ethanol. This prolonged exposure ensures the quantitative reduction of the ketone moiety to the corresponding secondary alcohol, 2,7-dichlorofluorene-4-ethanol, which is essential for the fidelity of the subsequent amination step.
Impurity control is meticulously managed through the specific workup conditions described in the patent. After the reduction phase, the solid product is separated and washed with water until neutral. This washing step is vital for removing residual borohydride species and inorganic salts that could otherwise catalyze decomposition or interfere with the downstream amination reaction. The careful pH control during the final salification step, maintaining a pH value between 1 and 6 (preferably 2 to 4), ensures that the amine functionality is fully protonated to form the stable hydrochloride salt without promoting acid-catalyzed dehydration of the alcohol group. This attention to detail in the mechanistic execution guarantees a high-purity pharmaceutical intermediate suitable for the rigorous standards of antimalarial drug production. The ability to isolate the product simply by cooling and filtration, rather than complex chromatographic separations, underscores the robustness of this chemical design for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Alpha-(di-n-butylaminomethyl)-2,7-dichloro-4-fluorenemethanol Efficiently
The operational execution of this synthesis requires precise adherence to the temperature and stoichiometric parameters outlined in the patent embodiments to ensure reproducibility and safety. The process begins with the preparation of the acylating agent mixture, followed by the controlled addition of the fluorene substrate to manage heat evolution. Subsequent reduction must be monitored to ensure complete consumption of the ketone before proceeding to the amination stage. The integration of the mother liquor recycle loop is a critical operational step that demands careful volume management to maintain the correct concentration for the final coupling reaction. For a detailed breakdown of the specific reagent quantities, temperature profiles, and isolation techniques, please refer to the standardized synthesis guide below.
- Perform Friedel-Crafts acylation of 2,7-dichlorofluorene with chloroacetyl chloride using anhydrous aluminum trichloride in a chlorinated solvent.
- Reduce the resulting chloroacetyl fluorene derivative using potassium borohydride or sodium borohydride in ethanol for 10 to 24 hours.
- React the reduced alcohol intermediate with di-n-butylamine in concentrated ethanol mother liquor to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers distinct strategic advantages that extend beyond mere chemical yield. The primary value proposition lies in the drastic simplification of the material flow and the reduction of variable costs associated with solvent consumption. By eliminating the need for fresh ethanol in the amination step through the reuse of reduction mother liquors, the process decouples production costs from volatile solvent market prices. This internal recycling mechanism creates a more predictable cost structure, allowing for more accurate long-term budgeting and pricing stability for downstream partners. Furthermore, the use of common, commodity-grade reagents like 2,7-dichlorofluorene and chloroacetyl chloride ensures that the supply chain is not dependent on exotic or single-source specialty chemicals, thereby enhancing supply continuity and reducing the risk of raw material shortages.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of redundant solvent purchases and waste disposal fees. By recovering and reusing a significant portion of the ethanol solvent, the manufacturing facility effectively lowers its operating expenditure per batch. Additionally, the simplified workup procedure, which avoids energy-intensive distillation steps for solvent removal in the intermediate stages, contributes to lower utility costs. The avoidance of expensive extraction solvents like anhydrous diethyl ether in favor of recycled ethanol further compounds these savings, resulting in a leaner, more cost-effective production model that can withstand competitive market pressures.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route translates directly into supply chain resilience. The reaction conditions are relatively mild, utilizing temperatures between 0°C and 45°C, which reduces the stress on reactor equipment and minimizes the likelihood of thermal runaways or batch failures. The extended reduction time of 10 to 24 hours provides a wide operational window, allowing for flexible scheduling and reducing the pressure on operators to rush critical steps. This operational flexibility ensures consistent batch-to-batch quality and reliable delivery schedules, which are critical for maintaining the inventory levels required by global pharmaceutical manufacturers.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns well with modern green chemistry principles. The reduction in solvent waste volume lowers the burden on effluent treatment plants and reduces the facility's overall environmental footprint. The use of water washes to remove inorganic byproducts simplifies the waste stream, making it easier to treat and dispose of in compliance with stringent environmental regulations. The straightforward crystallization and filtration steps are inherently scalable, meaning that the transition from pilot plant to multi-ton commercial production can be achieved with minimal process re-engineering, facilitating rapid capacity expansion to meet surging demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and benefits of the technology. Understanding these details is crucial for technical teams evaluating the integration of this intermediate into their broader supply networks.
Q: What is the primary cost-saving mechanism in this synthesis route?
A: The process significantly lowers production costs by recycling 50% to 60% of the ethanol mother liquor from the reduction step, which is then reused as the solvent for the subsequent amination reaction.
Q: How does this method improve upon traditional synthesis routes for Benflumetol intermediates?
A: Unlike conventional methods that start from fluorene-4-carboxylic acid and require complex ether extractions and drying steps, this route utilizes direct acylation of 2,7-dichlorofluorene, resulting in simpler operations and higher suitability for industrial scale-up.
Q: What are the typical yield ranges for the final hydrochloride salt?
A: According to the patent data, the preparation of the hydrochloride salt achieves yields ranging from 59% to 65%, demonstrating high efficiency for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benflumetol Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against malaria. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101747210A are fully realized in practical manufacturing environments. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of alpha-(di-n-butylaminomethyl)-2,7-dichloro-4-fluorenemethanol meets the exacting standards required for API synthesis. Our infrastructure is designed to handle the specific solvent recycling and temperature control requirements of this process, delivering a product that supports your regulatory filings and commercial goals.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic efficiencies available through our manufacturing capabilities. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the quality and cost-effectiveness of your antimalarial drug supply chain.
