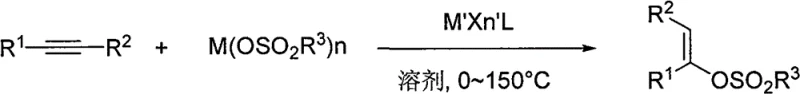
Advanced Gold-Catalyzed Synthesis of Enol Sulfonates for Commercial Scale-Up
The chemical industry is constantly evolving towards safer and more efficient synthetic methodologies, and patent CN101602638B represents a significant breakthrough in the preparation of enol sulfonic acid ester compounds. This intellectual property discloses a robust method that utilizes transition metal complexes as catalysts to facilitate the reaction between sulfonates and phenylacetylene compounds. Unlike traditional approaches that rely on hazardous reagents, this innovation employs phthalimide and acids as auxiliary catalysts within an inert organic solvent system. The process involves stirring and heating the mixture, followed by standard workup procedures including extraction, washing, drying, and silica gel column chromatography. The resulting enol sulfonate compounds are critical intermediates widely used in the synthesis of medicaments, agricultural pesticides, and various fine chemicals. By shifting from corrosive liquid acids to stable sulfonate salts, this technology addresses long-standing safety and equipment durability concerns in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of enol sulfonate compounds has been fraught with significant operational challenges and safety hazards that hinder large-scale industrial application. Conventional methods typically involve the enolization of ketones followed by reaction with sulfonic anhydrides, a process that generally necessitates strictly controlled low-temperature conditions to prevent decomposition or side reactions. Another prevalent technique, disclosed in earlier literature such as CN1013140A, utilizes free sulfonic acid in the presence of triphenylphosphine nitro-gold complexes. While this method can achieve yields ranging from 30% to 90%, the use of free sulfonic acid introduces severe corrosivity issues. In an industrial setting, strong sulfonic acids aggressively attack standard stainless steel reactors and piping, necessitating the use of expensive, specialized corrosion-resistant materials like glass-lined steel or Hastelloy. Furthermore, the handling of these corrosive liquids poses substantial risks to operators, requiring rigorous personal protective equipment and complex waste neutralization protocols. These factors collectively drive up capital expenditure and operational costs, limiting the economic viability of traditional routes for producing high-purity pharmaceutical intermediates.
The Novel Approach
The methodology outlined in patent CN101602638B offers a transformative solution by replacing corrosive free acids with stable metal sulfonate salts. This novel approach leverages a transition metal complex catalyst, specifically gold-based systems, alongside phthalimide and a catalytic amount of acid to drive the addition reaction across the alkyne triple bond. The reaction proceeds efficiently at temperatures between 50°C and 150°C, eliminating the need for energy-intensive cryogenic cooling required by older anhydride methods. By utilizing solid sulfonate salts such as sodium, potassium, copper, or calcium p-toluenesulfonate, the process inherently mitigates the corrosion risks associated with liquid sulfonic acids. This shift not only protects valuable manufacturing infrastructure but also simplifies the handling and storage of raw materials, as solid salts are generally more stable and less hazardous than their acidic counterparts. The result is a streamlined production workflow that delivers high-quality products with yields reaching up to 91.7% in optimized examples, demonstrating superior efficiency compared to many conventional protocols.
Mechanistic Insights into Gold-Catalyzed Alkyne Functionalization
The core of this synthetic advancement lies in the sophisticated activation of the alkyne moiety by the gold catalyst. In this mechanism, the transition metal complex, typically a cationic gold species generated in situ from precursors like [(PPh)3AuNO3], coordinates with the pi-electrons of the phenylacetylene triple bond. This coordination significantly increases the electrophilicity of the alkyne carbon atoms, making them susceptible to nucleophilic attack. The sulfonate anion, derived from the dissociation of the metal sulfonate salt in the presence of the acid co-catalyst, acts as the nucleophile. The presence of phthalimide as a co-catalyst is crucial; it likely assists in proton transfer steps or stabilizes intermediate species, facilitating the smooth progression of the reaction cycle. The acid co-catalyst, which can be sulfuric acid, oxalic acid, or pentafluorobenzoic acid, ensures the generation of the active sulfonic acid species in situ without requiring a large excess of corrosive liquid acid. This delicate balance allows for the regioselective formation of the vinyl sulfonate bond, preserving the stereochemistry required for downstream applications in drug synthesis.
From an impurity control perspective, this catalytic system offers distinct advantages over stoichiometric methods. Traditional routes often generate significant amounts of salt waste or require harsh quenching steps that can degrade sensitive functional groups on the aromatic rings. In contrast, the gold-catalyzed pathway described here operates under milder conditions, reducing the likelihood of thermal degradation or polymerization of the alkyne starting material. The use of specific metal sulfonates, such as copper p-toluenesulfonate, has been shown to enhance yields significantly, suggesting that the metal cation may play a secondary role in stabilizing the transition state or facilitating the leaving group ability of the sulfonate. Furthermore, the reaction tolerates a wide range of substituents on the alkyne, including electron-donating groups like methoxy and electron-withdrawing groups like fluoro, as well as heteroaromatic systems like thiophene. This broad substrate scope indicates a robust catalytic cycle that is less prone to deactivation by coordinating impurities, thereby ensuring a cleaner crude product profile and simplifying the subsequent purification via silica gel chromatography.
How to Synthesize Enol Sulfonate Efficiently
To implement this synthesis effectively, one must carefully control the stoichiometry and reaction environment as detailed in the patent examples. The process begins with the thorough drying of the sulfonate salt to prevent moisture from interfering with the Lewis acidic gold catalyst. The phenylacetylene derivative is then introduced into an inert organic solvent, such as 1,2-dichloroethane or chlorobenzene, along with the transition metal catalyst and phthalimide. The mixture is heated to the optimal temperature range, typically around 150°C for maximum conversion, although lower temperatures like 70°C can still provide respectable yields depending on the substrate reactivity.
- Dry the sulfonate salt and mix it with phenylacetylene compounds in an inert organic solvent system containing a transition metal complex catalyst.
- Add phthalimide and an acid as co-catalysts to the mixture, then stir and heat the system to a temperature between 50°C and 150°C.
- After the reaction completes (10-20 hours), perform extraction, washing, and drying, followed by silica gel column chromatography to isolate the pure enol sulfonate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates into tangible strategic benefits beyond mere chemical yield. The primary advantage lies in the drastic reduction of maintenance costs associated with reactor corrosion. By eliminating the need to handle bulk quantities of free sulfonic acid, manufacturing facilities can utilize standard glass-lined or high-grade stainless steel equipment without the fear of rapid degradation. This extends the lifecycle of capital assets and reduces the frequency of costly shutdowns for equipment replacement or repair. Additionally, the use of solid sulfonate salts simplifies logistics and storage; solids are easier to transport, have longer shelf lives, and pose fewer regulatory hurdles compared to hazardous corrosive liquids. This stability ensures a more reliable supply of raw materials, minimizing the risk of production delays caused by shipping restrictions or supplier shortages of specialized acids.
- Cost Reduction in Manufacturing: The elimination of corrosive reagents directly impacts the bottom line by lowering the total cost of ownership for production equipment. Facilities do not need to invest in exotic alloys or frequent linings, and the reduced hazard profile lowers insurance premiums and waste disposal costs. Furthermore, the high yields achieved with specific catalyst combinations, such as copper p-toluenesulfonate, mean less raw material is wasted per kilogram of final product. The process also avoids the generation of toxic byproducts, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations without the need for expensive scrubbing technologies.
- Enhanced Supply Chain Reliability: The raw materials for this process, including phenylacetylenes and various metal sulfonates, are commercially available and chemically stable. Unlike sensitive anhydrides that require cold chain logistics, these salts can be stored at ambient temperatures, reducing inventory holding costs. The robustness of the reaction conditions allows for flexible scheduling, as the process is less sensitive to minor fluctuations in temperature or mixing rates compared to low-temperature anhydride methods. This flexibility enables manufacturers to respond more quickly to market demand spikes for high-purity pharmaceutical intermediates, ensuring consistent delivery timelines to downstream clients.
- Scalability and Environmental Compliance: Scaling this reaction from laboratory to pilot and commercial plant is straightforward due to the absence of exothermic hazards associated with acid-anhydride mixing. The reaction proceeds smoothly upon heating, allowing for predictable heat management in large reactors. From an environmental standpoint, the process is inherently greener; it avoids the release of acidic vapors and reduces the volume of acidic wastewater generated during workup. The use of gold catalysts, while precious, is done at low loading levels, and the potential for catalyst recovery further enhances the sustainability profile. This aligns with the growing corporate mandate for green chemistry practices, making the final product more attractive to eco-conscious multinational buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enol sulfonate synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating process feasibility and procurement officers assessing supplier capabilities.
Q: What are the primary advantages of using sulfonate salts over sulfonic acids in this synthesis?
A: Using sulfonate salts eliminates the severe corrosion issues associated with free sulfonic acids, significantly extending equipment lifespan and improving operator safety during industrial production.
Q: Which catalysts are most effective for this enol sulfonate transformation?
A: Transition metal complexes, particularly gold complexes such as [(PPh)3AuNO3], [(PPh)3AuBF4], and [(PPh)3AuOTf], have demonstrated high efficacy, often yielding over 90% conversion under optimized conditions.
Q: Is this process scalable for commercial manufacturing of pharmaceutical intermediates?
A: Yes, the process operates at mild temperatures (50-150°C) using stable solid raw materials, making it highly suitable for commercial scale-up without the need for specialized corrosion-resistant reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Enol Sulfonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN101602638B to maintain competitiveness in the global fine chemical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. We are committed to delivering high-purity enol sulfonate intermediates that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against comprehensive analytical standards. Our facility is equipped to handle transition metal catalysis safely and efficiently, incorporating the necessary containment and recovery systems to maximize yield and minimize environmental impact.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis for switching from traditional acid-based routes or need specific COA data to validate our quality standards, we are ready to provide the support you need. Contact us today to request route feasibility assessments and discover how our expertise in gold-catalyzed transformations can secure your supply chain for next-generation pharmaceutical and agrochemical projects.
