Advanced Three-Step Synthesis Of Isotope Labeled Sulfadimidine For Veterinary Drug Analysis
Advanced Three-Step Synthesis Of Isotope Labeled Sulfadimidine For Veterinary Drug Analysis
The global demand for precise analytical standards in veterinary drug residue monitoring has never been more critical, driven by stringent food safety regulations and the need to mitigate ecological risks associated with sulfonamide usage. Patent CN110028457B introduces a groundbreaking synthesis method for isotope-labeled sulfabromodimethyl pyrimidine, specifically designed to serve as a high-fidelity internal standard for Isotope Dilution Mass Spectrometry (IDMS). This technology addresses the longstanding challenges of low yield and complex purification found in traditional routes, offering a robust solution for producing reference materials with exceptional isotopic abundance. By leveraging a streamlined three-step process starting from readily available isotope-labeled aniline, this innovation ensures that the final product meets the rigorous purity thresholds required for trace-level detection in complex biological matrices. For stakeholders in the analytical chemistry and veterinary pharmaceutical sectors, this patent represents a significant leap forward in securing reliable supply chains for essential quality control reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isotope-labeled sulfonamides has been plagued by inefficient multi-step pathways that rely on expensive and difficult-to-handle precursors like benzene-D6. Prior art, such as the method disclosed in Chinese patent document CN107879985A, typically necessitates a five-step reaction sequence which inherently accumulates impurities and drastically reduces overall yield at each stage. These conventional processes often suffer from harsh reaction conditions that can lead to isotopic scrambling or loss, compromising the very integrity of the labeled compound needed for accurate mass spectrometry. Furthermore, the extensive purification required after each step increases solvent consumption, waste generation, and production time, making the final standard reagent prohibitively expensive for routine laboratory use. The complexity of these legacy routes also poses significant scalability challenges, limiting the ability of suppliers to meet sudden surges in demand for food safety testing kits.
The Novel Approach
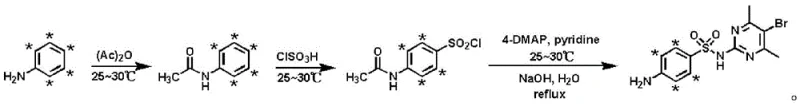
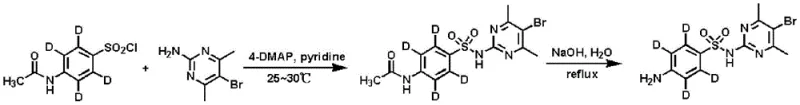
In stark contrast, the methodology outlined in CN110028457B utilizes a highly efficient three-step synthetic strategy that begins with the direct acetylation of isotope-labeled aniline, significantly shortening the process route while maximizing atom economy. This novel approach operates under mild conditions, typically maintaining temperatures between 25-30°C for key transformation steps, which minimizes energy consumption and reduces the risk of thermal degradation of sensitive intermediates. The strategic selection of aniline-D5 as the starting material allows for a more direct incorporation of the isotopic label into the aromatic ring, ensuring high retention of isotopic abundance throughout the synthesis. By simplifying the workflow to just acetylation, chlorosulfonation, and a final coupling-hydrolysis sequence, the process facilitates easier separation and purification, directly translating to higher throughput and reduced operational costs. This streamlined architecture not only enhances the economic viability of producing these specialized reagents but also ensures a consistent supply of high-quality standards for the industry.
Mechanistic Insights into Isotope-Labeled Sulfonamide Synthesis
The core of this technological advancement lies in the precise control of reaction kinetics and thermodynamics across the three distinct stages, ensuring that the deuterium labels remain intact while functional groups are selectively transformed. The initial acetylation step protects the amine group of the aniline-D5, preventing unwanted side reactions during the subsequent aggressive chlorosulfonation phase where chlorosulfonic acid is introduced in a controlled manner at low temperatures. This protection strategy is crucial for maintaining the structural integrity of the labeled ring system, as uncontrolled sulfonation could lead to polysubstitution or isotopic exchange with the acidic medium. The final stage involves a nucleophilic substitution where the sulfonyl chloride intermediate reacts with 2-amino-4,6-dimethyl-5-bromopyrimidine, followed by a carefully monitored alkaline hydrolysis to reveal the free amine. This cascade reaction is optimized to proceed with minimal byproduct formation, leveraging the specific reactivity of the pyrimidine ring to achieve high conversion rates without compromising the isotopic signature of the molecule.
Impurity control is rigorously managed through specific workup procedures designed to remove unreacted starting materials and acidic byproducts without losing the valuable labeled product. For instance, the quenching of excess chlorosulfonic acid is performed with ice water at strictly controlled temperatures of 0-5°C to prevent exothermic runaway reactions that could degrade the intermediate. The final purification involves pH adjustment to a narrow range of 5.0-6.0, a critical parameter identified in the patent where the product precipitates as a faint yellow solid with maximum yield, while soluble impurities remain in the filtrate. This precision in pH control demonstrates a deep understanding of the compound's solubility profile, allowing for a simple filtration step to replace complex chromatographic separations. Such mechanistic optimization ensures that the final API intermediate or standard reagent achieves a chemical purity and isotopic abundance both exceeding 99 percent, satisfying the most demanding analytical requirements.
How to Synthesize Isotope Labeled Sulfadimidine Efficiently
The synthesis protocol described in the patent provides a clear, reproducible pathway for manufacturing high-purity isotope-labeled sulfadimidine suitable for commercial scale-up. The process is designed to be robust against minor variations in operating conditions, making it ideal for transfer from laboratory bench to pilot plant operations. Key to the success of this method is the strict adherence to temperature controls during the addition of reagents, particularly the dropwise addition of acetic anhydride and chlorosulfonic acid under ice-salt baths to manage exotherms. Detailed standardized synthetic steps regarding molar ratios, solvent choices, and specific workup procedures are essential for replicating the high yields reported in the examples. Manufacturers looking to adopt this technology should focus on the precise stoichiometry of the coupling agents and the timing of the hydrolysis step to ensure optimal product quality.
- React isotope-labeled aniline with acetic anhydride at 25-30°C to prepare isotope-labeled acetaminophenyl.
- Treat the labeled acetaminophenyl with chlorosulfonic acid at 25-30°C to form isotope-labeled p-acetaminophenylsulfonyl chloride.
- Couple the sulfonyl chloride with 2-amino-4,6-dimethyl-5-bromopyrimidine and hydrolyze under alkaline conditions to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method offers tangible benefits in terms of cost structure and supply reliability, primarily driven by the reduction in process complexity and raw material costs. The shift from a five-step to a three-step process inherently reduces the consumption of solvents, reagents, and energy, leading to a significantly lower cost of goods sold (COGS) for the final isotope-labeled product. Moreover, the use of aniline-D5 as a starting material, which is generally more accessible and cost-effective than the benzene-D6 derivatives used in older methods, further enhances the economic efficiency of the production line. This cost reduction in veterinary drug intermediate manufacturing allows suppliers to offer more competitive pricing for analytical standards, making high-quality food safety testing more accessible to a broader range of laboratories and regulatory bodies.
- Cost Reduction in Manufacturing: The elimination of two entire synthetic steps drastically cuts down on labor hours, equipment usage time, and waste disposal costs associated with intermediate purification. By avoiding the need for expensive transition metal catalysts or complex chromatographic columns for purification, the process relies on simple crystallization and filtration techniques that are inexpensive to operate at scale. This simplification means that the capital expenditure required for setting up production lines is lower, and the operational expenditure is minimized through reduced solvent recovery loads. Consequently, the overall manufacturing footprint is smaller, aligning with modern green chemistry principles and reducing the environmental compliance burden on the facility.
- Enhanced Supply Chain Reliability: The robustness of the three-step route ensures a more predictable production schedule, as there are fewer points of failure where a batch could be lost due to low yield or failed purification. The mild reaction conditions reduce the dependency on specialized high-pressure or cryogenic equipment, meaning that production can be maintained even if specific utility systems face temporary constraints. Additionally, the high yield of each step, reported to be as high as 99 percent in the acetylation stage, ensures that raw material inventory is converted into finished goods with maximum efficiency, reducing the need for large safety stocks of expensive isotope-labeled precursors. This reliability is crucial for maintaining continuous supply to customers who depend on these standards for regulatory compliance testing.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids hazardous reagents and extreme conditions that are difficult to manage in large reactors. The workup procedures involve neutralization and precipitation, which are unit operations that scale linearly and predictably from grams to tons without requiring re-optimization. Furthermore, the reduction in solvent volume and the ability to recover and reuse solvents like dichloromethane and pyridine contributes to a lower E-factor (environmental factor), making the process more sustainable. This alignment with environmental, social, and governance (ESG) goals is increasingly important for multinational corporations seeking to partner with suppliers who demonstrate a commitment to responsible manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of isotope-labeled sulfadimidine, based on the specific disclosures within the patent documentation. Understanding these details helps R&D and procurement teams evaluate the suitability of this material for their specific analytical workflows. The answers provided reflect the optimized conditions and performance metrics achieved through the novel synthetic route, highlighting its superiority over legacy methods. Stakeholders are encouraged to review these insights to fully appreciate the value proposition of this advanced manufacturing technology.
Q: What is the primary advantage of this synthesis method over prior art?
A: Unlike previous methods requiring five steps from benzene-D6 with limited application, this patent utilizes a concise three-step route from aniline-D5, achieving yields over 89% and purity exceeding 99%.
Q: Why is isotope-labeled sulfadimidine critical for food safety testing?
A: It serves as an internal standard for Isotope Dilution Mass Spectrometry (IDMS), effectively eliminating recovery rate differences during sample pretreatment and ensuring accurate quantification of sulfonamide residues in animal products.
Q: What are the purity specifications of the final product?
A: The synthesized isotope-labeled sulfadimidine achieves both chemical purity and stable isotope abundance of over 99 percent, meeting the rigorous requirements for use as a quantitative standard reagent.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isotope Labeled Sulfadimidine Supplier
As a leader in the fine chemical industry, NINGBO INNO PHARMCHEM is uniquely positioned to leverage this patented technology to deliver high-purity isotope-labeled sulfadimidine to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of major diagnostic and pharmaceutical companies without compromising on quality. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art LC-MS and NMR instrumentation to verify both chemical purity and isotopic abundance for every batch we release. Our commitment to excellence means that every gram of material we supply is backed by comprehensive analytical data, giving our partners the confidence they need for their critical quantitative assays.
We invite you to collaborate with us to optimize your supply chain for veterinary drug analysis standards. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your long-term strategic goals. Let us be your trusted partner in securing a stable, high-quality supply of essential isotope-labeled intermediates.
