Advanced Palladium-Catalyzed Amination for High-Purity Ortho-Bicyclopropane Aniline Intermediates
Advanced Palladium-Catalyzed Amination for High-Purity Ortho-Bicyclopropane Aniline Intermediates
The pharmaceutical and agrochemical industries constantly seek robust synthetic routes for complex intermediates that balance cost efficiency with high purity standards. Patent CN101253144B introduces a groundbreaking methodology for the preparation of ortho-bicyclopropane-substituted primary anilines, specifically compounds of Formula I. These structures serve as critical building blocks for advanced fungicides, such as those described in WO 03/074491. The innovation lies in a streamlined two-step process that utilizes palladium-catalyzed cross-coupling followed by hydrogenation, effectively bypassing the limitations of traditional imine-based routes. By enabling the use of less reactive but economically superior chloro-substrates, this technology represents a significant leap forward for manufacturers aiming to optimize their supply chains for high-value agrochemical intermediates.
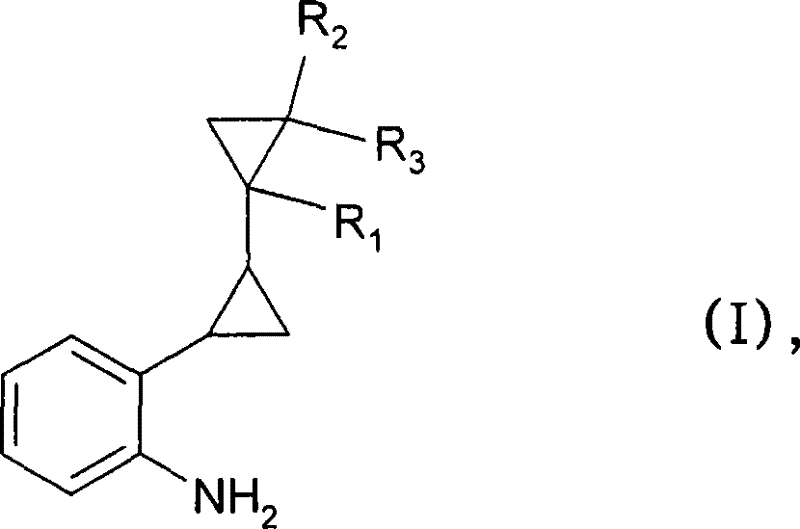
As a reliable agrochemical intermediate supplier, understanding the structural nuances of Formula I is essential. The substituents R1, R2, and R3 can vary from hydrogen to C1-C4 alkyl groups, allowing for significant chemical diversity. However, the core challenge has always been installing the amino group at the ortho position relative to the bulky bicyclopropane moiety. Conventional wisdom suggested that electron-rich, sterically hindered chlorobenzenes were poor candidates for direct amination. This patent shatters that assumption by identifying specific catalytic systems that activate the carbon-chlorine bond efficiently. For R&D directors focused on scalability, this means accessing a broader range of starting materials without compromising on reaction kinetics or final product quality, thereby securing a more resilient production pipeline for complex organic molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those detailed in WO 03/074491, relied heavily on the use of benzophenone imine as the nitrogen source. While chemically feasible, this approach suffers from severe economic and operational drawbacks that hinder large-scale adoption. The synthesis requires a two-step sequence where the halogenated substrate is first coupled with the expensive imine, followed by a cleavage step using hydroxylamine. Benzophenone imine is not only costly but also introduces additional processing steps that generate substantial waste. Furthermore, these legacy methods were largely restricted to bromo- or iodo-substrates due to the insufficient reactivity of chloro-derivatives under standard conditions.
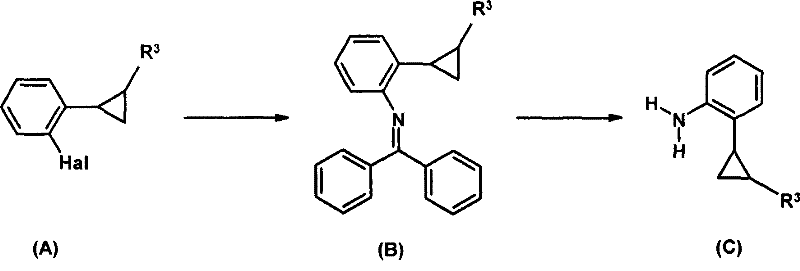
The reliance on bromo- or iodo-starting materials drastically inflates the raw material costs, making the final active ingredient less competitive in the global market. Additionally, the cleavage of the benzophenone protecting group often leads to difficult purification challenges, as removing the resulting benzophenone by-product can require extensive chromatography or recrystallization. For procurement managers, this translates to higher COGS (Cost of Goods Sold) and longer lead times. The inability to utilize cheaper chlorobenzene precursors effectively locked manufacturers into a high-cost paradigm, limiting the commercial viability of fungicides dependent on this specific aniline scaffold. The search for a more direct, cost-effective amination strategy became imperative for the industry.
The Novel Approach
The methodology disclosed in CN101253144B revolutionizes this landscape by employing benzylamine (Formula III) as the nucleophile in a palladium-catalyzed coupling reaction. This shift allows for the direct formation of a secondary amine intermediate (Formula IV), which is subsequently converted to the primary aniline via simple catalytic hydrogenation. The brilliance of this approach lies in its compatibility with chloro-substrates. By utilizing advanced ligand systems, the process activates the robust carbon-chlorine bond, enabling the use of 2-(2-chlorophenyl)cyclopropanes which are significantly more economical than their bromo counterparts. This substitution alone drives a substantial cost reduction in fungicide manufacturing.
Moreover, the deprotection step using hydrogen and a metal catalyst (such as Pd/C) is exceptionally clean, producing toluene as the only major by-product, which is easily removed. This contrasts sharply with the messy hydroxylamine cleavage of the old method. The reaction conditions are also remarkably flexible, operating effectively at temperatures ranging from 50°C to 180°C and tolerating various solvent systems including dimethoxyethane and xylenes. For supply chain heads, this robustness means the process is less sensitive to minor fluctuations in reaction parameters, ensuring consistent batch-to-batch quality. The ability to run the reaction under normal pressure and without the need for inert atmosphere gloveboxes further simplifies the engineering requirements for commercial scale-up.
Mechanistic Insights into Pd-Catalyzed Amination of Hindered Substrates
The success of this novel route hinges on the precise selection of the palladium catalyst and ligand system. The patent elucidates that standard phosphine ligands often fail to promote the amination of sterically hindered ortho-bicyclopropane chlorobenzenes. Instead, the invention highlights the efficacy of specialized ligands such as ferrocenyl diphosphines (e.g., Josiphos variants), bulky monodentate phosphines (e.g., P(tBu)3, X-Phos), and N-heterocyclic carbenes (e.g., I-Pr). These ligands create a specific electronic and steric environment around the palladium center that facilitates the oxidative addition of the aryl chloride, which is typically the rate-determining step. The bulky nature of ligands like tri-tert-butylphosphine prevents the formation of inactive palladium black, maintaining the catalyst in a highly active monomeric state throughout the reaction cycle.
Impurity control is another critical aspect addressed by the mechanistic design. When using primary amines like benzylamine, there is a risk of beta-elimination side reactions or over-alkylation to form tertiary amines. The patent demonstrates that by carefully tuning the ligand-to-palladium ratio (often between 1:100 to 1:10000) and selecting ligands with appropriate bite angles, these side pathways are suppressed. For instance, the use of aminophosphine ligands like PCy2NMe2BiPh has shown exceptional yields (up to 98%) while minimizing the formation of by-products such as Formula IA or IB. This level of control is vital for R&D teams aiming to meet strict regulatory limits on genotoxic impurities in final drug substances. The ability to predict and mitigate these impurities at the intermediate stage ensures a smoother regulatory filing process.
How to Synthesize 2-Bicyclopropan-2-yl-aniline Efficiently
The synthesis of this high-value intermediate follows a logical two-step sequence that balances reactivity with operational simplicity. First, the halogenated bicyclopropane precursor is reacted with benzylamine in the presence of a strong base like sodium tert-butoxide and a catalytic amount of the optimized palladium complex. This step forms the protected benzyl-aniline intermediate. Following isolation or telescoping, the second step involves hydrogenation over a heterogeneous catalyst like Pd/C to remove the benzyl group. Detailed standardized synthesis steps see the guide below.
- React ortho-bicyclopropane-substituted halobenzene (Formula II) with benzylamine (Formula III) using a palladium catalyst and base.
- Isolate the intermediate benzyl(2-bicyclopropan-2-yl-phenyl)amine (Formula IV).
- Perform catalytic hydrogenation on the intermediate to yield the final primary aniline (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of the technology described in CN101253144B offers transformative benefits that extend beyond mere chemical yield. The primary advantage is the drastic reduction in raw material costs achieved by switching from bromo- to chloro-substrates. Chlorobenzenes are commodity chemicals with stable pricing and abundant global supply, whereas bromo-analogs are subject to greater volatility and higher baseline costs. This switch fundamentally alters the cost structure of the final fungicide, providing a competitive edge in price-sensitive agricultural markets. Additionally, the replacement of expensive benzophenone imine with commodity benzylamine further compresses the bill of materials.
- Cost Reduction in Manufacturing: The elimination of expensive reagents and the simplification of the workup procedure lead to significant operational savings. The hydrogenation step replaces a complex chemical cleavage, reducing solvent consumption and waste disposal costs. Since the process avoids the use of stoichiometric amounts of precious metal scavengers often required for older Pd-catalyzed methods, the overall metal burden is lower. This qualitative improvement in process efficiency translates directly to improved margins without the need for aggressive price hikes, ensuring long-term commercial viability for the product line.
- Enhanced Supply Chain Reliability: By relying on widely available starting materials like chlorobenzene derivatives and benzylamine, manufacturers reduce the risk of supply disruptions. The robustness of the catalytic system means that production is less susceptible to delays caused by the need for highly specialized, moisture-sensitive reagents that require glovebox handling. The reaction can be performed in standard stainless steel reactors under normal pressure, facilitating easier technology transfer between different manufacturing sites. This flexibility ensures continuity of supply even during periods of global logistical stress, a key metric for supply chain heads evaluating vendor reliability.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively from gram to multi-kilogram scales without loss of efficiency. The use of hydrogen gas as a reductant produces water as a benign by-product, aligning with green chemistry principles. Furthermore, the ability to recycle the palladium catalyst, as noted in the patent preferences, minimizes heavy metal waste. This environmental profile simplifies regulatory compliance and reduces the carbon footprint of the manufacturing process, appealing to increasingly eco-conscious stakeholders and regulatory bodies worldwide.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into existing production frameworks.
Q: Why is the chloro-substrate preferred over bromo-substrates in this synthesis?
A: Chloro-substrates are significantly more economical and readily available than bromo- or iodo-analogs. This patent demonstrates that specific palladium ligand systems can overcome the traditionally low reactivity of chlorobenzene derivatives, enabling cost-effective large-scale production without sacrificing yield.
Q: What are the key advantages of using benzylamine over benzophenone imine?
A: Benzylamine is a much cheaper and more accessible nucleophile compared to benzophenone imine. Furthermore, the deprotection step for benzylamine (hydrogenation) is cleaner and generates fewer by-products than the hydroxylamine cleavage required for benzophenone imines, simplifying downstream purification.
Q: How does this method improve impurity profiles for fungicide manufacturing?
A: By utilizing specific ligand systems like Josiphos or N-heterocyclic carbenes, the reaction minimizes side reactions such as beta-elimination or over-alkylation. This results in a cleaner intermediate profile, which is critical for meeting the stringent purity specifications required in final agrochemical active ingredients.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bicyclopropan-2-yl-aniline Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates in the agrochemical value chain. Our technical team has extensively analyzed the methodologies outlined in CN101253144B and possesses the expertise to execute this sophisticated palladium-catalyzed amination with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facilities are equipped with rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch meets stringent purity specifications required for downstream fungicide synthesis.
We invite you to collaborate with us to leverage this advanced chemistry for your product portfolio. Our experts can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how the switch to chloro-substrates can impact your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to drive efficiency and innovation in your agrochemical manufacturing operations.
