Revolutionizing ADC Manufacturing: Solid-Phase Synthesis of DS-8201 Intermediates for Commercial Scale
Revolutionizing ADC Manufacturing: Solid-Phase Synthesis of DS-8201 Intermediates for Commercial Scale
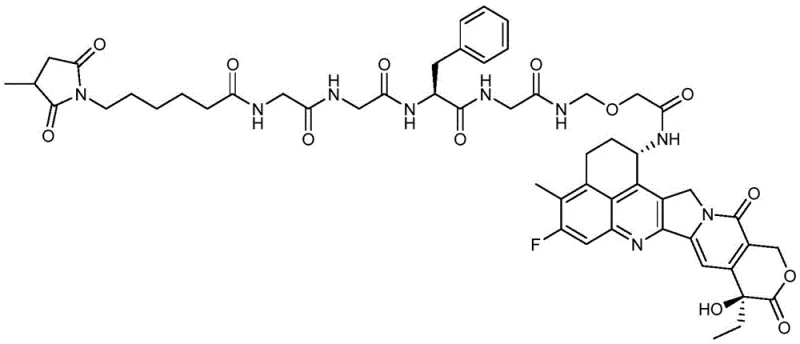
The landscape of Antibody-Drug Conjugate (ADC) manufacturing is undergoing a significant transformation, driven by the urgent need for more efficient and scalable synthesis routes for complex linker-payload systems. Patent CN115109118A, published in late 2022, introduces a groundbreaking method for the solid-phase synthesis of DS-8201 intermediates, specifically targeting the intricate linker region that connects the cytotoxic payload to the antibody. This technology addresses critical bottlenecks in current production workflows by shifting from traditional liquid-phase peptide synthesis to a robust solid-phase approach. By anchoring the synthesis to a resin support, the method streamlines the construction of the tetrapeptide-like linker, eliminating the need for tedious intermediate isolations and purifications that have historically plagued ADC manufacturing. This innovation not only enhances the chemical purity of the final construct but also establishes a foundation for reliable large-scale production of high-value oncology therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of DS-8201 and its intermediates relied heavily on liquid-phase strategies, which are inherently cumbersome and resource-intensive. As detailed in the background of the patent, existing routes often involve introducing the expensive and sensitive topoisomerase-I inhibitor payload (DXd derivative) too early in the synthetic sequence. In these conventional liquid-phase pathways, the payload is subjected to multiple reaction steps, including repeated deprotection and coupling cycles, which inevitably lead to significant degradation and yield loss. Furthermore, each step in a liquid-phase synthesis typically requires rigorous purification, such as column chromatography or recrystallization, to remove unreacted starting materials and by-products. This not only consumes vast quantities of organic solvents but also extends the production timeline considerably, making the process economically unsustainable for commercial-scale manufacturing of blockbuster drugs like DS-8201.
The Novel Approach
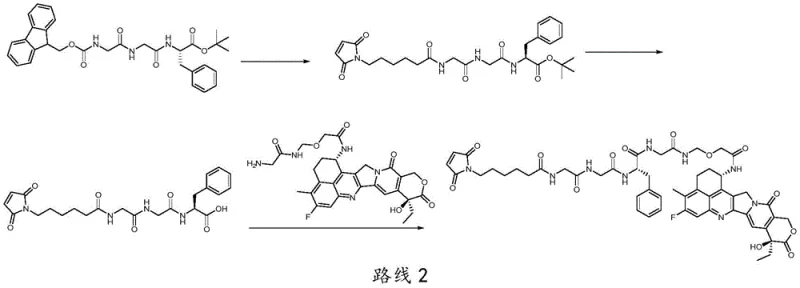
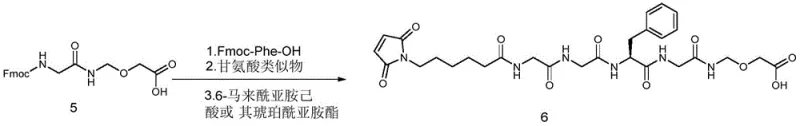
The novel approach disclosed in patent CN115109118A fundamentally reimagines the assembly of the linker fragment by employing solid-phase peptide synthesis (SPPS) techniques. Instead of building the molecule in solution, the synthesis begins by coupling the hydroxyl end of a precursor compound (Compound 5) directly onto a solid support, such as 2-CTC resin or Merrifield resin. This immobilization allows for the sequential addition of amino acid building blocks, specifically Fmoc-Phe-OH and glycine analogues, followed by the attachment of the maleimide functionality. The brilliance of this method lies in its simplicity: after each coupling reaction, impurities and excess reagents are simply washed away, removing the need for intermediate purification. Once the full linker sequence is assembled on the resin, it is cleaved under mild acidic conditions to release the high-purity linker intermediate (Compound 6). This strategy effectively decouples the linker synthesis from the payload handling until the very final stages, preserving the integrity of the cytotoxic agent.
Mechanistic Insights into Fmoc Solid-Phase Peptide Synthesis
The core mechanism driving this technological advancement is the Fluorenylmethyloxycarbonyl (Fmoc) solid-phase synthesis strategy, which offers orthogonal protection capabilities ideal for complex molecule assembly. The process initiates with the loading of Compound 5 onto the resin, where the hydroxyl group forms a stable ester linkage with the solid support. Subsequent cycles involve the removal of the Fmoc protecting group using a base like piperidine in DMF, exposing the free amine for the next coupling event. Coupling reagents such as HOBt, HOAt, or carbodiimides (e.g., DIC, EDC) activate the carboxylic acid of the incoming amino acid (Fmoc-Phe-OH or Fmoc-Gly-OH), facilitating rapid amide bond formation. This cycle is repeated to build the specific peptide sequence required for the DS-8201 linker. The use of solid support ensures that the growing chain remains localized, allowing for the use of excess reagents to drive equilibrium towards product formation without complicating the workup. Finally, the maleimide caproic acid is attached, completing the linker architecture before the entire construct is cleaved from the resin.
Impurity control is intrinsically managed through the physical nature of solid-phase synthesis. In liquid-phase reactions, side products and incomplete reactions create a complex mixture that is difficult to separate, especially when dealing with hydrophobic payloads. In contrast, the solid-phase method ensures that any unreacted species or soluble by-products are physically separated from the resin-bound product during the washing steps. The patent specifies the use of mild cleavage reagents, such as 0.5-5% trifluoroacetic acid (TFA) in dichloromethane (DCM) or acetic acid (AcOH) mixtures. This mildness is crucial because it prevents the premature hydrolysis of the ester bonds or the degradation of the maleimide ring, which are sensitive to harsh acidic or basic conditions. By optimizing the cleavage conditions, the method ensures that the final linker intermediate (Compound 6) is obtained with high chemical fidelity, ready for the final conjugation step with the payload.
How to Synthesize DS-8201 Linker Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing the critical linker intermediate with high efficiency and reproducibility. The process leverages standard peptide synthesis equipment, making it accessible for facilities already equipped for oligonucleotide or peptide manufacturing. The key to success lies in the precise control of resin loading and the selection of appropriate coupling reagents to minimize racemization and ensure complete conversion at each step. By adhering to the specified molar ratios and reaction times, manufacturers can achieve consistent quality batch after batch. For a detailed breakdown of the specific operational parameters, reagent concentrations, and step-by-step instructions, please refer to the standardized guide below.
- Couple the hydroxyl end of Compound 5 to 2-CTC or Merrifield resin, followed by sequential Fmoc-deprotection and coupling of Fmoc-Phe-OH and glycine analogues.
- Extend the peptide chain by coupling 6-maleimidocaproic acid or its succinimidyl ester to the resin-bound intermediate.
- Cleave the linker from the resin using mild acidic conditions (e.g., 1% TFA/DCM) and purify to obtain Compound 6, then couple with the payload Compound 7.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this solid-phase synthesis method represents a substantial opportunity to optimize the cost structure and reliability of ADC production. Traditional liquid-phase synthesis is often a bottleneck due to its low throughput and high dependency on skilled labor for complex purification tasks. By adopting the method described in CN115109118A, organizations can transition to a more automated and linear production flow. The elimination of intermediate purification steps means a drastic reduction in the consumption of chromatography media and organic solvents, which are significant cost drivers in fine chemical manufacturing. Furthermore, the simplified workflow reduces the overall cycle time, allowing for faster turnover of batches and improved responsiveness to market demand. This efficiency gain is particularly valuable for high-demand oncology drugs where supply continuity is critical for patient access.
- Cost Reduction in Manufacturing: The economic benefits of this solid-phase approach are derived primarily from the simplification of the downstream processing. In conventional routes, the repeated need for column chromatography after every synthetic step incurs high costs in terms of silica gel, solvents, and labor hours. By replacing these isolation steps with simple filtration and washing protocols, the new method significantly lowers the variable costs associated with production. Additionally, the ability to use excess reagents to drive reactions to completion without fear of difficult separations improves the effective yield of the expensive amino acid and linker building blocks. This reduction in material waste and processing complexity translates directly into a lower cost of goods sold (COGS), providing a competitive edge in the pricing of the final API or intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and scalability of the solid-phase technique. Liquid-phase syntheses of complex linkers are prone to variability; a single failed purification can result in the loss of an entire batch of high-value intermediate. The solid-phase method mitigates this risk by ensuring that the intermediate remains anchored and protected throughout the synthesis, reducing the likelihood of catastrophic batch failures. Moreover, the reagents required for this process, such as Fmoc-amino acids and standard coupling agents, are commodity chemicals with stable global supply chains. This reduces the dependency on custom-synthesized building blocks that might have long lead times, thereby shortening the overall procurement cycle and ensuring a steady flow of materials for continuous manufacturing operations.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method aligns well with green chemistry principles and industrial scalability requirements. The reduction in solvent usage, particularly the avoidance of large volumes of eluents for chromatography, significantly decreases the facility's hazardous waste footprint. This simplifies waste treatment processes and helps manufacturers meet increasingly stringent environmental regulations. Scalability is also enhanced because solid-phase reactors can be easily scaled up or operated in parallel to increase capacity without a proportional increase in floor space or equipment complexity. The ability to produce high-purity intermediates consistently makes the technology suitable for commercial-scale production, ranging from pilot plant quantities to multi-ton annual outputs required for global drug distribution.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this solid-phase synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the practical aspects of adopting this method. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing lines.
Q: What are the advantages of using solid-phase synthesis over liquid-phase for DS-8201 linkers?
A: Solid-phase synthesis eliminates the need for intermediate purification steps like column chromatography after every coupling. This significantly reduces solvent consumption, processing time, and product loss, leading to higher overall yields and purity compared to traditional liquid-phase methods.
Q: Which resins and cleavage agents are compatible with this synthesis method?
A: The process utilizes 2-CTC Resin or Merrifield Resin with a substitution degree of 0.2 to 2.0 mmol/g. Cleavage is achieved using mild acidic reagents such as 0.5-5% TFA in DCM, or mixtures of AcOH/TFE/DCM, ensuring the stability of sensitive functional groups during release.
Q: How does this method impact the cost of goods for ADC production?
A: By simplifying the workflow to simple washing steps between couplings and avoiding repeated protection/deprotection cycles in solution, the method drastically lowers operational costs. It also allows for the use of excess reagents to drive reactions to completion without complicating downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DS-8201 Intermediate Supplier
As the demand for next-generation ADC therapies continues to surge, having a manufacturing partner with deep expertise in complex linker synthesis is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this field, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the stringent purity specifications required for oncology intermediates, supported by rigorous QC labs that ensure every batch meets the highest international standards. We understand the critical nature of DS-8201 supply chains and are committed to delivering consistent quality and reliability to our global partners.
We invite pharmaceutical companies and CROs to collaborate with us to leverage this advanced solid-phase synthesis technology for their ADC programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to clinical and commercial success.
