Advanced Palladium-Catalyzed Cyanation for Scalable Production of High-Purity Pyridine Derivatives
Advanced Palladium-Catalyzed Cyanation for Scalable Production of High-Purity Pyridine Derivatives
The global demand for functionalized pyridine derivatives continues to surge, driven by their critical role as building blocks in the synthesis of advanced agrochemicals and pharmaceutical agents. Among these, 2-cyano-3,6-dichloropyridine stands out as a pivotal intermediate, yet its traditional manufacturing routes have long been plagued by significant safety and environmental challenges. A groundbreaking technical solution is detailed in patent CN101671300B, which discloses a novel method for preparing this valuable compound using potassium ferrocyanide as a safe cyanide source. This innovation marks a substantial departure from legacy processes that rely on highly toxic reagents, offering a pathway that aligns perfectly with modern green chemistry principles and stringent regulatory frameworks. By leveraging a palladium-catalyzed system, this technology not only enhances operational safety but also delivers superior reaction efficiency, making it an ideal candidate for reliable agrochemical intermediate suppliers seeking to optimize their supply chains.
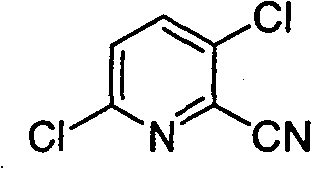
The strategic importance of this synthesis route cannot be overstated for procurement managers and R&D directors alike, as it directly addresses the dual pressures of cost containment and compliance. The utilization of potassium ferrocyanide effectively mitigates the risks associated with handling virulent cyanides like potassium cyanide or sodium cyanide, which are notorious for their acute toxicity and the complex waste treatment protocols they necessitate. Furthermore, the process operates under relatively mild conditions, typically ranging from 40°C to 200°C, utilizing common organic solvents such as N,N-dimethylformamide or dimethyl sulfoxide. This flexibility in reaction parameters allows for fine-tuning to maximize yield and purity, ensuring that the final product meets the rigorous specifications required for downstream applications in fungicide and herbicide manufacturing. As the industry moves towards more sustainable practices, adopting such a green processing method becomes a competitive advantage for any organization aiming to reduce lead time for high-purity agrochemical intermediates while maintaining robust safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-cyano-3,6-dichloropyridine has been fraught with significant technical and safety hurdles that hinder efficient commercial scale-up of complex pyridine derivatives. Traditional pathways, such as those described in older patents like US4766219, often involve a multi-step sequence starting with fluorination followed by cyanation. These legacy methods typically require the use of highly active and dangerous fluorinating agents, coupled with the subsequent use of potassium cyanide or sodium cyanide for the introduction of the cyano group. The reliance on these virulent cyanides introduces severe operational dangers, necessitating specialized containment facilities and extensive personal protective equipment, which drastically inflates capital and operational expenditures. Moreover, the total recovery of these two-step reactions is frequently restricted by suboptimal reaction conditions, leading to lower overall yields and the generation of substantial quantities of hazardous waste acids. The residual unreacted matters and toxic byproducts impose a heavy burden on post-treatment systems, requiring complex neutralization and detoxification processes that further erode profit margins and complicate environmental compliance.
The Novel Approach
In stark contrast to these archaic techniques, the methodology outlined in patent CN101671300B presents a streamlined, one-step cyanation strategy that fundamentally reshapes the production landscape. This innovative approach utilizes 2,3,6-trichloropyridine as the starting material and employs potassium ferrocyanide as the cyanating agent in the presence of a palladium catalyst system. The substitution of free cyanide salts with the stable ferrocyanide complex is a game-changer, as it virtually eliminates the risk of acute cyanide poisoning and significantly reduces the hazard level of the waste stream. The reaction proceeds smoothly in various polar aprotic solvents, with the patent demonstrating successful outcomes in solvents like 1,4-dioxane and N-methylpyrrolidone. This versatility allows manufacturers to select solvents that best fit their existing infrastructure, facilitating easier integration into current production lines. By simplifying the synthetic route and removing the need for hazardous fluorination pre-steps, this method achieves a remarkable balance between high efficiency and environmental stewardship, positioning it as a superior choice for cost reduction in agrochemical intermediates manufacturing.
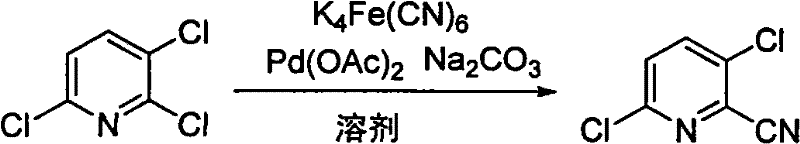
Mechanistic Insights into Palladium-Catalyzed Cyanation
The core of this technological breakthrough lies in the sophisticated palladium-catalyzed mechanism that enables the nucleophilic substitution of the chlorine atom at the 2-position of the pyridine ring. The catalytic cycle initiates with the oxidative addition of the aryl chloride bond of 2,3,6-trichloropyridine to the palladium(0) species generated in situ from palladium diacetate. This step is crucial as it activates the relatively inert carbon-chlorine bond, forming an organopalladium intermediate that is primed for further transformation. The presence of sodium carbonate acts as a base, facilitating the activation of the potassium ferrocyanide and assisting in the transmetallation or ligand exchange process where the cyanide group is transferred to the palladium center. Unlike free cyanide ions which can be unpredictable and dangerous, the cyanide ligands in ferrocyanide are tightly bound, releasing them in a controlled manner only in the presence of the active catalyst. This controlled release mechanism is key to minimizing side reactions such as hydrolysis of the nitrile group or formation of biaryl byproducts, thereby ensuring a cleaner reaction profile. The final reductive elimination step releases the desired 2-cyano-3,6-dichloropyridine product and regenerates the active palladium catalyst, allowing the cycle to continue with high turnover numbers.
From an impurity control perspective, this mechanism offers distinct advantages over traditional nucleophilic aromatic substitution methods. The use of potassium ferrocyanide significantly reduces the formation of tar-like polymeric byproducts that are often observed when using alkali metal cyanides under harsh thermal conditions. The mildness of the reaction conditions, particularly when optimized within the preferred range of 60°C to 120°C, helps preserve the integrity of the sensitive pyridine ring and prevents the degradation of the newly formed cyano group. Furthermore, the specific molar ratios recommended in the patent, such as a ratio of 1:0.2:0.2:0.005 for the substrate, ferrocyanide, base, and catalyst respectively, are finely tuned to maximize conversion while minimizing the accumulation of palladium black or other catalyst deactivation products. This precise stoichiometric control ensures that the crude product obtained after reaction completion requires less intensive purification, directly translating to higher isolated yields and reduced solvent consumption during workup. For R&D teams, understanding these mechanistic nuances is vital for troubleshooting and optimizing the process for specific reactor configurations, ensuring consistent quality across different batch sizes.
How to Synthesize 2-Cyano-3,6-Dichloropyridine Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the specific reaction parameters outlined in the patent to achieve optimal results. The process begins by charging a reaction vessel with the requisite amounts of 2,3,6-trichloropyridine and potassium ferrocyanide, ensuring that the atmosphere is purged with an inert gas such as nitrogen or argon to prevent oxidation of the catalyst. The choice of solvent is critical, with options including dimethylformamide, dimethyl sulfoxide, or tetrahydrofuran, each offering different solubility profiles and boiling points that can influence the reaction kinetics. Once the reagents are dissolved or suspended, the palladium diacetate and sodium carbonate are introduced, and the mixture is heated to the target temperature, which can range broadly from 40°C to 200°C depending on the desired reaction rate and solvent stability. Monitoring the reaction progress via HPLC or GC is recommended to determine the precise endpoint, typically falling within a 2 to 20-hour window, after which the mixture is cooled and subjected to a straightforward workup procedure involving filtration and washing with dilute ammonia solution to remove residual palladium and inorganic salts. The detailed standardized synthesis steps see the guide below for a comprehensive walkthrough of the experimental protocol.
- Combine 2,3,6-trichloropyridine and potassium ferrocyanide in an organic solvent such as DMF or DMSO under inert gas protection.
- Add sodium carbonate and palladium diacetate catalysts to the reaction mixture and heat to temperatures between 60°C and 120°C.
- Maintain reaction for 2 to 20 hours, then cool, filter, wash with ammonia solution, and purify to obtain the final cyano-product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers profound strategic benefits that extend far beyond simple chemical transformation. The primary advantage lies in the drastic simplification of the supply chain for raw materials; potassium ferrocyanide is a widely available, non-regulated commodity chemical, whereas potassium cyanide is a heavily controlled substance subject to strict transportation and storage regulations. This shift eliminates the logistical bottlenecks and security costs associated with procuring and handling virulent cyanides, thereby enhancing supply chain reliability and reducing the administrative burden on EHS departments. Additionally, the reduction in hazardous waste generation translates directly into lower disposal costs and a smaller environmental footprint, which is increasingly important for maintaining corporate social responsibility credentials and meeting sustainability goals. By streamlining the production process and removing high-risk steps, companies can achieve substantial cost savings in overall manufacturing operations without compromising on product quality or throughput.
- Cost Reduction in Manufacturing: The economic implications of switching to this ferrocyanide-based route are significant, primarily driven by the elimination of expensive safety infrastructure and waste treatment protocols. Traditional cyanation processes require dedicated scrubbing systems and specialized neutralization tanks to handle toxic off-gases and liquid effluents, all of which represent substantial capital investment and ongoing maintenance costs. By utilizing a non-toxic cyanide source, the need for such extreme mitigation measures is vastly reduced, allowing facilities to operate with leaner overheads. Furthermore, the high yields demonstrated in the patent examples, reaching up to 92%, mean that less raw material is wasted per unit of product, improving the overall material efficiency and lowering the cost of goods sold. This efficiency gain is compounded by the simplified post-treatment process, which requires fewer extraction and purification steps, thereby saving on solvent usage and energy consumption during distillation and drying phases.
- Enhanced Supply Chain Reliability: In the volatile landscape of fine chemical sourcing, reliability is paramount, and this method significantly de-risks the supply of critical intermediates. Since potassium ferrocyanide is not subject to the same stringent regulatory controls as free cyanides, supply disruptions due to regulatory crackdowns or transportation bans are virtually non-existent. This stability ensures a continuous flow of materials, preventing production stoppages that can ripple through the entire value chain and delay deliveries to downstream customers. Moreover, the robustness of the reaction conditions allows for greater flexibility in scheduling and batch sizing, enabling manufacturers to respond more agilely to fluctuating market demands. For supply chain planners, this means improved forecast accuracy and the ability to maintain lower safety stock levels, freeing up working capital and warehouse space for other strategic initiatives.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the ton-scale often reveals hidden complexities, but this technology is inherently designed for industrial viability. The use of common solvents and standard heating equipment means that existing reactors can be utilized without major retrofitting, accelerating the timeline from development to commercial production. From an environmental compliance standpoint, the process generates significantly less hazardous waste, simplifying the permitting process for new facilities or the expansion of existing ones. The reduction in toxic load makes it easier to meet increasingly strict discharge limits imposed by environmental agencies, reducing the risk of fines and operational shutdowns. This alignment with green chemistry principles not only safeguards the license to operate but also enhances the brand reputation of the manufacturer as a responsible and forward-thinking partner in the global chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced cyanation technology, providing clarity for stakeholders evaluating its potential impact on their operations. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for assessing the feasibility of integrating this route into existing manufacturing portfolios and for anticipating the operational changes required for successful adoption. We encourage technical teams to review these points carefully as they formulate their strategies for process optimization and supplier selection.
Q: Why is potassium ferrocyanide preferred over potassium cyanide in this synthesis?
A: Potassium ferrocyanide is significantly less toxic and safer to handle than potassium cyanide or sodium cyanide. It eliminates the severe operational hazards associated with virulent cyanides and reduces the environmental burden of waste acid treatment, making the process greener and more compliant with modern EHS standards.
Q: What represents the key advantage of this palladium-catalyzed method regarding yield?
A: Unlike conventional methods which often suffer from low total recovery due to harsh fluorination steps or side reactions, this novel approach demonstrates high efficiency. Experimental data within the patent indicates yields reaching up to 92% with purity levels around 95-97%, offering a robust pathway for industrial scaling.
Q: Can this process be scaled for commercial agrochemical production?
A: Yes, the method is explicitly designed for industrial production. It utilizes readily available raw materials, operates under relatively mild conditions (40-200°C), and simplifies post-treatment by avoiding complex detoxification steps required for free cyanide waste, thereby facilitating large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Cyano-3,6-Dichloropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed cyanation technology and are fully equipped to leverage it for our global clientele. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless and efficient. Our state-of-the-art facilities are designed to handle complex chemistries with the highest regard for safety and quality, supported by rigorous QC labs that enforce stringent purity specifications on every batch we produce. We understand that consistency is key in the fine chemical industry, and our commitment to process excellence guarantees that our 2-cyano-3,6-dichloropyridine meets the exacting standards required for the synthesis of high-value agrochemicals and pharmaceuticals.
We invite you to collaborate with us to unlock the full commercial potential of this innovative synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this greener methodology can optimize your bottom line. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing you to make informed decisions with confidence. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner dedicated to driving value through technological innovation and operational excellence.
