Advanced One-Pot Catalytic Route for High-Purity Flometoquin Intermediate Manufacturing
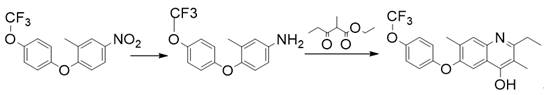
The pharmaceutical and agrochemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways for synthesizing complex heterocyclic intermediates. A significant breakthrough in this domain is documented in patent CN111808020B, which details a novel synthetic method for producing the flometoquin intermediate, specifically 2-ethyl-3,7-dimethyl-6-(4-(trifluoromethoxy)phenoxy)quinoline-4-ol. This patent represents a paradigm shift from traditional multi-step batch processes to a streamlined one-pot catalytic hydrogenation strategy. By integrating the reduction of the nitro group and the subsequent cyclization condensation into a single operational unit, this technology addresses critical pain points regarding waste generation, operational complexity, and overall process economics. For R&D directors and process chemists, this approach offers a compelling alternative to legacy methods that rely on stoichiometric metal reductants, promising a cleaner profile and superior scalability for commercial manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoline-based agrochemical intermediates like the flometoquin precursor has relied heavily on classical reduction techniques that are increasingly viewed as environmentally unsustainable and economically inefficient. Traditional literature methods, such as those cited in prior art, typically employ iron powder or tin chloride as the reducing agents to convert the nitro-substituted benzene starting material into the corresponding aniline intermediate. While chemically effective, these stoichiometric reductions generate massive quantities of inorganic solid waste, often referred to as iron mud or metallic sludge, which poses severe challenges for waste treatment facilities and significantly inflates disposal costs. Furthermore, these conventional routes are inherently discontinuous, requiring the isolation and purification of the aniline intermediate before proceeding to the cyclization step, which introduces additional unit operations, increases solvent consumption, and inevitably leads to yield losses during transfer and handling stages.
The Novel Approach
In stark contrast to the cumbersome traditional workflows, the methodology disclosed in patent CN111808020B introduces a highly integrated one-pot synthesis that elegantly bypasses the need for isolating unstable or sensitive intermediates. This innovative route utilizes molecular hydrogen as the clean reductant in the presence of a heterogeneous Platinum on Carbon (Pt/C) catalyst, coupled with methanesulfonic acid to facilitate the subsequent cyclization. By combining the reduction and ring-closing steps into a single reactor charge, the process drastically simplifies the operational workflow, eliminating the need for intermediate workups and the associated solvent exchanges. This consolidation not only enhances the overall throughput of the manufacturing line but also aligns perfectly with the principles of green chemistry by minimizing material usage and maximizing atom economy, making it an ideal candidate for modern, high-efficiency chemical production facilities.
Mechanistic Insights into Pt/C-Catalyzed Reductive Cyclization
The core of this technological advancement lies in the synergistic action of the Pt/C catalyst and the acidic promoter within a toluene solvent system under hydrogen pressure. The mechanism initiates with the catalytic hydrogenation of the nitro group on the 2-methyl-4-nitro-1-(4-(trifluoromethoxy)phenoxy)benzene substrate, efficiently converting it into the reactive aniline species in situ without the accumulation of hazardous byproducts. Once the aniline is formed, it immediately undergoes a condensation reaction with ethyl 2-methyl-3-oxopentanoate, driven by the presence of methanesulfonic acid which acts as a potent Brønsted acid catalyst to activate the carbonyl electrophile. The reaction system is designed with a reflux water diversion apparatus, which is critical for shifting the equilibrium of the cyclodehydration step towards the formation of the quinoline ring by continuously removing the water byproduct, thus ensuring high conversion rates and preventing the reverse hydrolysis reaction.
From an impurity control perspective, this catalytic route offers distinct advantages over metal-mediated reductions. The use of a heterogeneous catalyst like Pt/C allows for precise control over the reduction potential, minimizing the risk of over-reduction or side reactions that often plague iron powder reductions, such as the formation of azo-compounds or hydroxylamines. Additionally, the absence of soluble metal salts in the reaction mixture means that the final product is not contaminated with trace metals that would require expensive scavenging steps to meet regulatory standards for agrochemical active ingredients. The simplicity of the workup, involving merely filtering off the solid catalyst and adjusting the pH to precipitate the product, ensures that the impurity profile remains clean and consistent, facilitating easier downstream purification and quality control assurance for the final high-purity flometoquin intermediate.
How to Synthesize 2-Ethyl-3,7-dimethyl-6-(4-(trifluoromethoxy)phenoxy)quinoline-4-ol Efficiently
Implementing this synthesis on an industrial scale requires careful attention to reaction parameters to maximize the benefits of the one-pot design. The process begins by charging a pressure-rated reactor with toluene, the specific loading of Pt/C catalyst, methanesulfonic acid, and the two key organic starting materials. The system is then pressurized with hydrogen gas to a mild pressure range of 0.01 to 0.1 MPa and heated to reflux, with a specialized setup to remove water azeotropically as the reaction progresses. Monitoring the reaction endpoint via HPLC is crucial to determine the precise moment when the nitro starting material is fully consumed and the cyclization is complete, ensuring optimal yield before cooling and isolation.
- Charge a pressure reactor with toluene solvent, Pt/C catalyst, methanesulfonic acid, the nitro-substituted benzene substrate, and ethyl 2-methyl-3-oxopentanoate.
- Introduce hydrogen gas to pressurize the system to 0.01-0.1 MPa and initiate reflux with continuous water diversion to drive the cyclization equilibrium.
- Upon completion, filter to recover the Pt/C catalyst, pour the filtrate into water, adjust pH to 5 with hydrochloric acid, and isolate the white solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented one-pot technology translates directly into tangible operational efficiencies and risk mitigation strategies. The elimination of stoichiometric metal reductants like iron powder removes a major variable from the supply chain, reducing dependency on bulk commodity metals whose prices can be volatile and whose quality can vary significantly between suppliers. Furthermore, the drastic reduction in solid waste generation simplifies the environmental compliance landscape, lowering the overhead costs associated with hazardous waste disposal and treatment, which are often hidden but substantial expenses in fine chemical manufacturing. This streamlined process also shortens the overall production cycle time by removing intermediate isolation steps, thereby increasing the asset utilization rate of the manufacturing plant and allowing for faster response times to market demand fluctuations.
- Cost Reduction in Manufacturing: The transition from a two-step batch process to a one-pot catalytic sequence fundamentally alters the cost structure of producing this agrochemical intermediate. By removing the requirement for large quantities of iron powder or tin chloride, the direct material costs are significantly lowered, and the labor costs associated with multiple charging, filtering, and drying steps are drastically reduced. Additionally, the ability to recover and potentially recycle the heterogeneous Pt/C catalyst after simple filtration offers a pathway to further optimize the catalyst cost per kilogram of product, contributing to substantial long-term savings in the cost of goods sold without compromising on quality or yield.
- Enhanced Supply Chain Reliability: Relying on a simplified synthetic route with fewer unit operations inherently reduces the number of potential failure points in the manufacturing process, leading to more predictable and reliable delivery schedules. The use of common, commercially available solvents like toluene and stable catalysts ensures that raw material sourcing is robust and less susceptible to geopolitical or logistical disruptions that might affect specialized reagents. This stability is crucial for maintaining continuous supply to downstream formulators, ensuring that the production of the final flometoquin insecticide is not interrupted by upstream intermediate shortages, thus securing the value chain for our partners.
- Scalability and Environmental Compliance: The green chemistry attributes of this method, specifically the avoidance of heavy metal waste and the use of catalytic hydrogenation, make it exceptionally well-suited for scale-up in regulated jurisdictions with strict environmental standards. The process generates minimal solid waste, primarily consisting of the spent catalyst which can be sent for precious metal recovery, thereby simplifying the permitting process for new production lines or capacity expansions. This environmental compatibility not only future-proofs the supply against tightening regulations but also enhances the corporate sustainability profile of the final agrochemical product, a factor that is increasingly important for global brand owners and distributors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent specifications and process data. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing supply networks or production schedules.
Q: What are the primary environmental advantages of this new synthesis method compared to traditional iron powder reduction?
A: The new method eliminates the use of stoichiometric iron powder or tin chloride, thereby preventing the generation of large quantities of difficult-to-treat metallic sludge (iron mud) and significantly reducing solid waste disposal costs.
Q: What yield and purity levels can be expected from this one-pot catalytic process?
A: According to patent data, the process consistently achieves yields greater than or equal to 80% (with examples reaching ~87%) and product purity levels exceeding 95%.
Q: Is the platinum catalyst recoverable for reuse in subsequent batches?
A: Yes, the heterogeneous Pt/C catalyst can be easily recovered via simple filtration after the reaction is complete, allowing for potential recycling and further reduction in material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Flometoquin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes for key agrochemical building blocks like the flometoquin intermediate. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped to handle catalytic hydrogenation processes safely and efficiently, adhering to stringent purity specifications and operating within rigorous QC labs to guarantee that every batch meets the highest international standards for quality and performance.
We invite you to collaborate with us to optimize your supply chain for this vital intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our implementation of this one-pot technology can drive value for your organization. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global agrochemical market through superior manufacturing excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
