Industrial Scale-Up of 2-Chloro-3-Amino-4-Picoline: Advanced Synthetic Route for Global Pharma Supply Chains
Industrial Scale-Up of 2-Chloro-3-Amino-4-Picoline: Advanced Synthetic Route for Global Pharma Supply Chains
The pharmaceutical industry constantly demands robust, scalable, and cost-effective synthetic routes for critical antiretroviral intermediates. A pivotal development in this domain is documented in patent CN101157654A, which outlines a novel seven-step preparation method for 2-chloro-3-amino-4-picoline. This compound serves as an essential building block for Nevirapine and other non-nucleoside reverse transcriptase inhibitors. The technical breakthrough lies in the strategic selection of starting materials, shifting away from complex nitro-reduction pathways to a more accessible acetone-based condensation strategy. This approach not only simplifies the reaction sequence but also enhances the overall controllability of the process, addressing key pain points for R&D directors focused on impurity profiles and process safety. By leveraging common industrial reagents, this methodology offers a compelling alternative for manufacturers seeking to optimize their supply chain for high-purity pharmaceutical intermediates.
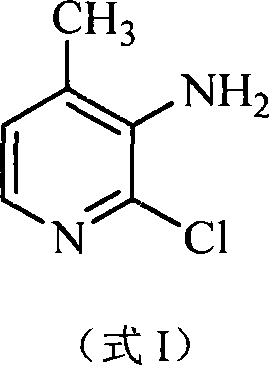
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-chloro-3-amino-4-picoline has been plagued by significant technical hurdles that impede efficient commercial production. Traditional routes often rely on the reduction of 2-chloro-3-nitro-4-methyl pyridine, a process that introduces severe safety risks due to the handling of energetic nitro compounds and the requirement for high-pressure hydrogenation equipment. Furthermore, alternative pathways involving the chlorination of 2,6-dihydroxy-4-methyl-3-nitrile pyridine suffer from poor regioselectivity, leading to difficult-to-remove isomers and a complex purification burden. These legacy methods frequently exhibit unstable yields, often fluctuating wildly between batches, which creates unpredictability for supply chain planners. The reliance on specialized starting materials that are not commoditized further exacerbates cost volatility, making long-term procurement planning difficult for large-scale API manufacturers who require consistent quality and volume.
The Novel Approach
In stark contrast, the methodology described in CN101157654A introduces a paradigm shift by utilizing acetone, sodium methoxide, and methyl formate as primary feedstocks. This novel approach constructs the pyridine ring de novo through a sequence of condensation, cyclization, and functional group transformations. The elimination of nitro-reduction steps significantly lowers the safety profile of the plant operations, reducing the need for explosion-proof infrastructure. Moreover, the reaction conditions are notably milder, with key steps operating at atmospheric pressure and moderate temperatures, which simplifies equipment requirements and lowers capital expenditure. The process demonstrates a remarkable ability to control impurities, achieving a final product purity greater than 99.7 percent without the need for excessive recrystallization cycles. This streamlined workflow translates directly into operational efficiency, allowing manufacturers to achieve a total output rate exceeding 18.4 percent with much greater consistency than previous generations of technology.
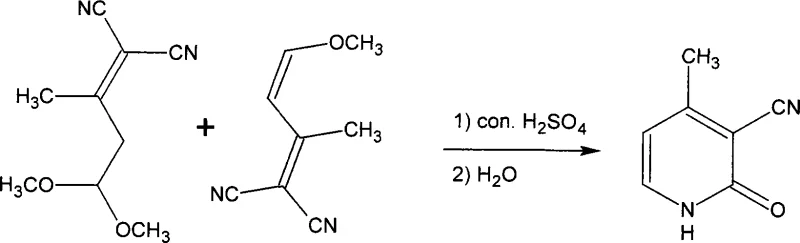
Mechanistic Insights into the Seven-Step Cyclization and Degradation
The core of this synthetic innovation lies in the precise construction of the pyridine nucleus and the subsequent functionalization. The initial steps involve the formation of a sodium enolate from acetone and methyl formate, which then undergoes condensation with malononitrile. This creates a highly reactive intermediate that is primed for cyclization. The critical ring-closing step utilizes concentrated sulfuric acid to facilitate the formation of 3-cyano-4-picoline-2-one. This cyclization is exothermic and requires careful thermal management to prevent polymerization or decomposition of the sensitive nitrile groups. Following ring formation, the 2-position is selectively chlorinated using phosphorus oxychloride and phosphorus pentachloride. This chlorination step is vital for setting up the final amination, as the chlorine atom serves as a leaving group in potential nucleophilic substitutions, although in this specific route, it is retained as a key structural feature of the final API intermediate.
The final transformation involves the conversion of the amide to the amine via Hofmann degradation, a reaction that demands rigorous control over stoichiometry and temperature. In this process, 2-chloro-4-picoline-3-methane amide is treated with sodium hypohalite generated in situ from bromine and sodium hydroxide. The reaction is initiated at low temperatures, specifically between 0°C and 5°C, to control the formation of the N-bromoamide intermediate and prevent over-oxidation or ring degradation. As the reaction progresses, the temperature is carefully raised to facilitate the rearrangement and loss of carbon dioxide, yielding the primary amine. This mechanism ensures that the amino group is introduced exclusively at the 3-position, maintaining the regiochemical integrity required for downstream coupling reactions in Nevirapine synthesis. The ability to execute this degradation with a yield of over 91 percent in the final step underscores the robustness of the chemical design.
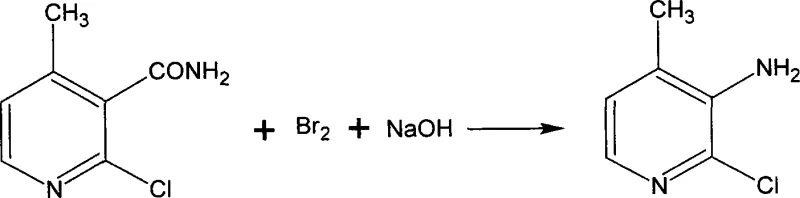
How to Synthesize 2-Chloro-3-Amino-4-Picoline Efficiently
Implementing this seven-step synthesis requires a disciplined approach to process control, particularly regarding the handling of reactive intermediates and the management of exothermic events. The protocol begins with the preparation of the sodium enolate, followed by sequential addition of reagents to build the carbon skeleton. Each transition between steps involves specific work-up procedures, such as pH adjustments and solvent extractions, which are critical for removing inorganic salts and byproducts that could poison subsequent catalysts or reactions. For R&D teams looking to adopt this technology, it is essential to replicate the specific temperature gradients and addition rates described in the patent examples to maximize yield. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational parameters required for successful execution.
- Condensation of acetone with methyl formate and sodium methoxide to form the sodium enolate intermediate.
- Acidification and reaction with malononitrile followed by cyclization using concentrated sulfuric acid to form the pyridone ring.
- Chlorination using phosphorus oxychloride followed by hydrolysis and final Hofmann degradation to yield the target amine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial advantages that resonate deeply with procurement managers and supply chain heads focused on cost reduction and reliability. The primary driver of value is the substitution of expensive or hazardous starting materials with commodity chemicals like acetone and methyl formate. This shift drastically simplifies the sourcing strategy, as these materials are available from a wide network of global suppliers, reducing the risk of supply disruption due to single-source dependency. Furthermore, the elimination of high-pressure hydrogenation and nitro-handling steps reduces the regulatory burden and insurance costs associated with plant operations. The process is inherently safer and more environmentally compliant, minimizing the generation of hazardous waste streams that require costly disposal. These factors combine to create a manufacturing profile that is not only economically superior but also more resilient to market fluctuations and regulatory changes.
- Cost Reduction in Manufacturing: The economic benefits of this route are derived from the fundamental chemistry rather than speculative savings. By utilizing acetone and sodium methoxide, the raw material cost base is significantly lowered compared to routes requiring specialized nitro-pyridines. The high purity of the intermediates reduces the need for extensive purification steps, which in turn lowers solvent consumption and energy usage per kilogram of product. Additionally, the high yield in the final Hofmann degradation step ensures that valuable precursors are not wasted, maximizing the return on material investment. This efficiency translates into a more competitive cost structure for the final API, allowing pharmaceutical companies to maintain healthy margins even in price-sensitive markets.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of widely available industrial feedstocks. Unlike specialized intermediates that may have long lead times or limited production capacity, acetone and formates are produced at massive scales globally. This abundance ensures that production schedules can be maintained without the fear of raw material shortages. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in reagent quality, further stabilizing the supply output. For supply chain heads, this reliability is crucial for meeting the strict delivery timelines required by global pharmaceutical clients, ensuring that drug production lines never halt due to intermediate shortages.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, featuring steps that are easily transferable from pilot plant to multi-ton production. The reaction conditions are moderate, avoiding the need for exotic materials of construction for reactors, which facilitates faster capacity expansion. From an environmental standpoint, the route avoids the generation of heavy metal waste associated with some catalytic hydrogenations and reduces the load of nitrogenous waste compared to nitro-reduction routes. This alignment with green chemistry principles simplifies the permitting process for new manufacturing sites and ensures long-term compliance with increasingly stringent environmental regulations, safeguarding the asset against future regulatory risks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-chloro-3-amino-4-picoline. These answers are derived directly from the technical specifications and experimental data provided in the patent literature. They are intended to provide clarity on the feasibility, quality, and logistical aspects of adopting this synthetic route for commercial manufacturing. Understanding these details is essential for stakeholders evaluating the integration of this intermediate into their broader supply chain strategy.
Q: What is the overall yield and purity of this synthetic route?
A: According to patent CN101157654A, the seven-step process achieves a total output rate greater than 18.4 percent, with the final product purity exceeding 99.7 percent, making it highly suitable for API manufacturing.
Q: Why is the Hofmann degradation step critical in this process?
A: The Hofmann degradation converts the amide intermediate into the primary amine. This step is optimized to occur at controlled low temperatures (0-5°C) to prevent side reactions and ensure high selectivity for the 3-amino position.
Q: Are the raw materials for this process readily available?
A: Yes, the process utilizes simple and easy-to-source industrial chemicals such as acetone, sodium methoxide, and methyl formate, which significantly reduces supply chain risks compared to routes requiring specialized nitro-compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-3-Amino-4-Picoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against HIV and other viral infections. Our technical team has extensively analyzed the route described in CN101157654A and possesses the expertise to scale this diverse pathway from 100 kgs to 100 MT/annual commercial production. We are committed to delivering this key pharmaceutical intermediate with stringent purity specifications, ensuring that every batch meets the rigorous standards required for API synthesis. Our rigorous QC labs employ advanced analytical techniques to verify the impurity profile and structural integrity of the product, providing our partners with the confidence they need to proceed with their own manufacturing processes without delay or quality concerns.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate how our optimized production capabilities can enhance your supply chain efficiency. Whether you are looking to secure a long-term supply agreement or need support in scaling up a new drug candidate, our team is ready to provide the technical and logistical support necessary to ensure your project's success. Contact us today to discuss how we can support your production needs with reliability and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
