Advanced Synthesis of Chiral Diamine Ruthenium Catalysts for Industrial Asymmetric Catalysis
Introduction to Next-Generation Chiral Catalyst Manufacturing
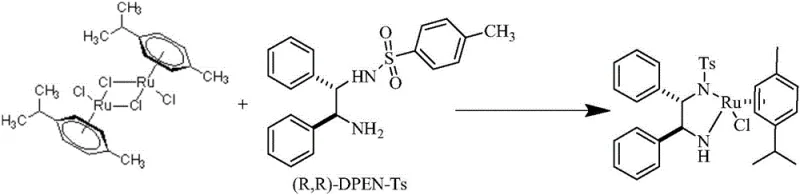
The landscape of asymmetric catalysis is undergoing a significant transformation driven by the demand for higher efficiency and purity in the synthesis of active pharmaceutical ingredients and fine chemicals. A pivotal advancement in this domain is detailed in patent CN114560890A, which discloses a robust preparation method for a chiral diamine coordination metal catalyst, specifically {[(1R,2R)-(-)-2-amino-1,2-diphenylethyl](p-toluenesulfonyl)amino)}(p-cymene)ruthenium chloride. This technology addresses critical bottlenecks in traditional catalyst manufacturing by introducing a novel mixed-solvent system coupled with precise alkali reagent coordination. For R&D directors and procurement specialists seeking a reliable chiral catalyst supplier, this innovation represents a paradigm shift from labor-intensive, low-yield processes to a streamlined, high-output manufacturing protocol. The ability to produce high-purity catalysts without the need for post-reaction recrystallization not only accelerates time-to-market but also drastically simplifies the supply chain for downstream asymmetric hydrogenation applications.
The strategic value of this patent lies in its ability to overcome the inherent limitations of prior art synthesis routes. Conventional methods often suffer from complicated preparation processes, low product purity, and significant metal consumption due to the necessity of multiple purification steps. By contrast, the methodology outlined in CN114560890A utilizes a p-cymene ruthenium chloride dimer as a metal precursor, reacting it directly with the chiral ligand under mild heating conditions. This approach ensures that the product precipitates directly in a crystal form, eliminating the need for further recrystallization purification. Consequently, the preparation process is significantly shortened, leading to substantial cost savings and a marked improvement in production efficiency. For industries relying on cost reduction in pharmaceutical intermediate manufacturing, adopting this synthesis route offers a tangible competitive advantage by optimizing resource utilization and minimizing waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral diamine coordination metal catalysts has been plagued by inefficiencies that hinder large-scale commercial viability. The background section of the patent highlights that existing techniques typically involve a direct coordination reaction between a ligand and a metal compound, a process that is notoriously complicated and prone to generating impurities. Specifically, the crude products obtained from these traditional methods often contain dechlorinated chiral substances, resulting in low overall purity. To meet the stringent quality standards required for asymmetric catalysis, these crude materials necessitate further recrystallization purification. This additional step is not merely a technical inconvenience; it is a major economic burden that results in reduced efficiency, diminished product yield, and increased consumption of valuable precious metals. In comparative examples provided within the patent data, conventional direct reaction methods yielded only 50.3% of the target product, demonstrating the severe material losses associated with legacy technologies.
The Novel Approach
The innovative strategy presented in this patent fundamentally reengineers the reaction environment to maximize yield and purity simultaneously. Instead of relying on a single solvent or harsh conditions, the novel approach employs a sophisticated mixed solvent system where Solution A (containing the ligand) and Solution B (containing the metal precursor) are prepared in distinct solvent environments before being combined. Solution A utilizes polar solvents like ethanol or methanol to ensure complete dissolution and activation of the ligand, while Solution B employs non-polar solvents such as petroleum ether or toluene to create an optimal reaction medium for the metal complex. By dropwise adding the ligand solution into the metal precursor solution in the presence of an alkali reagent, the reaction proceeds smoothly under mild temperatures ranging from 30°C to 75°C. This controlled addition and specific solvent pairing promote the immediate precipitation of the target catalyst in a high-purity crystalline form, effectively bypassing the need for energy-intensive recrystallization steps and achieving yields consistently above 95%.
Mechanistic Insights into Mixed-Solvent Coordinated Synthesis
Understanding the underlying chemical mechanisms is crucial for R&D teams aiming to replicate or scale this process effectively. The core of this invention's success lies in the synergistic effect of the mixed solvent system and the alkali reagent. The patent elucidates that the polar solvent in Solution A plays a vital role in facilitating the deprotonation of the ligand, (1R,2R)-N-(2-amino-1,2-diphenylethyl)p-toluenesulfonamide. Under the influence of the alkali reagent, the ligand loses a proton to form a reactive anionic species, which is significantly more nucleophilic and ready to coordinate with the ruthenium center. This deprotonation step is critical for initiating the coordination bond formation efficiently. Without the appropriate polar environment provided by solvents like ethanol, the ligand might remain protonated or poorly solvated, leading to sluggish reaction kinetics and incomplete conversion.
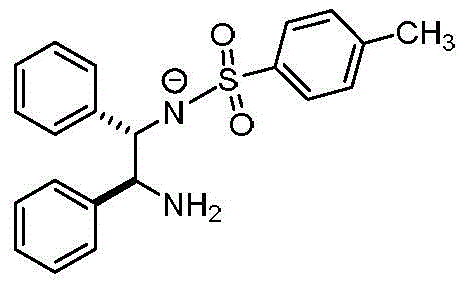
Furthermore, the inclusion of a non-polar solvent in Solution B serves a dual purpose: it modulates the overall polarity of the reaction mixture and creates an environment favorable for the precipitation of the aromatic target product. As the reaction progresses and the chiral diamine coordination metal catalyst forms, its solubility in the mixed solvent system decreases, prompting it to crystallize out of the solution. This phenomenon is driven by the fact that the product is an aromatic hydrocarbon substance, which is less soluble in the adjusted polarity of the mixed solvent compared to the reactants. The alkali reagent, such as sodium hydroxide or potassium hydroxide, further accelerates the reaction by neutralizing acidic by-products (like HCl generated from the ruthenium dimer) and promoting the dechlorination of the metal precursor. This ensures that the ruthenium center is available for coordination with the deprotonated ligand, driving the equilibrium towards the formation of the desired catalyst.
Impurity control is another critical aspect managed by this mechanistic design. In conventional single-solvent systems, side reactions such as the formation of dechlorinated chiral ligands or incomplete coordination complexes are common, leading to a messy crude product profile. However, the specific ratio of polar to non-polar solvents described in the patent (where the volume of organic solvent B is 1.0 to 3.0 times that of organic solvent A) creates a selective environment that suppresses these side pathways. The rapid precipitation of the correct product essentially removes it from the reactive solution, preventing it from undergoing further degradation or participating in reverse reactions. This "crystallization-driven synthesis" ensures that the final product possesses a purity of greater than 98.5% directly after filtration, a level of quality that typically requires multiple purification cycles in other methods. For manufacturers of high-purity asymmetric catalysts, this intrinsic purity control is a game-changer that simplifies quality assurance protocols.
How to Synthesize Chiral Diamine Ruthenium Catalyst Efficiently
The practical implementation of this synthesis route involves a carefully orchestrated sequence of mixing, heating, and separation steps designed to maximize safety and yield. The process begins with the preparation of two distinct solution systems under anaerobic conditions to prevent oxidation of the sensitive metal centers or ligands. Solution A is formed by dissolving the chiral ligand in a polar solvent like absolute ethanol at room temperature until clear. Simultaneously, Solution B is prepared by suspending the p-cymene ruthenium chloride dimer in a non-polar solvent like petroleum ether at elevated temperatures (30-60°C), followed by the addition of a stoichiometric amount of an alkali reagent such as sodium hydroxide. The critical operational step involves the dropwise addition of Solution A into Solution B over a period of 15 to 60 minutes while maintaining vigorous stirring. This controlled addition prevents local concentration spikes that could lead to oligomerization or impurity formation. Following the addition, the mixture is stirred at 30-75°C for 3 to 8 hours to ensure complete reaction. The detailed standardized synthesis steps for this procedure are outlined below.
- Prepare Solution A by dissolving the chiral ligand (1R,2R)-N-(2-amino-1,2-diphenylethyl)p-toluenesulfonamide in a polar organic solvent such as ethanol under anaerobic conditions.
- Prepare Solution B by mixing p-cymene ruthenium chloride dimer with a non-polar organic solvent like petroleum ether at 30-60°C, then add an alkali reagent such as sodium hydroxide.
- Dropwise add Solution A into Solution B under stirring at 30-75°C for 3-8 hours, then separate the solid product and vacuum dry to obtain the target catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis method offers profound strategic benefits that extend beyond simple technical metrics. The primary advantage is the drastic simplification of the production workflow, which directly translates to cost reduction in fine chemical manufacturing. By eliminating the recrystallization step, manufacturers save significant amounts of time, energy, and solvent resources. Recrystallization is traditionally one of the most material-intensive unit operations in fine chemistry, often requiring large volumes of hot solvents and subsequent cooling cycles. Removing this step not only lowers utility costs but also reduces the facility's environmental footprint by minimizing solvent waste. Additionally, the high yield of over 95% means that less raw material is required to produce the same amount of finished catalyst, optimizing the cost of goods sold (COGS) and improving margin potential for downstream applications.
- Cost Reduction in Manufacturing: The elimination of the recrystallization purification step represents a major operational saving. In traditional processes, recrystallization can account for a significant portion of production time and solvent consumption. By achieving high purity directly upon precipitation, this method removes the need for these expensive downstream processing units. Furthermore, the high reaction yield minimizes the loss of expensive ruthenium metal, which is a critical cost driver in catalyst production. The use of common, inexpensive solvents like petroleum ether and ethanol further contributes to a leaner cost structure compared to methods requiring exotic or highly purified solvent systems.
- Enhanced Supply Chain Reliability: The robustness of this synthesis route enhances supply chain stability by reducing the complexity of the manufacturing process. Fewer processing steps mean fewer points of failure and a shorter overall production cycle time. The use of readily available raw materials, such as p-cymene ruthenium chloride dimer and common alkali reagents, ensures that supply disruptions are unlikely. Moreover, the mild reaction conditions (30-75°C) allow the process to be run in standard glass-lined or stainless steel reactors without the need for specialized high-pressure or cryogenic equipment, making it easier for contract manufacturing organizations (CMOs) to adopt and scale the technology quickly to meet market demand.
- Scalability and Environmental Compliance: The patent data explicitly demonstrates the scalability of this method, with successful examples running at nearly 1 kg scale (Example 8) while maintaining consistent quality. This proves the feasibility of commercial scale-up of complex organometallic compounds using this route. From an environmental perspective, the process avoids the use of dangerous chemicals and operates under relatively safe conditions. The reduction in solvent usage due to the lack of recrystallization aligns with green chemistry principles, helping companies meet increasingly strict environmental regulations and sustainability goals. The anaerobic condition requirement is standard in organometallic chemistry and can be easily managed with nitrogen blanketing in industrial settings.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this chiral diamine ruthenium catalyst. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these nuances is essential for evaluating the feasibility of integrating this catalyst into your existing synthetic workflows or supply chain strategies.
Q: Why is a mixed solvent system critical for this catalyst synthesis?
A: The patent data indicates that using a mixed solvent system comprising a polar solvent (Solution A) and a non-polar solvent (Solution B) significantly improves selectivity and yield compared to single solvents. The polar solvent facilitates the deprotonation of the ligand to form a reactive anion, while the non-polar solvent adjusts the overall polarity of the reaction environment, promoting the precipitation of the aromatic target product in a crystalline form without the need for further recrystallization.
Q: What are the purity and yield advantages of this novel method?
A: Experimental data from the patent demonstrates that this method achieves yields exceeding 95.0% and purity levels greater than 98.5% directly after filtration and drying. In contrast, conventional direct coordination methods often result in yields as low as 50.3% and require additional recrystallization steps to reach similar purity levels, thereby increasing production costs and reducing overall efficiency.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. The patent includes examples of scaling up to nearly 1 kg batches (Example 8) with consistent high yields (96.4%) and purity (98.9%). Furthermore, the reaction conditions are mild (30-75°C), do not involve dangerous chemicals, and utilize common industrial solvents, making it highly suitable for commercial scale-up of complex organometallic compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Diamine Ruthenium Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance catalysts play in the efficient synthesis of complex pharmaceutical intermediates and fine chemicals. Our team of expert chemists has thoroughly analyzed the technological potential of the preparation method described in CN114560890A and is fully equipped to translate this laboratory-scale innovation into industrial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-quality catalysts. Our state-of-the-art facilities are designed to handle sensitive organometallic syntheses under strict anaerobic conditions, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for asymmetric catalysis applications.
We invite global partners to collaborate with us to optimize their supply chains and reduce manufacturing costs through the adoption of this advanced catalyst technology. Whether you are looking to secure a long-term supply of high-purity asymmetric catalysts or need assistance in adapting this synthesis route for your specific process needs, our technical team is ready to support you. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your production volumes. By engaging with us, you can access specific COA data, route feasibility assessments, and comprehensive project timelines that will help you make informed decisions and accelerate your path to commercialization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
