Advanced Manufacturing of Isoxazolin-3-yl Acylbenzene Intermediates for High-Efficiency Herbicide Production
Advanced Manufacturing of Isoxazolin-3-yl Acylbenzene Intermediates for High-Efficiency Herbicide Production
The chemical landscape of agrochemical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable synthetic routes. Patent CN1139578C represents a significant technological breakthrough in the preparation of isoxazolin-3-yl acylbenzene derivatives, which serve as critical precursors for potent herbicidally active compounds. This proprietary methodology addresses long-standing challenges in the synthesis of 3-heterocyclyl-substituted benzoyl derivatives, offering a streamlined pathway that enhances both yield and purity. By leveraging novel intermediates and optimized reaction conditions, this process provides a robust framework for the large-scale production of high-value crop protection agents. The technology is particularly relevant for manufacturers seeking to optimize their supply chains for complex heterocyclic structures.
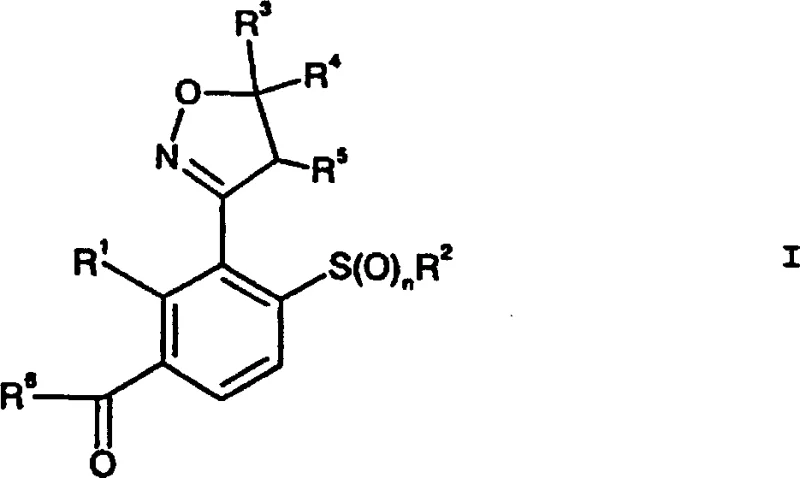
The core innovation lies in the strategic construction of the isoxazoline ring coupled with a highly functionalized benzene scaffold. Unlike traditional methods that often suffer from low atom economy and cumbersome purification steps, this approach utilizes a sequence of chemoselective transformations. The process begins with the preparation of specific oxime intermediates, followed by cyclization, selective reduction, and thio-functionalization. Each step has been meticulously engineered to minimize by-product formation and maximize throughput. For R&D directors and process chemists, understanding the nuances of this pathway is essential for evaluating its potential integration into existing manufacturing portfolios. The ability to produce these intermediates with high fidelity ensures consistent quality in the final herbicidal formulations, a key metric for regulatory compliance and market success.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described in WO 98/31681, have historically plagued the industry with inefficiencies that hinder commercial viability. These conventional routes typically involve an excessive number of synthetic steps, each introducing potential points of failure and yield loss. The cumulative effect of these multi-step sequences results in a significantly low overall yield of the desired final product relative to the starting materials. Furthermore, the intermediates required in these older processes, such as specific 3-(4,5-dihydroisoxazol-3-yl)benzoic acid derivatives, are notoriously difficult to obtain and often require complex, multi-step preparations themselves. This complexity not only drives up the cost of goods sold but also introduces significant supply chain vulnerabilities. Additionally, some prior methods rely on harsh reaction conditions or expensive reagents that are not conducive to safe, large-scale industrial operations, creating barriers to entry for many potential manufacturers.
The Novel Approach
In stark contrast, the process disclosed in CN1139578C offers a paradigm shift by drastically reducing the number of process steps required to reach the target molecule. By utilizing selected novel intermediates, specifically the formula VI aniline and formula X bromobenzene derivatives, the synthesis becomes far more direct and economical. The overall yield of the final formula I compound, as well as the key intermediate X, is substantially higher when calculated based on the starting materials used. This improvement is not merely incremental; it represents a fundamental optimization of the synthetic logic. Moreover, the individual steps within this new process are designed to produce intermediates in high yields, ensuring that material is not lost at critical junctures. The use of basic, readily available chemicals as starting materials further enhances the economic attractiveness, allowing for sourcing from multiple suppliers and ensuring supply continuity even in volatile market conditions.
Mechanistic Insights into Chemoselective Hydrogenation and Carbonylation
A pivotal aspect of this technology is the chemoselective hydrogenation step, where the aromatic nitro group is reduced to an amine in the presence of the sensitive isoxazoline ring. Historically, catalytic hydrogenation of nitro compounds is well-known, but the presence of the isoxazoline N-O bond poses a significant challenge, as it is prone to cleavage under standard hydrogenation conditions using catalysts like Raney nickel or palladium. However, this patent reveals that by carefully selecting reaction conditions—specifically using platinum or palladium on activated carbon at temperatures between 0°C and 50°C—the nitro group can be reduced exclusively without disrupting the heterocyclic ring. This selectivity is crucial for maintaining the structural integrity of the molecule. The use of sulfur or selenium-doped palladium catalysts can further enhance this selectivity, demonstrating a deep understanding of catalyst-substrate interactions that prevents unwanted side reactions and ensures high purity of the resulting aniline intermediate.
Another mechanistic highlight is the palladium-catalyzed carbonylation step used to couple the bromobenzene derivative with hydroxypyrazoles. Conventional methods, such as those in EP-A 344 775, often require extremely high carbon monoxide pressures, up to 150 kg/cm², necessitating specialized and expensive high-pressure equipment. This new process surprisingly achieves excellent yields at significantly lower pressures, typically between 5-20 kg/cm², and preferably 5-8 kg/cm². This reduction in pressure not only lowers capital expenditure requirements but also enhances operational safety. Furthermore, the catalyst system is designed such that the palladium precipitates as elemental metal with a specific pore size (1-10 μm), allowing for facile recovery via simple filtration. This eliminates the need for complex and costly concentration steps to recover the precious metal from the reaction solution, thereby reducing waste and improving the overall environmental profile of the manufacturing process.
How to Synthesize Isoxazolin-3-yl Acylbenzene Efficiently
The synthesis of these high-value agrochemical intermediates requires precise control over reaction parameters to ensure reproducibility and safety. The process generally involves a sequence starting with the oximation of substituted nitro-o-xylene derivatives using organic nitrites in dipolar aprotic solvents like DMF or NMP at low temperatures ranging from -60°C to -20°C. This is followed by a cyclization step with alkenes to form the isoxazoline core. Subsequent steps involve the chemoselective reduction of the nitro group, thioalkylation using dialkyl disulfides and copper catalysts, bromination, and oxidation to the sulfone. The final coupling is achieved through carbonylation. Each stage demands strict adherence to stoichiometric ratios and temperature controls to avoid by-product formation, such as dimerization during oximation or over-bromination. The detailed standardized synthesis steps for implementing this protocol are outlined below.
- Perform oximation of substituted nitro-o-xylene using organic nitrite and base at low temperatures (-60°C to -20°C) to form benzaldoxime intermediates.
- Execute cyclization with alkenes via nitrile oxide intermediates to construct the isoxazoline ring system with high stereocontrol.
- Conduct chemoselective hydrogenation of the nitro group to an amine using Pt/C or Pd/C catalysts without cleaving the isoxazoline N-O bond.
- Transform the aniline derivative into a thioether using dialkyl disulfide and copper catalyst, followed by bromination and oxidation to the sulfone.
- Finalize synthesis via palladium-catalyzed carbonylation with hydroxypyrazoles under moderate CO pressure (5-20 kg/cm²) to yield the final acylbenzene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the substantial cost reduction potential derived from the simplification of the manufacturing process. By eliminating the need for hard-to-obtain intermediates and reducing the total number of unit operations, the operational overhead is significantly lowered. The reliance on basic, commodity chemicals as starting materials means that raw material costs are stable and predictable, shielding the production budget from the volatility associated with specialty reagents. Furthermore, the ability to recover and reuse expensive palladium catalysts directly from the reaction mixture translates into direct savings on precious metal consumption, a major cost driver in fine chemical synthesis.
- Cost Reduction in Manufacturing: The streamlined nature of this process directly impacts the bottom line by minimizing waste and maximizing material throughput. The elimination of complex purification steps, such as those required to remove polybrominated by-products in conventional aniline bromination, reduces solvent usage and energy consumption. Additionally, the operation at lower pressures for the carbonylation step removes the necessity for investing in high-specification pressure vessels, thereby lowering capital depreciation costs. The high yields reported for each individual step ensure that less raw material is needed to produce the same amount of final product, effectively lowering the cost per kilogram of the active intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. Unlike processes that depend on proprietary or scarce reagents, this method utilizes chemicals like nitro-o-xylene, alkyl nitrites, and dialkyl disulfides, which are produced by numerous global suppliers. This diversification of the supply base mitigates the risk of shortages and allows for competitive bidding on raw materials. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in feedstock quality, ensuring consistent production schedules and reliable delivery timelines to downstream customers.
- Scalability and Environmental Compliance: From a scalability perspective, the process is designed with industrial application in mind. The exothermic nature of certain steps, such as the oximation, is managed through controlled addition rates and cooling, making it safe to scale from pilot plants to multi-ton reactors. The environmental footprint is minimized through the avoidance of alkali metal hypohalites and the efficient recovery of catalysts and solvents. The reduction in hazardous waste generation simplifies compliance with increasingly stringent environmental regulations, reducing the administrative and financial burden associated with waste disposal and permitting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy and reliability for decision-makers. Understanding these specifics is vital for assessing the feasibility of adopting this route for your specific production needs.
Q: How does this process improve upon previous methods like WO 98/31681?
A: The process described in CN1139578C significantly reduces the number of synthetic steps compared to WO 98/31681. It utilizes readily available basic chemicals and achieves higher overall yields for the final formula I compounds and key intermediates, making it more economically viable for industrial scale-up.
Q: What are the critical safety parameters for the carbonylation step?
A: Unlike prior art requiring pressures up to 150 kg/cm², this optimized process operates safely at reduced pressures between 5-20 kg/cm², preferably 5-8 kg/cm². This reduction eliminates the need for expensive high-pressure vessels while maintaining reaction efficiency and yield.
Q: How is catalyst recovery handled in the final coupling step?
A: The palladium catalyst precipitates predominantly as elemental palladium with a pore size of 1-10 μm. This allows for easy separation via simple filtration, avoiding complex concentration steps and enabling low-cost recovery and recycling of the precious metal.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoxazolin-3-yl Acylbenzene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation agrochemicals. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex heterocyclic chemistry, including the sensitive isoxazoline motifs described in CN1139578C, positions us as a strategic partner for your long-term supply needs.
We invite you to engage with our technical procurement team to discuss how we can tailor this manufacturing process to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this optimized route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your supply chain.
