Advanced Synthesis of Polynitrogen Heterocycles for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust pathways for constructing complex nitrogen-containing scaffolds, which serve as critical backbones for numerous bioactive molecules. Patent CN101619064A introduces a groundbreaking methodology for the synthesis of novel polynitrogen heterocyclic rings, specifically targeting the efficient production of bicyclic polynitrogen compounds. This technology addresses the persistent challenges in medicinal chemistry by providing a versatile route that encompasses aliphatic, aromatic, and heterocyclic variants. The core innovation lies in the strategic utilization of inexpensive and readily available starting materials, such as various maleic acid esters and nitrogen-substituted glycine or pyridine dicarboxylic acids. By streamlining the synthetic sequence into a manageable four or five-step reaction process, this method significantly enhances the feasibility of producing high-purity pharmaceutical intermediates. For R&D directors and process chemists, this represents a pivotal shift towards more sustainable and economically viable manufacturing protocols that do not compromise on structural complexity or purity standards.
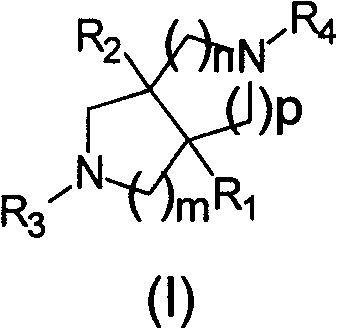
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polynitrogen heterocycles has been plagued by significant inefficiencies that hinder commercial viability and rapid drug development. Conventional synthetic routes are often characterized by excessive length, requiring numerous steps that cumulatively degrade overall yield and increase operational complexity. The aftertreatment processes associated with these traditional methods are frequently loaded down with trivial difficulties, involving complex purification sequences to remove stubborn impurities and byproducts. Furthermore, the raw material costs for many established pathways are prohibitively high, relying on specialized reagents that are difficult to source in bulk quantities. From an environmental and operational standpoint, these older methods generate substantial amounts of three wastes, leading to significant pollution and increased disposal costs. The combination of low yields, high costs, and environmental burden creates a bottleneck for supply chain heads who require consistent, scalable, and cost-effective sources of critical intermediates for downstream API production.
The Novel Approach
In stark contrast to the cumbersome traditional methodologies, the novel approach detailed in the patent offers a streamlined and highly efficient alternative that resolves these longstanding industrial pain points. This method achieves the synthesis of multi-series polynitrogen heterocycle pharmaceutical intermediates through a short-circuit line that begins with simply available raw materials. The process is engineered for low cost and high yield, ensuring that the economic barriers to entry are significantly lowered for manufacturers. Crucially, the reaction conditions are simple and easy to control, reducing the risk of batch failure and ensuring consistent quality across large-scale production runs. The handling and processing steps are simplified, minimizing the need for complex equipment or hazardous operational procedures. By drastically reducing the generation of three wastes, this approach aligns with modern green chemistry principles, offering a practical and feasible synthesis route that supports sustainable industrial production without sacrificing the structural diversity required for advanced drug discovery programs.
Mechanistic Insights into Maleic Ester-Based Cyclization and Reduction
The core of this synthetic strategy relies on a sophisticated yet robust sequence of cyclization and reduction reactions that construct the bicyclic framework with high precision. The initial phase involves the reaction of maleic acid esters with N-substituted glycine, where the molar ratio is carefully optimized, typically ranging from 3:1 to 1:1, with a recommended substitution of 1.5 to 1 molar weight of the maleic ester. This reaction is conducted in solvents such as toluene, benzene, dimethylbenzene, DMF, DMSO, or DMA at temperatures between 80 and 150 degrees Celsius, facilitating the formation of the initial diester intermediate. Subsequent steps involve the reaction of this diester with benzylamine in solvents like ethanol or methanol at 50 to 90 degrees Celsius to form the bicyclic lactam structure. The precise control of stoichiometry and temperature during these cyclization events is critical for minimizing side reactions and ensuring the formation of the desired regioisomers, which is essential for maintaining the integrity of the final pharmaceutical intermediate.
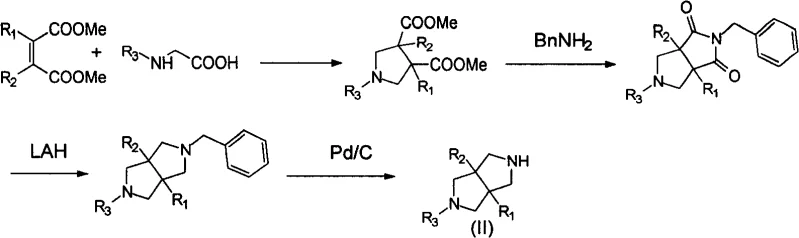
Following the construction of the bicyclic core, the process employs powerful reduction strategies to achieve the final polynitrogen heterocycle structure. The reduction of the carbonyl groups is typically executed using lithium aluminium hydride (LAH), with a molar ratio of carbonyl compound to LAH optimized at 1:3 to 1:6 to ensure complete conversion. This step is often followed by a catalytic hydrogenation process using catalysts such as Pd/C or Pd(OH)2/C to remove protecting groups like benzyl moieties. The impurity control mechanism is inherent in the simplicity of the reaction pathway; by avoiding complex transition metal catalyzed cross-couplings that often leave trace metal residues, the process naturally yields a cleaner crude product. The final purification is straightforward, often requiring only standard extraction and distillation techniques, which allows for the production of high-purity compounds with GC purity exceeding 98.0%. This level of purity is vital for R&D directors who need to ensure that impurity profiles do not complicate subsequent toxicological studies or regulatory filings.
How to Synthesize 3,7-Diazabicyclo[3.3.0]Octanes Efficiently
The synthesis of specific targets such as 3,7-diazabicyclo[3.3.0]octanes exemplifies the practical application of this patented technology in a real-world manufacturing context. The process begins with the cyclization of sarcosine and dimethyl maleate in toluene under reflux, followed by reaction with benzylamine to form the bridged lactam. Subsequent reduction with LAH in anhydrous THF and final hydrogenation over Pd/C yields the target amine with high efficiency. This route demonstrates the versatility of the method in handling different substituents while maintaining high yields and operational simplicity. For process chemists looking to implement this technology, the detailed standardized synthesis steps provided in the patent serve as a robust foundation for scale-up activities. The following guide outlines the critical operational parameters required to replicate this success in a commercial setting.
- Cyclization of N-substituted amino acid with substituted maleic acid ester to form substituted pyrrolidine diester.
- Reaction of the tetramethyleneimine diester with organic amine to create a bridged ring tetramethyleneimine lactam.
- Reduction of the lactam using lithium aluminium hydride (LAH) followed by hydrogenation deprotection to yield the final 3,7-diazabicyclo[3.3.0]octane structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates into tangible strategic advantages that extend beyond mere technical feasibility. The primary benefit lies in the substantial cost reduction in pharmaceutical intermediates manufacturing driven by the use of commodity chemicals. By utilizing inexpensive and readily available raw materials like maleic acid esters and glycine derivatives, the direct material cost is significantly lowered compared to routes relying on exotic or proprietary reagents. This cost efficiency is compounded by the simplified processing requirements, which reduce labor and utility consumption during production. The elimination of complex purification steps further drives down operational expenditures, allowing for more competitive pricing structures in the global market. These factors collectively enhance the margin potential for downstream API manufacturers, making this intermediate a highly attractive option for cost-sensitive drug development projects.
- Cost Reduction in Manufacturing: The economic advantage of this process is rooted in the fundamental chemistry that avoids expensive catalysts and reagents. By eliminating the need for transition metal catalysts that require costly removal steps, the process inherently reduces the financial burden associated with raw material procurement and waste treatment. The high yield of the reaction sequence means that less raw material is wasted per unit of product, maximizing the efficiency of every kilogram of input. Furthermore, the use of common solvents like toluene and ethanol simplifies solvent recovery and recycling processes, contributing to additional long-term savings. This qualitative improvement in cost structure ensures that the supply of these intermediates remains stable and affordable even during fluctuations in the broader chemical market.
- Enhanced Supply Chain Reliability: Supply chain continuity is critically dependent on the availability of raw materials and the robustness of the manufacturing process. Since this method relies on widely available commodity chemicals, the risk of supply disruption due to raw material scarcity is drastically minimized. The simplicity of the reaction control also means that the manufacturing process is less prone to deviations that could lead to batch rejections or delays. This reliability is essential for supply chain heads who must guarantee consistent delivery schedules to API manufacturers. The ability to source materials from multiple vendors and the ease of scaling the reaction ensure that production can be ramped up quickly to meet surging demand without compromising on quality or lead time.
- Scalability and Environmental Compliance: The design of this synthetic route inherently supports commercial scale-up of complex pharmaceutical intermediates. The reaction conditions are mild and easy to control, which reduces the engineering challenges associated with transferring the process from the laboratory to the pilot plant and finally to full commercial production. Additionally, the reduction in three wastes aligns with increasingly stringent environmental regulations, reducing the compliance burden on manufacturing facilities. The green chemistry aspects of the process not only mitigate environmental risk but also enhance the corporate social responsibility profile of the supply chain. This makes the intermediate more attractive to global pharmaceutical companies that prioritize sustainable sourcing in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this polynitrogen heterocycle synthesis technology. These answers are derived directly from the patent specifications and are intended to provide clarity on the process capabilities and limitations. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this intermediate into their existing drug development pipelines. The responses cover aspects ranging from raw material sourcing to the specific advantages of the reduction steps employed in the synthesis.
Q: What are the primary raw materials for this polynitrogen heterocycle synthesis?
A: The process utilizes inexpensive and readily available raw materials such as various maleic acid esters and nitrogen-substituted glycine or pyridine dicarboxylic acids, significantly lowering the entry barrier for production.
Q: How does this method improve upon conventional synthesis routes?
A: Unlike traditional methods which often involve long synthetic routes, complex aftertreatment, and high raw material costs, this patented approach offers a shorter 4 to 5-step reaction sequence with simple processing and high yields.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method is designed for ease of scale-up. The reaction conditions are easy to control, and the purification process is simple, making it highly feasible for green chemical industry applications and commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polynitrogen Heterocycles Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthesis pathways play in accelerating drug discovery and development. Our team of expert process chemists has extensively evaluated the technology described in CN101619064A and is fully prepared to support its implementation at any scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from clinical trials to market launch. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of polynitrogen heterocycles meets the highest international standards. We are committed to providing a reliable supply of high-purity pharmaceutical intermediates that empower your R&D efforts.
We invite you to collaborate with us to optimize your supply chain and reduce your overall development costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project requirements. By partnering with us, you gain access to specific COA data and comprehensive route feasibility assessments that will help you make informed decisions about your manufacturing strategy. Contact us today to discuss how we can support your need for reliable polynitrogen heterocycles supplier services and drive your project forward with confidence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
