Unlocking Efficient Production of Critical Pyridine Intermediates for Global Pharmaceutical Supply Chains
Unlocking Efficient Production of Critical Pyridine Intermediates for Global Pharmaceutical Supply Chains
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and scalable synthetic routes. A pivotal advancement in this domain is detailed in patent CN1042705A, which discloses a novel derivatization method for producing 3,5-dimethyl-4-methoxypyridine derivatives. These compounds serve as extremely valuable building blocks in the synthesis of ATDase inhibitors, a class of compounds with significant therapeutic potential. The patent outlines a strategic shift away from cumbersome traditional methodologies, proposing a streamlined pathway that begins with 3,5-dimethyl-4-methoxy-2-cyanopyridine. By leveraging catalytic hydrogenation and subsequent functional group transformations, this technology offers a robust solution for generating high-purity intermediates such as 3,5-dimethyl-4-methoxy-2-aminomethyl pyridine. For R&D directors and procurement specialists alike, understanding the nuances of this patented process is crucial for optimizing supply chains and reducing the cost of goods sold in complex API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted pyridines like 3,5-dimethyl-4-methoxy-2-hydroxymethylpyridine relied heavily on starting materials that are increasingly difficult to source and expensive to produce. Prior art, such as US-A4472409, describes a multi-step sequence initiating with 2,3,5-trimethylpyridine. This conventional route necessitates the formation of an N-oxide compound, followed by nitration to introduce a nitro group, and subsequently exchanging the nitro group for a methoxy group using sodium methylate. This sequence is not only chemically intensive, requiring seven distinct operational steps to reach the necessary precursors, but it also suffers from poor overall yields and the handling of hazardous nitrating agents. Furthermore, the starting material, 2,3,5-trimethylpyridine, is often obtained through low-yield reactions involving 3,5-lutidine and lithium methide at cryogenic temperatures, creating a significant bottleneck in the supply chain. These factors collectively result in a process that is economically inefficient and operationally risky for large-scale commercial production.
The Novel Approach
In stark contrast, the methodology presented in CN1042705A introduces a paradigm shift by utilizing 3,5-dimethyl-4-methoxy-2-cyanopyridine as the primary starting material. This approach bypasses the problematic N-oxide formation and nitration steps entirely. The core innovation lies in the direct conversion of the cyano group into an aminomethyl functionality, which serves as a versatile handle for further derivatization. As illustrated in the structural representation below, the starting material possesses a stable pyridine core with strategically placed methyl and methoxy groups that facilitate selective reactivity.
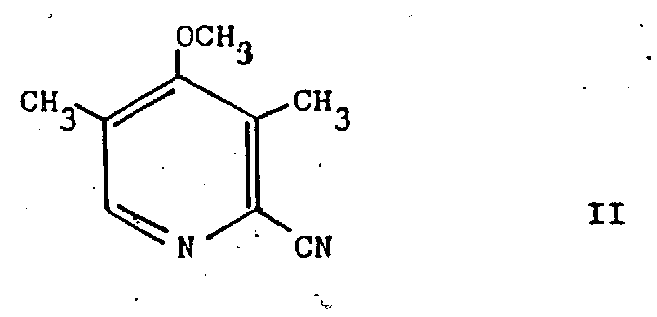
This novel route allows for the direct synthesis of 3,5-dimethyl-4-methoxy-2-aminomethyl pyridine, a key intermediate that can be subsequently converted into hydroxymethyl or chloromethyl derivatives with high efficiency. By shortening the synthetic sequence and utilizing more accessible raw materials derived from 3,5-lutidine oxidation and cyanation, this method drastically reduces the complexity of the manufacturing process. For procurement managers, this translates to a more reliable sourcing strategy, as the dependency on scarce trimethylpyridine derivatives is eliminated, thereby enhancing the resilience of the supply chain against raw material shortages.
Mechanistic Insights into Catalytic Hydrogenation and Diazotization
The heart of this synthetic strategy involves a highly selective catalytic hydrogenation step, which converts the nitrile group of the starting cyanopyridine into a primary amine. This transformation is critical because nitriles are prone to over-reduction or coupling reactions that can lead to secondary amine impurities. The patent specifies the use of heterogeneous catalysts such as Raney nickel or Palladium on Carbon (Pd/C) under hydrogen pressure ranging from 1 to 5 atmospheres. Crucially, the reaction environment is modulated by the addition of ammonia or mineral acids. When using Raney nickel, the presence of anhydrous ammonia acts as a competitive inhibitor, preventing the newly formed primary amine from reacting with intermediate imines, thus ensuring high selectivity for the primary amine product. Alternatively, when using Pd/C, the addition of acid protonates the amine as it forms, similarly blocking side reactions. This mechanistic control is vital for maintaining the purity profile required for pharmaceutical applications, ensuring that the impurity spectrum remains manageable without extensive downstream purification.
Following the hydrogenation, the resulting 3,5-dimethyl-4-methoxy-2-aminomethyl pyridine undergoes a classic diazotization-hydrolysis sequence to install the hydroxymethyl group. This step involves treating the amine with sodium nitrite in an acidic aqueous medium, typically acetic acid, at controlled low temperatures between -5°C and 10°C. The formation of the diazonium salt is transient and unstable, requiring immediate hydrolysis to yield the alcohol. The structural integrity of the pyridine ring is maintained throughout this acidic treatment, demonstrating the robustness of the substitution pattern. The resulting amine intermediate, depicted below, is a novel compound in itself and represents a pivotal junction in the synthesis, allowing for divergence into either alcohol or chloride derivatives depending on the final API requirements.
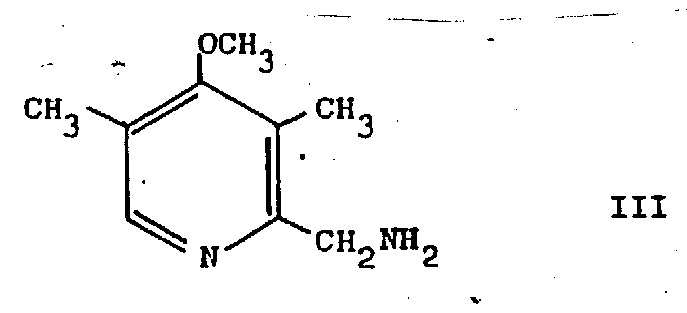
The final conversion to the chloromethyl derivative is achieved using thionyl chloride, a standard chlorinating agent that reacts efficiently with the hydroxymethyl group. The entire sequence from cyanide to chloride is designed to minimize isolation steps; in preferred embodiments, the intermediate amine is not isolated but carried forward directly into the diazotization pot. This telescoping of steps not only saves time but also reduces solvent consumption and waste generation, aligning with green chemistry principles. For R&D teams, understanding these mechanistic details allows for better troubleshooting during tech transfer, particularly in controlling exotherms during the diazotization and managing the evolution of gases during the thionyl chloride reaction.
How to Synthesize 3,5-Dimethyl-4-Methoxypyridine Derivatives Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity. The process begins with the preparation of the hydrogenation mixture, where the cyanopyridine substrate is dissolved in a solvent such as methanol saturated with ammonia. The choice of solvent is critical; alcohols like methanol or ethanol provide excellent solubility for the polar intermediates while remaining inert under hydrogenation conditions. Following the reduction, the catalyst is removed by filtration, and the solvent is evaporated to yield the crude amine. This crude material is then subjected to diazotization in an aqueous acetic acid solution, where temperature control is paramount to prevent the decomposition of the diazonium species. Finally, the organic phase containing the hydroxymethyl product is extracted and reacted with thionyl chloride in a chlorinated solvent like dichloromethane. The detailed standardized operating procedures for scaling this route from gram to kilogram quantities are outlined in the technical guide below.
- Hydrogenate 3,5-dimethyl-4-methoxy-2-cyanopyridine using Raney nickel or Pd/C catalyst in the presence of ammonia or acid to form the aminomethyl intermediate.
- Perform diazotization on the aminomethyl intermediate using sodium nitrite in an acidic aqueous solution to convert the amino group into a hydroxymethyl group.
- React the resulting hydroxymethyl derivative with thionyl chloride to obtain the final chloromethyl pyridine product, followed by purification via distillation or crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN1042705A offers substantial strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By shifting away from the reliance on 2,3,5-trimethylpyridine, which is noted in the background art as difficult to obtain and expensive, manufacturers can source 3,5-lutidine and simple cyanation reagents that are commodity chemicals with stable global availability. This shift mitigates the risk of supply disruptions caused by the scarcity of specialized starting materials. Furthermore, the reduction in the number of synthetic steps—from seven complex operations in the old route to a concise three-step sequence in the new route—directly correlates to lower operational expenditures. Fewer unit operations mean reduced labor costs, lower energy consumption for heating and cooling, and decreased demand for reactor time, all of which contribute to a more competitive cost structure for the final intermediate.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts in certain steps and the avoidance of hazardous nitration reagents significantly lowers the cost of waste disposal and environmental compliance. The process utilizes Raney nickel, which is recoverable, and avoids the generation of heavy metal waste streams associated with other catalytic systems. Additionally, the ability to telescope steps, such as carrying the amine intermediate directly into diazotization without isolation, reduces solvent usage and processing time. These efficiencies compound to deliver substantial cost savings in the manufacturing of high-purity pharmaceutical intermediates, allowing for better margin management in a price-sensitive market.
- Enhanced Supply Chain Reliability: The robustness of the hydrogenation and diazotization steps ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with API manufacturers. The use of standard equipment, such as atmospheric hydrogenation reactors and glass-lined vessels for acid reactions, means that the process can be easily replicated across multiple manufacturing sites without requiring specialized or custom-built infrastructure. This flexibility enhances supply continuity, as production can be shifted between facilities if necessary without significant re-validation efforts. Moreover, the stability of the key intermediates allows for strategic stockpiling, providing a buffer against unexpected demand surges.
- Scalability and Environmental Compliance: The reaction conditions described, such as atmospheric pressure hydrogenation and moderate temperatures for diazotization, are inherently safer and easier to scale than high-pressure or cryogenic processes. The process generates minimal hazardous byproducts, primarily nitrogen gas and salts, which are easier to treat than the complex organic waste streams from nitration reactions. This aligns with increasingly stringent environmental regulations, reducing the regulatory burden on manufacturing sites. The scalability of the route ensures that as demand for the downstream ATDase inhibitors grows, the supply of this critical intermediate can be ramped up seamlessly from pilot scale to multi-ton commercial production without encountering fundamental engineering bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims within patent CN1042705A, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their existing portfolios and assess the technical risks associated with the scale-up process.
Q: What are the primary advantages of the cyanopyridine route over traditional trimethylpyridine methods?
A: The cyanopyridine route described in patent CN1042705A eliminates the need for difficult-to-obtain 2,3,5-trimethylpyridine and avoids complex N-oxide formation and nitration steps, significantly simplifying the synthetic pathway and improving overall process safety.
Q: Which catalysts are recommended for the hydrogenation step to prevent secondary amine formation?
A: To prevent the formation of unwanted secondary amines during the reduction of the cyano group, the process recommends using Raney nickel coupled with ammonia, or alternatively, Palladium on Carbon (Pd/C) in the presence of a mineral acid or toluenesulfonic acid.
Q: Is this synthesis method scalable for commercial production of ATDase inhibitor intermediates?
A: Yes, the method utilizes standard industrial reagents like Raney nickel and thionyl chloride under moderate conditions (atmospheric pressure hydrogenation, mild temperatures for diazotization), making it highly suitable for commercial scale-up with robust quality control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Dimethyl-4-Methoxypyridine Derivative Supplier
The technological advancements detailed in patent CN1042705A represent a significant opportunity for optimizing the production of ATDase inhibitor intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these laboratory-scale innovations into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot batches to full-scale manufacturing is smooth and compliant. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to monitor impurity profiles and ensure that every batch meets the exacting standards required by global pharmaceutical regulators.
We invite you to collaborate with us to leverage this efficient synthetic route for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our mastery of pyridine chemistry can drive value and reliability in your pharmaceutical manufacturing operations.
