Advanced Manufacturing of Thienopyrrole Intermediates for Glycogen Phosphorylase Inhibitors
The pharmaceutical industry's relentless pursuit of effective treatments for metabolic disorders has placed glycogen phosphorylase inhibitors at the forefront of therapeutic research. Patent CN1688588A discloses a groundbreaking methodology for the preparation of thienopyrrole derivatives, which serve as critical intermediates in the synthesis of these potent bioactive compounds. As a leading entity in fine chemical manufacturing, we recognize that the transition from laboratory discovery to commercial viability hinges on process safety and reproducibility. This patent addresses a critical bottleneck in the prior art, specifically the instability and potential explosiveness associated with traditional synthetic routes for thiophene-based acids. By introducing a novel cyclization strategy involving protected amino-thiophene precursors, the inventors have established a robust pathway that mitigates significant safety risks while maintaining high chemical fidelity. For R&D directors and procurement specialists alike, understanding the nuances of this chemistry is essential for securing a reliable supply chain of high-purity pharmaceutical intermediates.
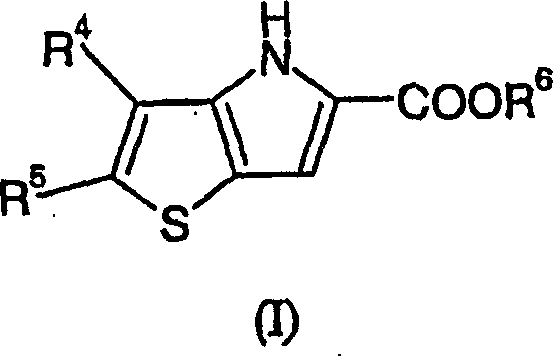
The structural complexity of Formula (I) compounds, characterized by the fused thieno[3,2-b]pyrrole core, demands precise control over regiochemistry and functional group tolerance. The patent details a comprehensive suite of substituents (R4, R5, R6) that allow for extensive medicinal chemistry optimization, enabling the fine-tuning of pharmacokinetic properties. However, the true value proposition lies not just in the molecular diversity but in the manufacturability of the scaffold itself. The disclosed process circumvents the hazardous conditions previously required to construct this heterocyclic system, offering a safer alternative that aligns with modern green chemistry principles and strict industrial safety regulations. This shift represents a pivotal advancement for manufacturers aiming to scale production without compromising on worker safety or environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carboxylic acid precursors for thienopyrrole derivatives has been fraught with significant operational hazards. As noted in the background section of the patent data, conventional methods for preparing certain thiophene acids involve reaction conditions where explosions can occur, presenting an unacceptable risk for large-scale manufacturing. These traditional routes often rely on harsh reagents or unstable intermediates that require specialized containment infrastructure, driving up capital expenditure and operational costs. Furthermore, the lack of robust protecting group strategies in older methodologies frequently leads to poor regioselectivity, resulting in complex impurity profiles that are difficult and costly to purge. For supply chain managers, these factors translate into volatile lead times and inconsistent batch quality, undermining the reliability required for GMP production of active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the novel approach detailed in CN1688588A leverages a sophisticated sequence of protection, functionalization, and cyclization to ensure both safety and precision. The core innovation involves the cyclization of a compound of Formula (II), where the nitrogen atom is masked with a removable protecting group (R7). This strategic protection allows for the subsequent introduction of functional groups without unwanted side reactions. The cyclization step itself is conducted under mild basic conditions, typically using alkali metal carbonates like potassium carbonate in polar aprotic solvents such as DMF at moderate temperatures ranging from 40°C to 100°C. This eliminates the need for extreme thermal inputs or hazardous reagents, drastically simplifying the engineering controls required for production. By decoupling the ring formation from the hazardous steps of precursor synthesis, this method offers a streamlined, safer, and more economically viable route to high-value thienopyrrole intermediates.
Mechanistic Insights into Base-Mediated Cyclization and Directed Metallation
The mechanistic elegance of this process is best understood through the interplay of steric hindrance and electronic activation during the synthesis of the key intermediates. A critical feature of the invention is the use of a directional nitrogen protecting group, such as the tert-butoxycarbonyl (Boc) group or other bulky moieties defined in Formula (ii). During the ortho-directed metallation step, this bulky group serves a dual purpose: it protects the nitrogen lone pair from reacting with electrophiles and sterically directs lithiating agents, such as n-butyllithium, exclusively to the adjacent carbon position on the thiophene ring. This ensures that subsequent formylation occurs with high regioselectivity at the 2-position, preventing the formation of isomeric byproducts that would complicate downstream purification. The precision of this step is paramount for maintaining the integrity of the final API structure.
Following the installation of the aldehyde and ester side chains, the final ring closure proceeds via an intramolecular nucleophilic attack. Under basic conditions, the deprotonated nitrogen attacks the ester carbonyl or facilitates the elimination of the protecting group to form the pyrrole ring. The patent specifies that this cyclization is suitably carried out in the presence of weak bases like potassium carbonate or bicarbonate. The reaction mechanism likely involves the initial deprotection of the nitrogen followed by immediate cyclization, driven by the thermodynamic stability of the aromatic thienopyrrole system. This tandem deprotection-cyclization sequence minimizes the number of isolation steps, thereby reducing material loss and solvent consumption. The ability to perform this transformation in a single pot or with minimal workup significantly enhances the overall process efficiency and yield.
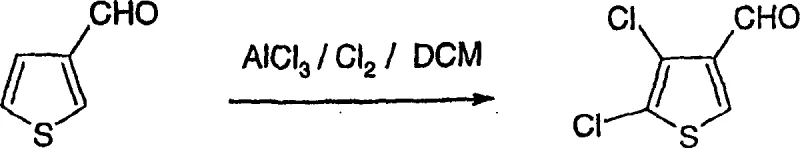
How to Synthesize 2,3-Dichloro-4H-thieno[3,2-b]pyrrole-5-carboxylic Acid Efficiently
The practical implementation of this chemistry is exemplified in the patent's experimental section, which outlines a scalable route starting from commercially available thiophene-3-carbaldehyde. The process begins with a controlled chlorination to install the necessary halogen substituents, followed by oxidation to the carboxylic acid. This acid is then subjected to a Curtius rearrangement to generate the protected amine, setting the stage for the critical lithiation and cyclization steps. The detailed protocol demonstrates that high purity can be achieved through standard crystallization and chromatographic techniques, validating the method's suitability for industrial application. For technical teams looking to replicate or adapt this chemistry, the following guide summarizes the critical operational parameters derived from the patent examples.
- Chlorinate thiophene-3-carbaldehyde using aluminum chloride and chlorine gas in dichloromethane to form 4,5-dichlorothiophene-3-carbaldehyde.
- Oxidize the aldehyde to the corresponding carboxylic acid using potassium permanganate in aqueous sodium hydroxide.
- Perform a Curtius rearrangement using diphenylphosphoryl azide (DPPA) to convert the acid to a protected amine (carbamate).
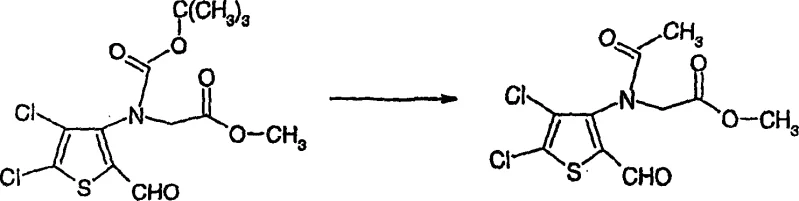
- Execute directed ortho-lithiation followed by formylation to introduce the aldehyde group at the 2-position.
- Alkylate the nitrogen with methyl bromoacetate and subsequently cyclize under basic conditions to form the thienopyrrole ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible strategic benefits beyond mere chemical curiosity. The primary advantage lies in the substantial reduction of operational risk. By eliminating the explosive hazards associated with prior art methods, manufacturers can operate with lower insurance premiums and reduced need for specialized blast-proof infrastructure. This directly translates to cost reduction in pharmaceutical manufacturing, as the capital intensity of the production facility is lowered. Furthermore, the use of common, non-proprietary reagents such as potassium carbonate, DMF, and standard halogenating agents ensures that the supply chain remains resilient against raw material shortages. The reliance on commodity chemicals rather than exotic catalysts enhances the long-term sustainability of the supply line.
- Cost Reduction in Manufacturing: The process design inherently lowers production costs by minimizing the number of unit operations and avoiding expensive transition metal catalysts. The cyclization step utilizes inexpensive inorganic bases and proceeds at moderate temperatures, reducing energy consumption compared to high-temperature or high-pressure alternatives. Additionally, the high regioselectivity achieved through the directing group strategy reduces the burden on purification processes, leading to higher overall yields and less waste generation. This efficiency gain allows for a more competitive pricing structure for the final intermediate, providing a clear economic advantage in a cost-sensitive market.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance in API production. The starting materials, such as thiophene-3-carbaldehyde, are widely available from multiple global suppliers, mitigating the risk of single-source dependency. The mild reaction conditions also mean that the process can be easily transferred between different manufacturing sites without requiring extensive re-validation or specialized equipment modifications. This flexibility strengthens the supply chain against geopolitical disruptions or localized production issues, ensuring continuous availability of key intermediates.
- Scalability and Environmental Compliance: From an environmental perspective, the process aligns well with green chemistry initiatives by avoiding toxic heavy metals and minimizing hazardous waste streams. The solvent systems employed, primarily DMF and dichloromethane, are well-understood and can be efficiently recovered and recycled using standard distillation technologies. The absence of explosive intermediates simplifies waste treatment protocols and reduces the environmental footprint of the manufacturing site. This compliance with stringent environmental regulations facilitates faster regulatory approvals and enhances the corporate social responsibility profile of the manufacturing partner, making it an attractive option for multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patent, we have compiled answers to common inquiries regarding the synthesis and application of these thienopyrrole derivatives. These insights are derived directly from the experimental data and claims presented in CN1688588A, providing a factual basis for decision-making. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this chemistry into their existing pipelines.
Q: What safety advantages does this new process offer over conventional methods?
A: The patent explicitly states that conventional methods for preparing similar acids can involve explosion hazards. This novel process utilizes a Curtius rearrangement and mild cyclization conditions (40-100°C) to avoid unstable intermediates, significantly enhancing operational safety.
Q: How is regioselectivity controlled during the synthesis?
A: Regioselectivity is achieved through the use of a bulky directional nitrogen protecting group (such as a tert-butoxycarbonyl group). This group prevents substitution at the nitrogen atom and directs lithiation reagents specifically to the adjacent ring position, ensuring high purity of the 2-formyl intermediate.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process employs standard industrial reagents like potassium carbonate, DMF, and common solvents. The reaction temperatures are moderate (e.g., 60°C for cyclization), and the workup involves standard extraction and chromatography techniques, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thienopyrrole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to bring this advanced synthetic methodology to life on a commercial scale. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from gram-scale optimization to multi-ton manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of thienopyrrole intermediate meets the highest industry standards. Our commitment to quality and safety makes us the ideal partner for developing complex pharmaceutical intermediates.
We invite you to collaborate with us to leverage this innovative chemistry for your next drug development program. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline and reduce your overall project costs. Let us be your trusted partner in delivering high-quality chemical solutions.
