Advanced Synthesis of Felbamate and PPD Intermediates for Commercial Scale Production
The pharmaceutical industry continuously seeks robust, scalable, and safe methodologies for the production of active pharmaceutical ingredients (APIs) and their critical precursors. Patent CN1092759A presents a groundbreaking advancement in the synthesis of Felbamate, a potent antiepileptic agent, and its vital intermediate, 2-phenyl-1,3-propanediol (PPD). This intellectual property outlines a novel chemical pathway that circumvents the severe safety hazards and environmental liabilities associated with legacy manufacturing techniques. By leveraging specific borohydride reduction strategies and innovative carbamation protocols, this technology offers a viable solution for producing high-purity Felbamate while mitigating the risks of pyrophoric reagents and halogenated waste streams. For global supply chain leaders, adopting such a refined process translates directly into enhanced operational continuity and reduced regulatory friction.
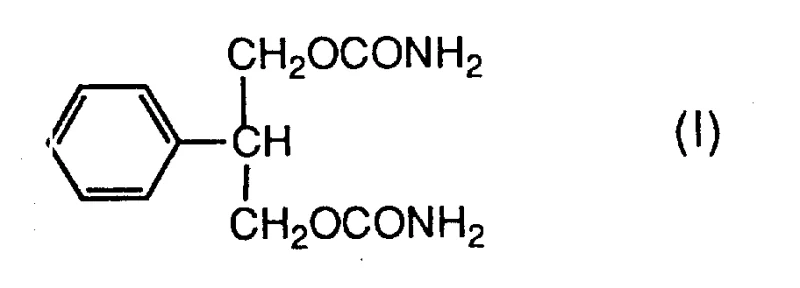
Felbamate, chemically defined as 2-phenyl-1,3-propanediol dicarbamate, serves as a cornerstone therapy for managing various types of epilepsy. The structural integrity and purity of this molecule are paramount for its therapeutic efficacy and patient safety. The patent explicitly details the molecular architecture of Felbamate, designated as formula (I), highlighting the symmetrical dicarbamate functionality attached to the central propane chain. Achieving this specific substitution pattern efficiently has historically been a bottleneck in large-scale manufacturing. The disclosed invention not only provides a route to the final API but also secures the supply of the diol intermediate (PPD), which is a valuable building block in its own right. Understanding the precise chemical identity of the target molecule is the first step in appreciating the magnitude of the process improvements offered by this patent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-phenyl-1,3-propanediol (PPD) has been plagued by significant technical and safety challenges that hinder cost-effective manufacturing. Prior art methods, such as those described in US Patents 4,982,016 and 5,072,056, rely heavily on the reduction of diethyl phenylmalonate. This approach necessitates the use of extremely hazardous, pyrophoric reducing agents like borane dimethyl sulfide, lithium aluminum hydride (LiAlH4), and diisobutylaluminum hydride (DIBAL). These reagents are not only prohibitively expensive but also pose severe fire and explosion risks, requiring specialized containment infrastructure and driving up capital expenditure. Furthermore, alternative routes based on benzaldehyde involve the formation of nitroalkane intermediates and the use of formaldehyde, a known carcinogen, alongside peracetic acid, which introduces additional explosion hazards. The cumulative effect of these dangers makes conventional methods unsatisfactory for modern, safety-conscious industrial operations.
The Novel Approach
In stark contrast to the perilous legacy techniques, the methodology disclosed in CN1092759A introduces a paradigm shift by utilizing alkali metal borohydrides for the critical reduction steps. This novel approach centers on the conversion of formylphenylacetate enolate salts (VIII), or their tautomeric equivalents, into PPD using reagents like sodium borohydride or potassium borohydride. These reducing agents are significantly more stable, easier to handle, and considerably less expensive than their aluminum-based counterparts. The process allows for the reaction to proceed in common protic solvents such as alcohols or water-compatible mixtures, eliminating the need for strictly anhydrous conditions required by LiAlH4. Moreover, the patent describes a carbamation step that avoids environmentally undesirable halogenated solvents like chloroform and halogenated acids, instead employing non-halogenated solvents such as acetonitrile or ethers. This transition represents a substantial improvement in both process safety and environmental compliance.
Mechanistic Insights into Borohydride-Mediated Reduction and Carbamation
The core of this technological breakthrough lies in the versatile reactivity of the formylphenylacetate enolate salt (VIII) and its subsequent transformation through borohydride reduction. The synthesis begins with the formation of the enolate salt, typically achieved by reacting a phenylacetate ester with a formate ester in the presence of a strong base like sodium methoxide. This step generates a stable nucleophilic species that serves as the precursor for the entire synthetic sequence. The patent elucidates that this enolate salt can exist in equilibrium with various tautomeric forms, including E-enols (X), Z-enols (XII), and the free aldehyde ester (XIV), depending on the solvent environment. For instance, in dimethyl sulfoxide (DMSO), the Z-enol form may predominate, whereas in chloroform, a mixture of all three species exists. This mechanistic understanding is crucial for R&D directors aiming to optimize reaction conditions, as the choice of solvent can influence the equilibrium position and potentially the kinetics of the subsequent reduction.
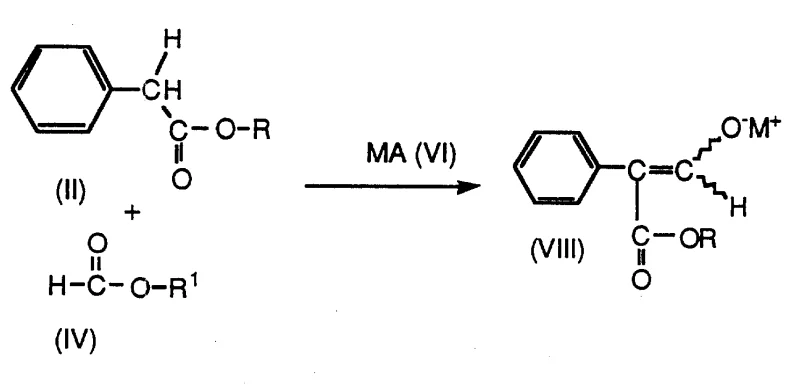
Upon exposure to the borohydride reducing agent, the aldehyde and ester functionalities of the intermediate are reduced to yield boron-containing species such as tropicate borates (XV), borate diesters (XVI), or boronate esters (XVII). These boron intermediates act as protected forms of the diol, which are subsequently hydrolyzed to release the free 2-phenyl-1,3-propanediol (PPD). The patent details multiple pathways (Routes A, B, C, and D) to achieve this transformation, offering flexibility in process design. For example, Route A involves protonating the enolate salt first to form the neutral tautomers before reduction, while Route B mixes the enolate salt directly with the borohydride followed by acidification. This mechanistic flexibility allows manufacturers to tailor the process to their specific equipment and safety protocols. Furthermore, the final conversion of PPD to Felbamate is achieved through a novel carbamation strategy using cyanates and strong acids in non-halogenated solvents, or alternatively, using chlorosulfonyl isocyanate. This avoids the use of phosgene, a highly toxic gas traditionally used in carbamate synthesis, thereby enhancing the overall safety profile of the manufacturing plant.
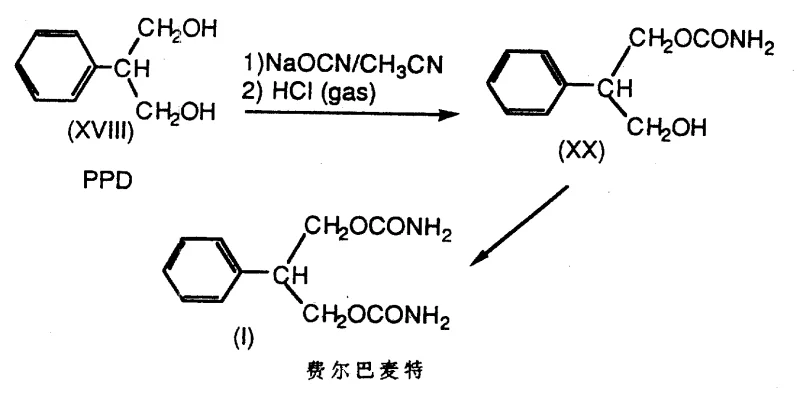
How to Synthesize 2-Phenyl-1,3-propanediol Efficiently
The practical implementation of this synthesis requires careful control of reaction parameters to maximize yield and purity while maintaining safety. The process generally initiates with the preparation of the sodium salt of methyl 2-formyl-2-phenylacetate, which can be isolated as a wet filter cake or used in situ. This intermediate is then subjected to reduction using an effective amount of sodium borohydride, typically in an alcoholic solvent like ethanol or isopropanol. The reaction temperature is carefully managed, often ranging from -20°C to 40°C, to control the exotherm associated with borohydride decomposition and hydrogen evolution. Following the reduction, the reaction mixture is treated with acid, such as sulfuric acid or acetic acid, to hydrolyze the boron intermediates and liberate the free diol. The resulting PPD can be recovered through standard workup procedures including extraction, crystallization, or distillation. For detailed operational parameters, stoichiometry, and specific workup instructions, please refer to the standardized guide below.
- Preparation of the enolate salt (VIII) by reacting methyl phenylacetate with methyl formate in the presence of sodium methoxide.
- Reduction of the enolate salt or its protonated tautomers (X, XII, XIV) using an effective amount of alkali metal borohydride (e.g., NaBH4) in a compatible solvent.
- Hydrolysis of the resulting borate intermediates (XV, XVI, XVII) with acid or water to isolate high-purity 2-phenyl-1,3-propanediol (PPD).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN1092759A offers compelling economic and logistical benefits that extend beyond simple chemical yield. The primary advantage lies in the drastic simplification of the safety infrastructure required for production. By replacing pyrophoric reagents like lithium aluminum hydride with stable borohydrides, facilities can eliminate the need for specialized inert atmosphere handling systems and expensive fire suppression measures dedicated to reactive metal hydrides. This reduction in hazard classification directly lowers insurance premiums and operational overheads. Additionally, the ability to use non-halogenated solvents removes the burden of costly hazardous waste disposal associated with chlorinated organics, further driving down the total cost of ownership for the manufacturing process. These factors combine to create a more resilient and cost-efficient supply chain for this critical pharmaceutical intermediate.
- Cost Reduction in Manufacturing: The substitution of expensive and dangerous reducing agents with commodity chemicals like sodium borohydride results in significant raw material cost savings. Sodium borohydride is widely available and priced substantially lower than lithium aluminum hydride or borane complexes. Furthermore, the process eliminates the need for rigorous drying of solients and reactants, which reduces energy consumption associated with solvent purification and distillation. The ability to store the enolate salt intermediates also allows for batch optimization, where the upstream formation of the salt can be decoupled from the reduction step, leading to better asset utilization and reduced downtime. These cumulative efficiencies contribute to a leaner manufacturing model with a lower cost basis per kilogram of finished product.
- Enhanced Supply Chain Reliability: Reliance on hazardous, specialty reagents often creates single points of failure in the supply chain, as few suppliers are willing or able to manufacture and transport pyrophoric materials in bulk. By shifting to a chemistry based on stable, commodity-grade reagents, manufacturers can diversify their supplier base and reduce the risk of supply disruptions. The robustness of the borohydride reduction also means that the process is less sensitive to minor fluctuations in moisture content or temperature compared to aluminum hydride reductions, leading to more consistent batch-to-batch quality. This reliability is critical for maintaining continuous production schedules and meeting the stringent delivery commitments required by downstream API manufacturers and pharmaceutical companies.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing reaction conditions that are compatible with standard stainless steel reactors and common processing equipment. The avoidance of halogenated solvents and toxic gases like phosgene simplifies the permitting process for new manufacturing lines and ensures compliance with increasingly strict environmental regulations such as REACH and TSCA. The waste streams generated are primarily aqueous borates and alcohols, which are easier to treat and dispose of than heavy metal residues or chlorinated organic waste. This environmental compatibility not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturer, a key factor for multinational pharmaceutical clients seeking green supply chain partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of technology transfer and scale-up.
Q: What are the safety advantages of this PPD synthesis route compared to traditional methods?
A: Traditional methods often utilize highly pyrophoric reagents like lithium aluminum hydride (LiAlH4) or diisobutylaluminum hydride (DIBAL), which pose significant fire hazards and require specialized handling infrastructure. The patented process utilizes alkali metal borohydrides, such as sodium borohydride, which are significantly safer, more stable, and easier to handle on an industrial scale, thereby reducing operational risks and insurance costs.
Q: How does this process address environmental concerns regarding solvent usage?
A: Conventional carbamation processes for converting PPD to Felbamate frequently rely on environmentally undesirable halogenated solvents like chloroform and halogenated acids such as trifluoroacetic acid. This invention enables the use of non-halogenated solvents, including acetonitrile, ethers, and alcohols, for both the reduction and the subsequent carbamation steps. This shift drastically simplifies waste disposal protocols and aligns with modern green chemistry standards.
Q: Can the intermediate enolate salts be stored for later use?
A: Yes, a distinct advantage of this methodology is the stability of the formylphenylacetate enolate salts (VIII). Unlike some transient intermediates in other synthetic routes, these salts can be conveniently separated from the reaction mixture, dried, and stored for subsequent reactions. This flexibility allows manufacturers to decouple the upstream enolate formation from the downstream reduction, optimizing production scheduling and inventory management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Felbamate Supplier
The technological advancements detailed in Patent CN1092759A represent a significant opportunity for optimizing the production of Felbamate and its intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage these innovations for our global clientele. As a premier CDMO partner, we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. Our facility is equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-phenyl-1,3-propanediol or Felbamate meets the highest international standards for pharmaceutical use.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this safer, more efficient synthesis route can be integrated into your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to request specific COA data and route feasibility assessments to validate the superior quality and economic advantages of our manufacturing capabilities. Let us collaborate to secure a sustainable and cost-effective supply of high-purity Felbamate intermediates for your critical therapeutic programs.
