Advanced Synthesis of 1,1,1-Tris(4-Hydroxyphenyl)Ethane for Commercial Polymer Applications
Introduction to Patent CN113461493B Technology
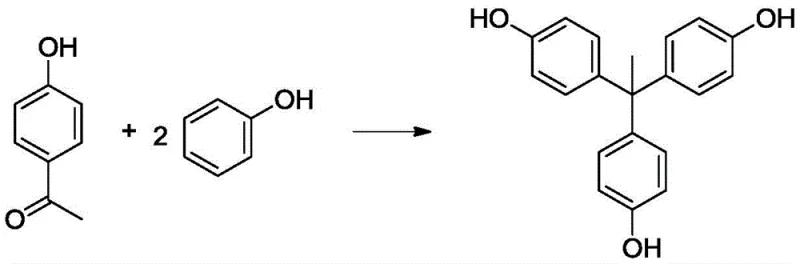
The chemical industry is constantly seeking more efficient pathways for producing high-value trifunctional phenolic compounds, and the technology disclosed in patent CN113461493B represents a significant leap forward in the synthesis of 1,1,1-tris(4-hydroxyphenyl)ethane (THPE). This specific patent outlines a refined preparation method that utilizes 4-hydroxyacetophenone and phenol as primary raw materials, employing an acid catalyst in conjunction with a novel auxiliary agent comprising weakly alkaline nitrogen-containing compounds. For R&D directors and procurement specialists, the core innovation lies in the ability to drastically improve the conversion rate of 4-hydroxyacetophenone without relying on the massive excess of phenol traditionally required to push equilibrium. By integrating additives such as 4-dimethylaminopyridine or 1,8-diazabicycloundecen-7-ene, the process achieves superior selectivity and yield, addressing long-standing pain points regarding raw material efficiency and downstream purification complexity in the manufacturing of polymer cross-linking agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
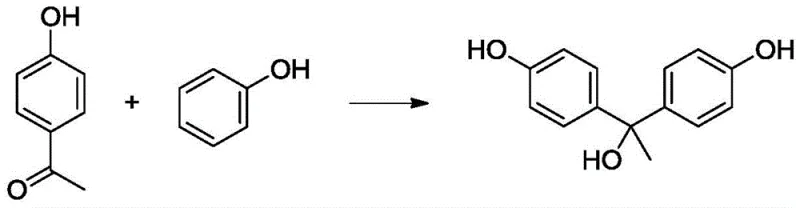
Historically, the industrial production of 1,1,1-tris(4-hydroxyphenyl)ethane has been plagued by inefficient stoichiometry and difficult purification protocols that inflate operational costs. Conventional acidic condensation processes typically necessitate the use of phenol in amounts ranging from 5 to 20 times the theoretical requirement to ensure adequate conversion of the ketone starting material. This massive excess creates a substantial burden on the supply chain, requiring extensive recovery systems and energy-intensive distillation columns to separate unreacted phenol from the product matrix. Furthermore, traditional methods often employ zinc salts like zinc chloride as cocatalysts, which introduces the risk of heavy metal contamination; residual zinc ions can persist in the final product, compromising its suitability for high-performance polymer applications or pharmaceutical intermediates where metal limits are strictly regulated. Additionally, the side reaction leading to 1,1-bis(hydroxyphenyl)ethane isomers is difficult to suppress under standard acidic conditions, resulting in crude products with high impurity loads that complicate crystallization and lower overall yield.
The Novel Approach
The methodology presented in CN113461493B fundamentally alters the reaction landscape by introducing a weakly alkaline nitrogen-containing compound as a promoter alongside the acid catalyst. This dual-catalyst system allows the reaction to proceed efficiently even when the molar ratio of phenol to 4-hydroxyacetophenone is reduced to a range of 1:2 to 1:6, which is only slightly higher than the theoretical stoichiometric demand. This reduction in phenol loading not only lowers raw material costs but also simplifies the post-reaction workup by minimizing the volume of solvent and unreacted starting material that must be removed. The presence of the nitrogenous base appears to facilitate the conversion of the intermediate bisphenol species into the final tris-phenol product, thereby suppressing the accumulation of the primary byproduct. As illustrated in the main reaction scheme below, the process yields a cleaner reaction profile with significantly higher selectivity for the target molecule, enabling a more streamlined path to high-purity material suitable for sensitive electronic or polymer applications.
Mechanistic Insights into Acid-Catalyzed Condensation with Base Promotion
The mechanistic advantage of this process stems from the synergistic interaction between the strong acid catalyst and the weak base additive during the electrophilic aromatic substitution steps. The acid, whether it be methanesulfonic acid, p-toluenesulfonic acid, or concentrated phosphoric acid, serves to protonate the carbonyl oxygen of the 4-hydroxyacetophenone, generating a highly reactive carbocation or carbocation-like intermediate. In traditional systems, the equilibrium favors the formation of the bis-substituted intermediate (bisphenol) unless a vast excess of phenol is present to drive the third substitution. However, the addition of a weak base with a pKb value between 0.1 and 10, such as imidazole derivatives or pyridines, likely modulates the acidity of the local environment or stabilizes transition states in a way that promotes the third nucleophilic attack by phenol. This results in a conversion rate of 4-hydroxyacetophenone exceeding 99% and a selectivity for the target tris-product greater than 97%, effectively shutting down the pathway that leads to the persistent bisphenol impurity.
Impurity control is further enhanced through a sophisticated purification protocol that leverages the chemical properties of the crude mixture. Following the reaction, the crude solid is subjected to a recrystallization process involving solvents like methanol or ethanol, water, and crucially, sodium borohydride and activated carbon. The inclusion of sodium borohydride serves a dual purpose: it acts as a reducing agent to eliminate colored oxidative impurities or quinone-like species that often form during phenolic condensations, while the activated carbon adsorbs trace organic contaminants and catalyst residues. This step ensures that the final product achieves a purity level exceeding 99%, with specific control over isomeric impurities like iso-THPE, which are maintained below 1%. Such rigorous impurity management is critical for customers in the polymer and fine chemical sectors who require consistent batch-to-batch performance and minimal interference with downstream polymerization kinetics.
How to Synthesize 1,1,1-Tris(4-Hydroxyphenyl)Ethane Efficiently
The synthesis protocol described in the patent offers a robust framework for scaling production, beginning with the melting of phenol under an inert nitrogen atmosphere to prevent oxidation. Once the phenol is molten, the acid catalyst and the specific weak base additive are introduced, and the mixture is stirred to ensure homogeneity before the slow addition of 4-hydroxyacetophenone. Temperature control is vital, with the reaction typically maintained between 45°C and 90°C depending on the specific catalyst system chosen; for instance, using methanesulfonic acid with 4-dimethylaminopyridine may allow for operation at higher temperatures around 90°C to accelerate kinetics, while phosphoric acid systems might operate optimally at lower temperatures near 55°C. The detailed standardized synthesis steps see the guide below for precise operational parameters regarding addition rates, stirring times, and quenching procedures.
- Melt phenol under inert gas and add acid catalyst along with a weakly alkaline nitrogen-containing compound such as 4-dimethylaminopyridine.
- Slowly add 4-hydroxyacetophenone to the mixture while maintaining temperature between 45-90°C for 1 to 24 hours.
- Cool the reaction, add dichloroethane, distill off solvent, filter the crude product, and purify via recrystallization with sodium borohydride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route translates into tangible operational efficiencies and risk mitigation strategies. The most immediate impact is the drastic reduction in the volume of phenol required per unit of product, which alleviates pressure on raw material sourcing and storage logistics. Since phenol is a hazardous material with strict handling regulations, reducing the inventory load and the volume of material requiring recovery and recycling significantly lowers the environmental compliance burden and associated disposal costs. Furthermore, the elimination of zinc-based cocatalysts removes the need for expensive and time-consuming metal scavenging steps or ion-exchange purification, which are often bottlenecks in traditional manufacturing flows. This simplification of the downstream processing train allows for faster batch turnover and reduced utility consumption, directly contributing to a leaner and more responsive supply chain capable of meeting tight delivery windows for high-purity polymer additives.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the optimization of raw material stoichiometry and the simplification of purification workflows. By operating with near-stoichiometric amounts of phenol rather than a large excess, the facility saves substantially on raw material procurement costs and reduces the energy load associated with distilling and recovering unreacted phenol. Additionally, the absence of heavy metal catalysts eliminates the cost of specialized filtration media or chelating agents required to meet stringent metal specifications, while the high selectivity of the reaction minimizes product loss during purification, leading to a higher overall mass balance and improved yield efficiency.
- Enhanced Supply Chain Reliability: From a supply continuity perspective, the reliance on readily available organic acids and nitrogenous bases rather than specialized metal salts enhances the resilience of the supply chain. The process is less susceptible to disruptions in the supply of specific inorganic cocatalysts, and the reduced dependency on massive volumes of phenol mitigates the risk associated with price volatility in the phenol market. Moreover, the simplified post-treatment process, which involves straightforward filtration and crystallization rather than complex multi-stage distillations, reduces the likelihood of equipment fouling or maintenance downtime, ensuring a more consistent and reliable output of finished goods for downstream customers.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly superior to prior art, aligning with modern green chemistry principles and regulatory expectations. The reduction in waste generation, particularly the avoidance of zinc-contaminated waste streams, simplifies effluent treatment and reduces the environmental footprint of the manufacturing site. The process is inherently scalable, as the reaction conditions are mild and do not require extreme pressures or temperatures that would necessitate specialized high-pressure reactors, allowing for seamless scale-up from pilot batches to multi-ton commercial production while maintaining strict adherence to environmental safety standards and waste discharge limits.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this synthesis technology, derived directly from the experimental data and comparative examples provided in the patent documentation. These insights are intended to clarify the operational advantages and quality improvements achievable through the adoption of the weak-base promoted acid catalysis method. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into existing production facilities or for procurement teams assessing the quality specifications of potential suppliers.
Q: How does the new method reduce phenol consumption compared to traditional processes?
A: Traditional methods require phenol in 5-20 times excess to drive conversion, whereas this patent enables near-stoichiometric ratios (1:2 to 1:6) by using weak base additives to improve 4-hydroxyacetophenone conversion.
Q: What is the primary advantage regarding impurity profiles in this synthesis?
A: The addition of weakly alkaline nitrogen compounds significantly suppresses the formation of 1,1-bis(hydroxyphenyl)ethane byproducts, reducing selectivity of this impurity to less than 2% compared to over 13% in conventional acid-only methods.
Q: Does this process introduce heavy metal contaminants like zinc?
A: No, unlike prior art methods utilizing zinc chloride or zinc sulfide cocatalysts which leave residual zinc ions, this method relies on organic acids and nitrogenous bases, ensuring a metal-free product suitable for sensitive polymer applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1,1-Tris(4-Hydroxyphenyl)Ethane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance cross-linking agents play in the development of advanced polymer materials and antioxidants. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering 1,1,1-tris(4-hydroxyphenyl)ethane with stringent purity specifications, leveraging rigorous QC labs to verify that every batch meets the exacting standards required for polymer synthesis and fine chemical applications. Our capability to implement the advanced catalytic strategies described in recent patents allows us to offer a product with superior impurity profiles and consistent physical properties.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing processes can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our supply chain. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that our 1,1,1-tris(4-hydroxyphenyl)ethane aligns perfectly with your formulation needs and quality targets.
