Advanced Synthesis of Fused Imidazopyridine Intermediates for Scalable Pharmaceutical Production
Introduction to Novel Amidine Derivative Synthesis
The pharmaceutical industry continuously seeks robust synthetic pathways for complex heterocyclic intermediates, particularly those serving as precursors for nootropic agents and cognitive enhancers. Patent CN1250550C discloses a groundbreaking preparation method for amidine derivatives, which are critical intermediates in the production of fused imidazopyridine derivatives. This technology addresses long-standing inefficiencies in traditional synthesis routes by fundamentally altering the solvent system and reaction conditions. By shifting away from environmentally hazardous and operationally difficult solvents like dichloromethane and dimethylformamide, this innovation offers a streamlined approach that enhances both chemical efficiency and process safety. For R&D teams and procurement strategists, understanding this shift is vital for optimizing the supply chain of high-purity pharmaceutical intermediates.
The core breakthrough lies in the strategic selection of acetonitrile and acetone as the primary reaction media. Historically, the synthesis of these fused ring systems has been plagued by side reactions and difficult workup procedures associated with high-boiling solvents. The new method described in the patent utilizes a three-step sequence that maximizes yield while minimizing by-product formation. This is particularly relevant for the production of compounds like 2-(3-isoxazolyl)-3,6,7,9-tetrahydroimidazo[4,5-d]pyrano[4,3-b]pyridine, where purity profiles directly impact the efficacy and safety of the final active pharmaceutical ingredient (API). As a reliable pharmaceutical intermediate supplier, leveraging such patented methodologies ensures a competitive edge in delivering cost-effective and high-quality materials to global markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, such as JP 1993/286973A and various Tetrahedron Letters publications, predominantly relied on dichloromethane (DCM) or dimethylformamide (DMF) as reaction solvents. While chemically feasible, these solvents present severe drawbacks for industrial scale-up. Dichloromethane is subject to stringent global emission control regulations, necessitating expensive recovery equipment and specialized containment infrastructure to prevent environmental release. Furthermore, reactions in DCM often suffer from slow progress and high yields of unwanted by-products, complicating the isolation of the desired amidine derivatives. On the other hand, DMF possesses a high boiling point of 153°C, making its removal via evaporation energy-intensive and technically challenging. This thermal stability often traps impurities within the reaction matrix, requiring complex purification protocols that erode overall process efficiency and increase manufacturing costs significantly.
The Novel Approach
The methodology outlined in CN1250550C circumvents these issues by employing acetonitrile for steps 1 and 3, and acetone for step 2. This solvent switch is not merely a substitution but a strategic optimization that alters the reaction kinetics and thermodynamics favorably. In the acetonitrile system, the formation of problematic oxazole by-products is drastically suppressed to approximately 0.1%, compared to significant levels in conventional routes. This reduction eliminates the need for filtration steps to remove precipitated impurities, allowing for a smoother workflow. Additionally, the lower boiling points of acetonitrile and acetone facilitate easier solvent removal and recycling, aligning with green chemistry principles. The process enables the efficient production of compound (IV) and subsequent intermediates through simple crystallization techniques, demonstrating a clear path toward cost reduction in API manufacturing.
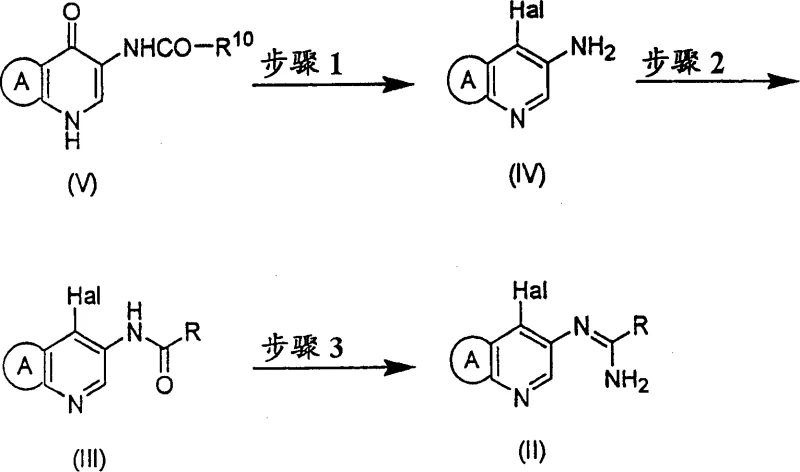
The visual representation of this three-step process highlights the elegance of the new route. Step 1 involves the conversion of a pyridone amide (V) to a chloro-amine (IV) via halogenation and hydrolysis. Step 2 proceeds with acylation in acetone to form the amide (III), and Step 3 concludes with the formation of the amidine (II) through halogenation and amination. This logical progression ensures that each intermediate can be isolated in high purity, providing multiple quality control checkpoints essential for GMP compliance. The ability to perform these transformations in readily available, low-toxicity solvents makes this route highly attractive for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into the Acetonitrile-Mediated Cyclization
The mechanistic underpinning of this synthesis relies on precise control over halogenation and nucleophilic substitution events. In Step 1, the reaction of compound (V) with a halogenating agent like phosphorus oxychloride (POCl3) in the presence of catalytic DMF generates a reactive Vilsmeier-Haack type intermediate. Conducting this in acetonitrile at temperatures ranging from -20°C to 100°C allows for controlled chlorination without excessive degradation. The subsequent hydrolysis step is critical; it reverses any transient oxazole formation back to the starting material or desired product, preventing the accumulation of insoluble impurities that typically clog filters in DCM-based processes. This dynamic equilibrium ensures that the reaction mixture remains homogeneous enough for efficient processing until the final precipitation of the target amine.
Step 3 represents the culmination of the amidine formation, where the amide (III) is activated by a halogenating agent such as phosphorus pentachloride (PCl5) or POCl3 in acetonitrile. The use of acetonitrile here is pivotal because it ensures sufficient miscibility with liquid ammonia during the subsequent amination step. In contrast, other solvents might lead to phase separation or insufficient mixing, slowing down the reaction and promoting side products. The presence of an organic base like pyridine further scavenges generated acid, driving the equilibrium toward the formation of the imidoyl chloride intermediate. This intermediate is then rapidly converted to the amidine (II) upon treatment with ammonia. The final cyclization to the fused imidazopyridine (I) involves a sulfinate-mediated displacement of the halogen, a reaction that proceeds efficiently at elevated temperatures (100°C to 145°C) in polar aprotic solvents like NMP or DMSO.
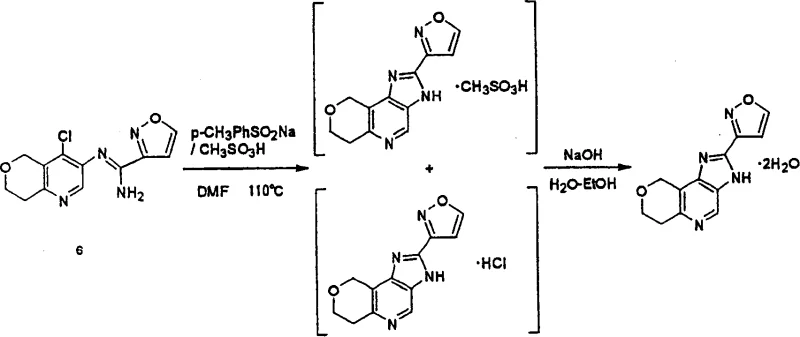
The final transformation, as depicted in the reaction scheme, showcases the conversion of the amidine derivative into the biologically active fused ring system. This step often involves heating compound (6) with sodium p-toluenesulfinate in a high-boiling solvent, followed by salt formation with phosphoric acid. The ability to isolate specific crystal forms, such as the monohydrate phosphate salt, underscores the importance of process control in defining the physical properties of the API. Understanding these mechanistic details allows process chemists to fine-tune reaction parameters, ensuring consistent batch-to-batch quality and minimizing the risk of polymorphic variations that could affect drug bioavailability.
How to Synthesize Fused Imidazopyridine Intermediates Efficiently
Implementing this synthesis requires strict adherence to the specified solvent systems and temperature profiles to maximize yield and purity. The protocol begins with the suspension of the starting pyridone amide in acetonitrile, followed by the controlled addition of phosphorus oxychloride at low temperatures to manage exotherms. The subsequent hydrolysis and workup involve careful pH adjustments to induce crystallization of the intermediate amine. For the acylation step, acetone serves as the ideal medium, allowing for the rapid precipitation of the amide product upon water addition, which simplifies isolation. The final amidine formation leverages the solubility characteristics of acetonitrile to facilitate the ammonia reaction. Detailed standard operating procedures for these steps are essential for technology transfer.
- Step 1: React pyridone amide with phosphorus oxychloride in acetonitrile with DMF catalyst, followed by hydrolysis to obtain the chloro-amine intermediate.
- Step 2: Acylate the chloro-amine with a carboxylic acid derivative in acetone using pyridine and phosphorus oxychloride to form the amide intermediate.
- Step 3: Convert the amide to an imidoyl chloride using phosphorus pentachloride in acetonitrile, followed by amination with ammonia to yield the amidine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers substantial benefits for supply chain stability and cost management. The elimination of dichloromethane removes the regulatory burden and capital expenditure associated with solvent recovery and emission control systems. This directly translates to lower overhead costs and reduced environmental compliance risks. Furthermore, the replacement of DMF with acetonitrile and acetone simplifies the distillation and recycling processes due to their lower boiling points. This efficiency gain reduces energy consumption and shortens cycle times, enabling faster throughput in manufacturing facilities. For procurement managers, this means a more resilient supply chain less susceptible to solvent shortages or regulatory crackdowns on chlorinated hydrocarbons.
- Cost Reduction in Manufacturing: The process significantly lowers operational expenses by eliminating the need for specialized equipment to handle and recover dichloromethane. The simplified workup procedures, which rely on crystallization rather than complex chromatography or extensive washing, reduce labor and material costs. Additionally, the high yields reported in the examples, such as 85.3% for compound 3 and 91.4% for compound 4, indicate a material-efficient process that minimizes waste generation. This efficiency is crucial for maintaining competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Acetonitrile and acetone are commodity chemicals with robust global supply networks, unlike some specialized solvents that may face availability fluctuations. By relying on these common solvents, manufacturers can secure raw material supplies more easily and negotiate better pricing terms. The robustness of the crystallization steps also ensures that the production schedule is less likely to be disrupted by purification bottlenecks. This reliability is paramount for meeting the just-in-time delivery requirements of major pharmaceutical clients who depend on consistent intermediate availability for their own API production schedules.
- Scalability and Environmental Compliance: The method is explicitly designed for industrial applicability, with examples demonstrating successful scaling from gram to kilogram quantities without loss of efficiency. The reduction in hazardous waste, particularly chlorinated solvents, aligns with increasingly strict environmental regulations worldwide. This compliance advantage future-proofs the manufacturing process against tightening emission standards. Moreover, the ability to produce specific crystal forms of the final phosphate salt ensures that the material meets the rigorous physical specifications required for downstream formulation, reducing the risk of batch rejection and ensuring seamless integration into the customer's production line.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in CN1250550C. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this technology for their specific production needs. The answers reflect the practical realities of scaling this chemistry from the laboratory to commercial manufacturing environments.
Q: What are the primary solvent advantages of this new synthesis method?
A: The process replaces dichloromethane (DCM) and dimethylformamide (DMF) with acetonitrile and acetone. This eliminates the need for complex DCM emission control equipment and avoids the difficulty of removing high-boiling DMF (153°C), significantly simplifying downstream purification and solvent recovery.
Q: How does this method improve impurity control compared to conventional routes?
A: Conventional methods often generate high yields of oxazole by-products that precipitate and complicate filtration. The acetonitrile-based route described in CN1250550C minimizes oxazole formation (to approximately 0.1%), allowing for direct crystallization without cumbersome filtration steps, thereby enhancing overall purity.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly highlights industrial applicability. The use of common solvents like acetonitrile and acetone, combined with robust crystallization steps that achieve yields over 85% in key stages, demonstrates strong scalability and operational simplicity suitable for multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fused Imidazopyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation therapeutics. Our expertise in process chemistry allows us to adapt advanced synthetic routes, such as the one described in CN1250550C, to meet the rigorous demands of the pharmaceutical industry. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that consistently meet stringent purity specifications. Our state-of-the-art facilities are equipped with rigorous QC labs capable of performing detailed impurity profiling and polymorph screening, guaranteeing that every batch of fused imidazopyridine intermediate performs reliably in your downstream synthesis.
We invite you to collaborate with our technical team to explore how this optimized synthesis can benefit your specific project requirements. By leveraging our manufacturing capabilities, you can achieve a Customized Cost-Saving Analysis tailored to your volume needs and timeline. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can accelerate the development of your drug candidates while optimizing production costs and ensuring a secure, compliant supply chain for your critical raw materials.
