Strategic Analysis of Novel NAD Biosynthesis for Commercial Scale-up and Procurement
Strategic Analysis of Novel NAD Biosynthesis for Commercial Scale-up and Procurement
Introduction to Patent CN110607335B and Technological Breakthroughs
The pharmaceutical and fine chemical industries are constantly seeking more efficient routes for producing essential cofactors, and patent CN110607335B presents a significant advancement in the biosynthesis of nicotinamide adenine dinucleotide compounds. This intellectual property discloses a novel synthetic route that fundamentally shifts the starting point of production to chorismate, a metabolite widely available across various life forms. By leveraging a specific enzymatic cascade involving PhzD and PhzE proteins, the method achieves a transamination rearrangement that generates aminodeoxyisochorismate without the traditional reliance on expensive amino acid precursors. This innovation is particularly critical for R&D directors focusing on purity and impurity profiles, as the decoupling from tryptophan or aspartate synthesis minimizes metabolic interference. The strategic value of this patent lies in its ability to utilize a common metabolic branch point, theoretically allowing for higher yields and simpler downstream processing. For procurement and supply chain leaders, this represents a potential shift towards more stable and cost-effective sourcing of high-purity NAD and its derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the de novo synthesis of NAD in biological systems has been constrained by its tight coupling with essential amino acid pathways, specifically the tryptophan-kynurenic acid pathway in eukaryotes or the aspartate pathway in bacteria. These conventional methods require the consumption of valuable amino acids like tryptophan or aspartate, which are themselves costly to produce and regulate within a fermentation broth. This dependency creates a metabolic bottleneck where the concentration of the target NAD compound is inherently limited by the organism's need to maintain protein synthesis balance. Furthermore, traditional yeast fermentation methods often suffer from relatively expensive production costs due to the complex media requirements and the need to purify corresponding proteases for in vitro conversion. The reliance on these coupled pathways not only increases the raw material costs but also complicates the impurity profile, making it challenging to achieve the stringent purity specifications required for pharmaceutical applications without extensive purification steps.
The Novel Approach
In contrast, the novel approach detailed in the patent utilizes chorismate as the primary initiator, effectively bypassing the metabolic constraints associated with amino acid consumption. This pathway employs a series of specific enzymes, including 2-amino-4-deoxychorismate synthase and 2,3-dihydro-3-hydroxyanthranilate synthase, to drive the reaction forward with high specificity. By removing the pyruvate part and proceeding through oxidative ring-opening rearrangement, the method generates quinolinic acid which then enters the NAD salvage pathway. This decoupling means that the production of NAD does not compete with the host organism's essential growth processes, theoretically allowing for much higher accumulation of the target product. The use of a recombinant Escherichia coli system further enhances this advantage, as E. coli offers a clear genetic background and mature fermentation processes that are well-understood in industrial settings. This shift represents a paradigm change in how we approach the manufacturing of complex biochemicals, moving from extraction or coupled synthesis to dedicated, optimized biosynthetic routes.
Mechanistic Insights into Enzymatic Cascade and Impurity Control
The core of this technological breakthrough lies in the precise orchestration of four key enzymatic reactions that transform chorismate into quinolinic acid. The process begins with the transamination rearrangement of chorismate, catalyzed by the PhzD protein, to yield aminodeoxyisochorismate. 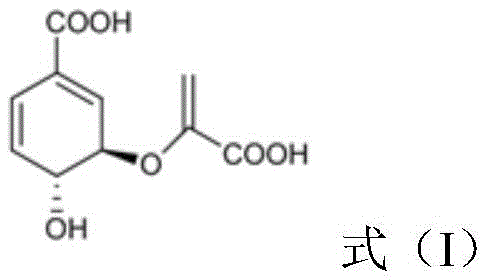
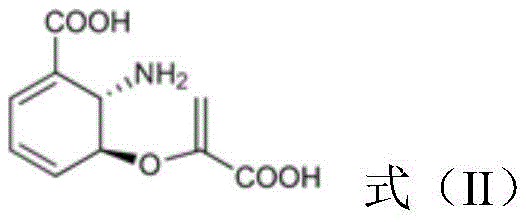 Subsequently, the PhzE protein catalyzes the removal of the pyruvate moiety from aminodeoxyisochorismate, generating 2,3-dihydro-3-hydroxyanthranilic acid.
Subsequently, the PhzE protein catalyzes the removal of the pyruvate moiety from aminodeoxyisochorismate, generating 2,3-dihydro-3-hydroxyanthranilic acid. 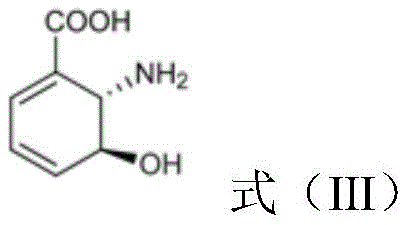 This intermediate is then dehydrogenated by a specific DHHA-2,3-dehydrogenase, such as Pau20 or its homologs, to produce 3-hydroxyanthranilic acid.
This intermediate is then dehydrogenated by a specific DHHA-2,3-dehydrogenase, such as Pau20 or its homologs, to produce 3-hydroxyanthranilic acid. 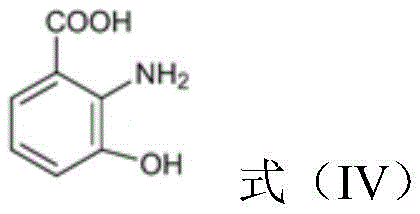 Finally, the NabC protein facilitates the oxidative ring-opening rearrangement to form quinolinic acid, which is the direct precursor for the salvage pathway.
Finally, the NabC protein facilitates the oxidative ring-opening rearrangement to form quinolinic acid, which is the direct precursor for the salvage pathway.  This stepwise mechanism ensures high regioselectivity, significantly reducing the formation of by-products that typically plague less specific chemical synthesis routes. For R&D teams, understanding this cascade is vital for optimizing expression vectors and fermentation conditions to maximize the flux through this specific pathway.
This stepwise mechanism ensures high regioselectivity, significantly reducing the formation of by-products that typically plague less specific chemical synthesis routes. For R&D teams, understanding this cascade is vital for optimizing expression vectors and fermentation conditions to maximize the flux through this specific pathway.
Impurity control is inherently built into this enzymatic design due to the high specificity of the biocatalysts involved. Unlike chemical synthesis which might generate various isomers or side-reaction products requiring complex chromatography, the enzymatic route leverages the stereospecificity of proteins like PhzD and PhzE. The patent highlights the use of specific dehydrogenases that utilize NAD+ as a cofactor, creating a self-regulating system where the cofactor balance can be managed within the cell. The elimination of transition metal catalysts, often used in traditional organic synthesis, means there is no risk of heavy metal contamination, a critical parameter for pharmaceutical intermediates. Furthermore, the use of a recombinant E. coli host allows for genetic engineering strategies to knock out competing pathways, such as the native nadA and nadB genes, ensuring that the metabolic flux is directed almost exclusively towards the desired NAD synthesis. This genetic precision results in a cleaner crude product, simplifying the downstream purification process and ensuring consistent quality across batches.
How to Synthesize Nicotinamide Adenine Dinucleotide Efficiently
Implementing this synthesis route requires a systematic approach to strain engineering and fermentation optimization to ensure robust production capabilities. The process begins with the construction of a recombinant organism, typically Escherichia coli, into which the coding genes for the four key enzymes are introduced via a specialized expression vector like pXB1a-QA. Detailed standardized synthesis steps see the guide below.
- Initiate the pathway using chorismate as the starting substrate in a recombinant organism.
- Catalyze transamination rearrangement using PhzD protein to generate aminodeoxyisochorismate.
- Convert intermediates to quinolinic acid using PhzE, DHHA-2,3-dehydrogenase, and NabC proteins.
- Complete NAD synthesis via the salvage pathway using quinolinic acid phosphotransferase.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this biosynthetic method offers substantial advantages that directly address the pain points of procurement managers and supply chain heads. The primary benefit is the potential for significant cost reduction in manufacturing, driven by the elimination of expensive amino acid precursors like tryptophan. By utilizing chorismate, which can be generated from simple inorganic salt and glucose media, the raw material costs are drastically simplified and lowered. This shift reduces the dependency on volatile amino acid markets and allows for more predictable budgeting and cost structures. Additionally, the removal of the need for expensive ATP precursors in in vitro conversion further enhances the economic viability of the process. For procurement teams, this translates into a more stable pricing model for high-purity NAD intermediates, allowing for better long-term contract negotiations and cost planning.
- Cost Reduction in Manufacturing: The decoupling of NAD synthesis from amino acid metabolism means that the production process no longer competes with the host cell's protein synthesis needs, leading to theoretically higher yields per unit of substrate. This efficiency gain directly impacts the cost of goods sold, as less feedstock is required to produce the same amount of active pharmaceutical ingredient. Furthermore, the simplified medium requirements reduce the complexity and cost of media preparation and sterilization. The absence of heavy metal catalysts also eliminates the costly downstream steps associated with metal scavenging and validation, streamlining the overall production workflow. These factors combine to create a leaner, more cost-effective manufacturing process that can withstand market fluctuations better than traditional methods.
- Enhanced Supply Chain Reliability: The use of Escherichia coli as the host organism leverages a well-established industrial platform with a clear genetic background and mature fermentation technology. This familiarity reduces the risk of technical failures during scale-up and ensures a more reliable supply continuity. Since the pathway relies on common metabolic intermediates and simple carbon sources, the supply chain is less vulnerable to disruptions in the availability of specialized fine chemicals. The robustness of the E. coli system also allows for flexible production scheduling, enabling manufacturers to respond quickly to changes in demand. For supply chain heads, this means a lower risk of stockouts and a more resilient sourcing strategy for critical cofactors.
- Scalability and Environmental Compliance: The biosynthetic nature of this process aligns well with modern environmental standards, as it operates under mild conditions and generates less hazardous waste compared to chemical synthesis. The ability to scale from laboratory flasks to industrial fermenters is supported by the extensive history of E. coli fermentation in the biotech industry. The reduced need for organic solvents and harsh reagents simplifies waste treatment and lowers the environmental footprint of the manufacturing site. This compliance advantage is increasingly important for multinational corporations aiming to meet sustainability goals. The process is designed to be scalable, allowing for a smooth transition from pilot studies to commercial production volumes without significant re-engineering of the core process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel NAD biosynthesis pathway. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing supply chains. The information provided here serves as a foundational guide for further technical discussions and feasibility assessments.
Q: How does this new pathway differ from traditional NAD synthesis?
A: Unlike traditional methods relying on tryptophan or aspartate, this pathway starts from chorismate, decoupling NAD production from essential amino acid metabolism and potentially lowering precursor costs.
Q: What are the key enzymes involved in this biosynthetic route?
A: The core enzymatic cascade includes PhzD, PhzE, DHHA-2,3-dehydrogenase, and NabC, which sequentially convert chorismate to quinolinic acid before entering the salvage pathway.
Q: Is this method suitable for large-scale industrial fermentation?
A: Yes, the use of Escherichia coli as a host organism, combined with a simplified inorganic salt-glucose medium, suggests high feasibility for commercial scale-up and robust supply chain integration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nicotinamide Adenine Dinucleotide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this biosynthetic route for the production of high-value cofactors and are well-positioned to support its commercialization. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to market is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Nicotinamide Adenine Dinucleotide meets the highest industry standards. We understand the critical nature of supply chain continuity for pharmaceutical manufacturers and are committed to delivering consistent quality and reliability. Our team is ready to collaborate with you to optimize this pathway for your specific production needs, leveraging our deep technical expertise to maximize yield and efficiency.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can benefit your specific product portfolio. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic advantages of adopting this technology. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project requirements. Our goal is to be your strategic partner in engineering bottleneck solutions, providing the technical support and manufacturing capacity needed to bring your products to market faster and more cost-effectively. Let us help you navigate the complexities of commercial scale-up and secure a competitive edge in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
