Advanced Synthesis of 6beta7beta-Methylene-Steride-Diol for Commercial API Production
The pharmaceutical industry continuously seeks robust synthetic pathways for complex steroidal intermediates, particularly those serving as precursors to next-generation progestins like Drospirenone. Patent CN101735300A introduces a groundbreaking methodology for preparing 6beta,7beta-methylene-steride-3beta,5beta-diol, addressing critical bottlenecks in traditional steroid functionalization. This technical disclosure outlines a streamlined sequence that bypasses the cumbersome protection groups and harsh oxidation conditions typical of legacy processes. By focusing on direct epoxidation and efficient cyclopropanation, the technology offers a viable route for producing high-purity pharmaceutical intermediates at scale. For R&D teams evaluating process chemistry, this approach represents a significant leap forward in atom economy and operational simplicity. The strategic integration of these steps ensures that the critical 6beta,7beta-methylene structural unit is constructed with exceptional stereocontrol. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances is essential for securing a stable supply chain for hormonal therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 6beta,7beta-methylene motif in steroid skeletons has relied on classical Michael additions using Corey methylenation reagents or Simmons-Smith reactions with Zn-Cu couples on conjugated dienes. While effective on a laboratory scale, these conventional methodologies often suffer from poor stereoselectivity and require extensive downstream purification to remove diastereomeric byproducts. Furthermore, earlier改进 routes necessitated the introduction of a 5beta-hydroxy-Delta6-ene unit through a tedious sequence involving group protection, allylic oxidation, selective reduction, and subsequent elimination. These multi-step sequences not only drive up the cost of goods sold due to reagent consumption but also increase the environmental footprint through excessive solvent use and waste generation. The reliance on expensive oxidants and the need for rigorous control over reaction parameters make these older methods less attractive for commercial-scale manufacturing. Consequently, procurement managers often face volatility in pricing and availability when sourcing intermediates produced via these inefficient legacy pathways.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes a direct and highly efficient strategy starting from readily available steroid-4,6-dien-3-one. The process initiates with a mild reduction to form the corresponding 3beta-alcohol, followed immediately by a stereoselective epoxidation to generate the key 4beta,5beta-epoxy intermediate. This pivotal intermediate serves as a versatile platform for the subsequent construction of the methylene bridge without the need for prior allylic oxidation at the 7-position. By eliminating several synthetic steps associated with traditional Winstein cyclopropanation methods, this new route drastically simplifies the overall operation. The ability to proceed directly from the epoxy-alkenyl alcohol to the target cyclopropane structure demonstrates a profound improvement in step economy. 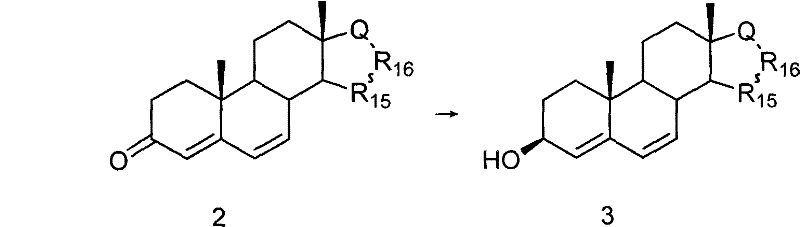
Mechanistic Insights into Stereoselective Cyclopropanation
The core innovation of this synthesis lies in the precise control of stereochemistry during the formation of the cyclopropane ring, which is critical for the biological activity of the final API. The mechanism leverages the directing effect of the 5beta-hydroxyl group established during the reductive ring-opening of the epoxide precursor. This hydroxyl group coordinates with the zinc species during the cyclopropanation step, ensuring that the methylene addition occurs exclusively from the beta-face to yield the desired 6beta,7beta configuration. Such high stereoselectivity is paramount for minimizing the formation of inactive or potentially harmful alpha-isomers, thereby reducing the burden on crystallization and chromatography units. The use of Zn-Cu powder or diethyl zinc as catalysts facilitates this transformation under relatively mild thermal conditions, typically between 10°C and 80°C. This mechanistic elegance ensures that the structural integrity of the sensitive steroid backbone is maintained throughout the rigorous chemical transformations.
Furthermore, the impurity profile is significantly managed by the specificity of the epoxidation reagent, such as m-chloroperoxybenzoic acid (m-CPBA), which selectively targets the Delta4,5 double bond. This selectivity prevents unwanted side reactions at other sites of the steroid nucleus, which are common pitfalls in less refined synthetic routes. The subsequent reductive ring-opening using hydride sources like lithium aluminum hydride proceeds cleanly to set the 5beta-hydroxy stereocenter, which acts as the anchor for the final cyclization. 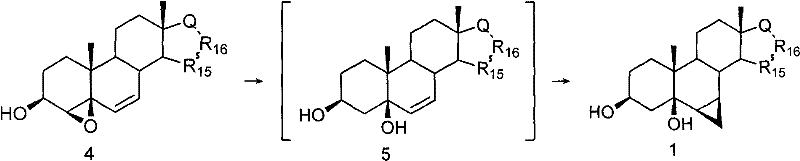
How to Synthesize 6beta7beta-Methylene-Steride-Diol Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and temperature control to maximize yield and purity. The process begins with the reduction of the starting ketone using sodium borohydride in ethanol, followed by extraction and recrystallization to ensure high purity before the epoxidation step. The subsequent epoxidation is conducted in dichloromethane at room temperature, utilizing m-CPBA to install the oxygen bridge with high fidelity. Finally, the cyclopropanation is executed in ethylene glycol dimethyl ether with a Zn-Cu couple and diiodomethane at elevated temperatures to drive the reaction to completion. Detailed standardized synthesis steps see the guide below.
- Reduce steroid-4,6-dien-3-one using sodium borohydride to form steroid-4,6-dien-3beta-ol.
- Perform epoxidation on the dien-ol intermediate using m-CPBA to generate the 4beta5beta-epoxy derivative.
- Execute reductive ring-opening followed by Zn-Cu mediated cyclopropanation to finalize the 6beta7beta-methylene structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible operational efficiencies and risk mitigation. The elimination of complex protection-deprotection sequences reduces the total number of unit operations, which directly correlates to lower labor costs and reduced equipment occupancy time. By utilizing commodity chemicals such as sodium borohydride, zinc powder, and common organic solvents, the dependency on exotic or supply-constrained reagents is minimized. This shift towards widely available raw materials enhances supply chain resilience, ensuring that production schedules are not disrupted by vendor shortages. Additionally, the simplified workflow reduces the potential for human error during manufacturing, leading to more consistent batch-to-batch quality.
- Cost Reduction in Manufacturing: The streamlined nature of this process inherently lowers the cost of goods by reducing solvent consumption and waste disposal requirements associated with additional synthetic steps. Eliminating the need for expensive transition metal catalysts or specialized oxidizing agents further contributes to substantial cost savings in pharmaceutical intermediate manufacturing. The high yields reported in the patent examples indicate that less starting material is wasted, optimizing the overall material balance. These factors combine to create a more economically viable production model that can withstand market price fluctuations.
- Enhanced Supply Chain Reliability: Sourcing intermediates produced via this route mitigates the risk of delays caused by complex logistics for specialty reagents. Since the protocol relies on standard industrial chemicals, suppliers can maintain robust inventory levels without fear of rapid degradation or obsolescence. This reliability is crucial for maintaining continuous API production lines and meeting strict regulatory delivery commitments. Partnerships with manufacturers utilizing this technology ensure a steady flow of high-quality materials essential for downstream drug formulation.
- Scalability and Environmental Compliance: The reaction conditions described, such as moderate temperatures and ambient pressure, are inherently safer and easier to scale from pilot plant to commercial metric-ton quantities. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, lowering the compliance burden for manufacturing facilities. This green chemistry aspect not only improves the corporate sustainability profile but also reduces the costs associated with effluent treatment. Scalability is further supported by the robustness of the Zn-Cu mediated cyclopropanation, which tolerates minor variations in process parameters without compromising product quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and benefits for potential partners. Understanding these details is vital for making informed decisions about process adoption and supplier selection. We encourage technical teams to review these points when evaluating the integration of this intermediate into their supply portfolio.
Q: What are the primary advantages of this synthesis route over conventional Winstein methods?
A: This method eliminates the need for lengthy protection-deprotection sequences and expensive allylic oxidation steps, significantly reducing operational complexity and raw material costs while maintaining high stereoselectivity.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the protocol utilizes common reagents like sodium borohydride and zinc-copper couple under moderate temperature conditions, ensuring safety and scalability for metric-ton production campaigns.
Q: How does this method impact the impurity profile of the final API intermediate?
A: By leveraging high stereoselective epoxidation and directed cyclopropanation, the route minimizes the formation of diastereomeric impurities, simplifying downstream purification and enhancing overall yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6beta7beta-Methylene-Steride-Diol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of hormonal therapeutics. Our CDMO capabilities are specifically designed to translate complex patent methodologies like CN101735300A into robust, commercial-scale processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications to guarantee that every batch meets the highest industry standards. By leveraging our technical expertise, we help clients navigate the challenges of steroid synthesis with confidence and efficiency.
We invite you to collaborate with us to optimize your supply chain and reduce overall production costs through advanced process engineering. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals. Let us be your partner in delivering excellence in pharmaceutical intermediate supply.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
