Advanced Synthesis Of Ether Substituted Imidazopyridines For Commercial Scale-Up Of Complex Pharmaceutical Intermediates
The pharmaceutical landscape continuously demands novel scaffolds capable of modulating immune responses with high efficacy and safety profiles. Patent CN1674894A introduces a significant advancement in this domain by disclosing a series of imidazopyridine compounds featuring unique ether or thioether functionalities at the 1-position. These structural modifications are not merely academic exercises but represent a strategic evolution in the design of immune response modifiers, specifically targeting the induction of cytokine biosynthesis such as interferon-alpha and tumor necrosis factor-alpha. The technical breakthrough lies in the ability to systematically vary the substituents at the 1-position while maintaining the core imidazo[4,5-c]pyridine architecture, which is known for its potent biological activity. This patent provides a comprehensive framework for synthesizing these complex heterocycles, offering a robust pathway for the development of next-generation antiviral and antitumor agents. For industry stakeholders, understanding the nuances of this chemistry is critical for evaluating the feasibility of integrating these intermediates into existing drug development pipelines.
The versatility of the disclosed compounds extends beyond simple structural variation; it encompasses a wide range of therapeutic applications including the treatment of viral diseases and neoplastic conditions. The presence of the ether linkage allows for fine-tuning of physicochemical properties, which is essential for optimizing pharmacokinetic parameters in vivo. By enabling the attachment of diverse side chains through the ether oxygen, chemists can explore a vast chemical space to identify candidates with superior potency and reduced toxicity. This level of molecular precision is paramount in modern drug discovery, where even minor structural changes can lead to significant differences in clinical outcomes. Consequently, the methods described in this patent serve as a foundational tool for researchers aiming to expand the horizon of immunomodulatory therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for imidazopyridine derivatives often suffer from significant limitations regarding regioselectivity and functional group tolerance. Conventional methods typically rely on harsh conditions that can degrade sensitive substituents or lead to complex mixtures of isomers, necessitating extensive and costly purification processes. Furthermore, introducing specific functionalities at the 1-position of the imidazole ring has historically been challenging due to the electronic properties of the precursor pyridines. Many prior art methods fail to provide a generalizable approach that accommodates a broad spectrum of ether or thioether groups, thereby restricting the structural diversity available for medicinal chemistry optimization. These constraints often result in lower overall yields and increased production costs, making the commercial viability of such compounds questionable for large-scale applications.
The Novel Approach
In contrast, the methodology outlined in CN1674894A offers a streamlined and highly adaptable solution to these longstanding challenges. By utilizing a stepwise approach that begins with readily available 2,4-dihydroxy-3-nitropyridines, the process ensures high regiocontrol during the initial functionalization steps. The strategic use of chlorinating agents followed by nucleophilic displacement with protected amino alcohols allows for the precise installation of the ether linkage before the formation of the imidazole ring. This sequence minimizes side reactions and protects sensitive functional groups until the final stages of synthesis. The novelty of this approach lies in its modularity; different ether chains can be introduced simply by varying the amino alcohol starting material, enabling rapid generation of analog libraries without redesigning the entire synthetic route. This flexibility is a game-changer for accelerating lead optimization programs.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
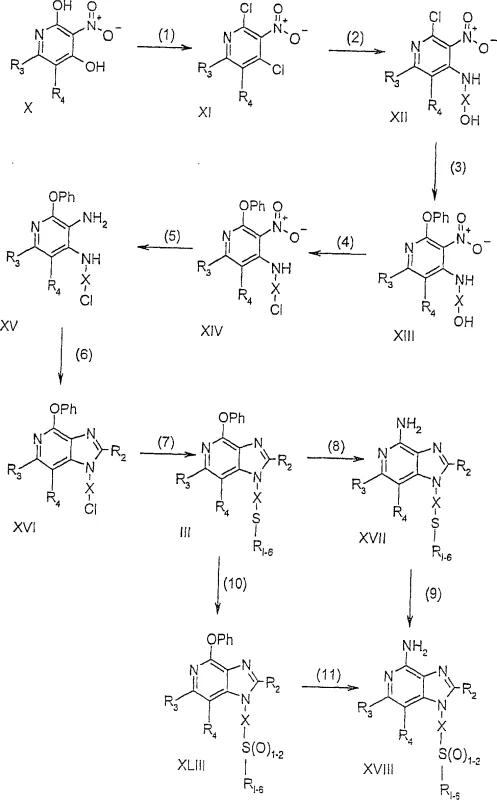
The core of the synthetic strategy involves a sophisticated sequence of transformations that convert simple pyridine precursors into the target imidazopyridine scaffold. The process initiates with the chlorination of the pyridine ring using phosphorus oxychloride, which activates the positions for subsequent nucleophilic attack. This is followed by the displacement of the chloro group with an amino alcohol, a step that requires careful control of temperature and base to prevent over-reaction. The resulting intermediate then undergoes a phenoxide displacement to install the ether moiety, a critical step that defines the unique properties of the final compound. The reduction of the nitro group to an amine is subsequently performed using catalytic hydrogenation or chemical reducing agents, setting the stage for the crucial cyclization event.
The cyclization step itself is a masterpiece of heterocyclic chemistry, involving the condensation of the diamine intermediate with an orthoester or acyl halide. This reaction forms the imidazole ring fused to the pyridine core, completing the tricyclic system. The mechanism proceeds through the formation of an amidine intermediate which then cyclizes under thermal conditions, often facilitated by acid catalysts. The final amination of the 4-position is achieved by heating with ammonium acetate, a mild yet effective reagent that ensures high conversion without compromising the integrity of the ether side chain. Throughout this sequence, impurity control is maintained by leveraging the differential solubility of intermediates and employing rigorous purification techniques such as recrystallization. This meticulous attention to detail ensures that the final product meets the stringent purity requirements necessary for pharmaceutical applications.
How to Synthesize Ether Substituted Imidazopyridines Efficiently
The synthesis of these high-value intermediates requires a disciplined approach to reaction conditions and workup procedures to ensure consistent quality and yield. The process begins with the activation of the pyridine ring, followed by the sequential introduction of the nitrogen and oxygen functionalities that define the molecule's architecture. Each step must be monitored closely using analytical techniques such as HPLC and NMR to confirm conversion and identify any potential byproducts. The detailed standardized synthesis steps provided in the guide below outline the specific reagents, temperatures, and durations required to replicate the patent's success on a laboratory scale. Adhering to these protocols is essential for achieving the reproducibility needed for technology transfer and scale-up activities.
- Chlorination of 2,4-dihydroxy-3-nitropyridine using phosphorus oxychloride to form 2,4-dichloro-3-nitropyridine intermediates.
- Nucleophilic substitution with amino alcohols followed by phenoxide displacement to introduce ether functionality at the pyridine ring.
- Catalytic reduction of nitro groups and subsequent cyclization with orthoesters to form the imidazo[4,5-c]pyridine core structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the synthetic route described in this patent offers substantial advantages that directly impact the bottom line and supply chain resilience. The use of commodity chemicals such as phosphorus oxychloride, phenol, and ammonium acetate ensures that raw material costs remain stable and predictable, shielding manufacturers from the volatility associated with exotic reagents. Furthermore, the modular nature of the synthesis allows for the production of multiple analogs using a common set of intermediates, which significantly reduces inventory complexity and warehousing costs. This efficiency translates into a more agile supply chain capable of responding quickly to changing market demands without the need for extensive retooling or process redevelopment. The robustness of the chemistry also minimizes the risk of batch failures, ensuring a steady flow of material to downstream formulation teams.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in key steps, relying instead on thermal cyclization and simple acid-base chemistry, leads to significant cost savings in reagent procurement and waste disposal. The process avoids the need for specialized equipment required for high-pressure hydrogenation in all steps, as alternative chemical reduction methods are viable, further lowering capital expenditure requirements. Additionally, the high selectivity of the reactions reduces the burden on purification units, saving both time and solvent consumption during the isolation of the final product. These cumulative efficiencies result in a lower cost of goods sold, making the technology economically attractive for generic and branded drug manufacturers alike.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials mitigates the risk of supply disruptions that often plague specialized chemical supply chains. Since the intermediates are stable and can be stored for extended periods without degradation, manufacturers can maintain strategic stockpiles to buffer against unforeseen logistical challenges. The scalability of the process from gram to kilogram scales has been demonstrated through the variety of examples provided, confirming that the chemistry holds up under increased production loads. This reliability is crucial for maintaining continuous manufacturing operations and meeting strict delivery commitments to global pharmaceutical partners.
- Scalability and Environmental Compliance: The synthetic pathway is designed with environmental considerations in mind, utilizing solvents that can be readily recovered and recycled, thereby reducing the overall environmental footprint of the manufacturing process. The absence of heavy metal residues in the final product simplifies the regulatory approval process and reduces the need for extensive metal scavenging steps. Moreover, the reaction conditions are generally mild, operating at atmospheric pressure and moderate temperatures, which enhances operational safety and reduces energy consumption. These factors collectively contribute to a sustainable manufacturing model that aligns with modern green chemistry principles and regulatory expectations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. They are derived from a thorough analysis of the patent's background and the specific advantages highlighted in the examples. Understanding these aspects is vital for decision-makers evaluating the potential integration of these intermediates into their product portfolios. The answers provided reflect the technical realities of the process and the strategic benefits it offers to the pharmaceutical industry.
Q: What are the key advantages of the ether substitution at the 1-position?
A: The ether functionality at the 1-position enhances solubility and metabolic stability compared to traditional alkyl substitutions, offering improved bioavailability for immune response modulation.
Q: Is the synthesis route scalable for commercial production?
A: Yes, the patent describes robust conditions using common reagents like phosphorus oxychloride and ammonium acetate, which are suitable for large-scale manufacturing with established safety protocols.
Q: How is impurity control managed during the cyclization step?
A: Impurity control is achieved through precise temperature regulation during the reflux cyclization and subsequent purification via recrystallization or column chromatography to ensure high purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ether Substituted Imidazopyridines Supplier
The technical potential of the synthetic route described in CN1674894A is immense, offering a clear path to high-quality immune response modifiers. NINGBO INNO PHARMCHEM stands ready to leverage this intellectual property, bringing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of Ether Substituted Imidazopyridines meets the exacting standards required for clinical and commercial use. We understand the critical nature of supply continuity in the pharmaceutical sector and have built our operations to guarantee reliability and consistency.
We invite you to initiate a dialogue regarding your specific supply chain optimization needs. Our technical procurement team is prepared to provide a Customized Cost-Saving Analysis tailored to your project requirements. Please contact us to request specific COA data and route feasibility assessments that will demonstrate how our capabilities align with your development goals. Let us partner with you to bring these innovative therapeutics to market efficiently and effectively.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
