Optimizing Antibiotic Quality Control: Scalable Synthesis of Cefditoren Pivoxil Ring-Opening Impurity
The pharmaceutical industry operates under increasingly stringent regulatory frameworks where the characterization of impurities is not merely a suggestion but a mandatory requirement for market approval. Patent CN110143957B introduces a pivotal advancement in the quality assurance of third-generation cephalosporin antibiotics by detailing a robust preparation method for the Cefditoren Pivoxil ring-opening product. This specific compound acts as a critical reference standard, enabling manufacturers to accurately quantify degradation pathways that could compromise patient safety. As global demand for high-quality generic antibiotics rises, the ability to synthesize these complex impurity standards with high purity and reproducibility becomes a strategic asset for any reliable pharmaceutical intermediate supplier. The disclosed methodology shifts the paradigm from inefficient isolation to directed synthesis, offering a scalable solution that aligns with modern Good Manufacturing Practice (GMP) expectations.
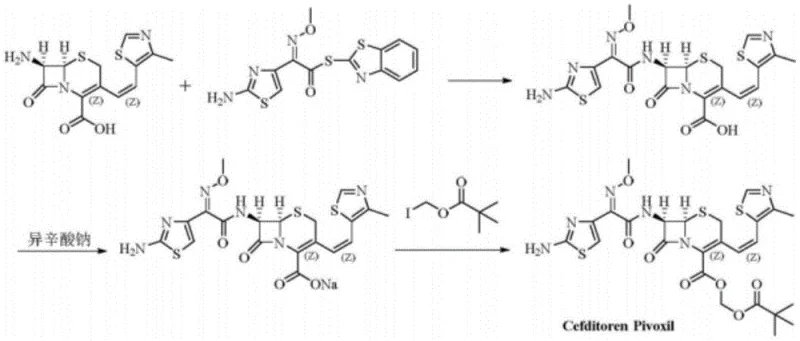
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of specific impurity reference standards like the Cefditoren Pivoxil ring-opening product has been a significant bottleneck in analytical development. Traditional approaches relied heavily on isolating these trace components directly from crude API batches or forced degradation studies. This method is inherently flawed due to the extremely low concentration of the target impurity within the crude matrix, leading to dismal recovery rates and extensive purification challenges. Furthermore, the structural similarity between the ring-opening product and other process-related impurities often results in co-elution during chromatographic separation, making it nearly impossible to obtain a reference standard with the requisite purity for validation. For procurement managers, relying on such inconsistent sources translates to unpredictable lead times and inflated costs associated with repetitive purification attempts.
The Novel Approach
The innovation described in the patent circumvents these inefficiencies by employing a targeted chemical transformation rather than passive isolation. By utilizing Cefditoren Pivoxil as the starting material and subjecting it to controlled alkaline conditions, the process deliberately induces the beta-lactam ring opening to generate the target impurity as the major product. This strategic inversion of the problem transforms a trace contaminant into a primary synthetic output. The result is a dramatic improvement in both yield and purity, with the patent reporting content levels exceeding 95.0% and isolated yields reaching 70%. This approach not only secures a steady supply of the reference standard but also significantly reduces the cost reduction in pharmaceutical intermediates manufacturing by eliminating the need for processing tons of crude material to extract milligrams of standard.
Mechanistic Insights into Alkaline Hydrolysis of Beta-Lactams
The core of this synthesis lies in the nucleophilic attack on the strained four-membered beta-lactam ring, a structural motif common to all cephalosporins. Under the specified conditions, the hydroxide ion or other alkaline species acts as a potent nucleophile, targeting the carbonyl carbon of the lactam ring. The reaction is initiated at cryogenic temperatures, typically between -20°C and -5°C, to modulate the reaction kinetics and prevent non-selective degradation of other sensitive functional groups, such as the vinyl thiazole moiety or the ester side chains. As the reaction progresses and the temperature is allowed to rise to 10°C-20°C, the ring opens irreversibly to form the corresponding amino acid derivative. Understanding this mechanistic pathway is crucial for R&D directors aiming to replicate the process, as slight deviations in pH or temperature can lead to the formation of secondary degradation products like the delta-3 isomers or dimeric species.
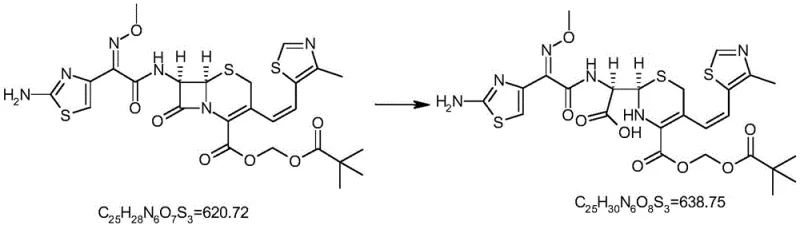
Controlling the impurity profile during this synthesis requires a deep understanding of the solvent effects and base strength. The patent highlights the utility of polar aprotic solvents such as DMSO, DMF, or N-methylpyrrolidone, which enhance the solubility of the lipophilic cephalosporin substrate while stabilizing the transition state of the hydrolysis. The choice of base—ranging from sodium hydroxide to potassium carbonate—is equally critical; stronger bases may accelerate the reaction but risk over-hydrolysis, whereas weaker bases might require longer reaction times that could promote isomerization. By optimizing these parameters, the process ensures that the resulting ring-opening product retains the stereochemical integrity of the original molecule, a factor that is paramount for its effectiveness as an analytical standard in chiral HPLC methods.
How to Synthesize Cefditoren Pivoxil Ring-Opening Product Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing this critical reference standard with high fidelity. The process begins with the dissolution of the API in a suitable organic solvent followed by the precise addition of a base under strict thermal control. This method is designed to be robust enough for laboratory scale-up while maintaining the purity levels required for regulatory submission. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below which encapsulates the critical process steps.
- Dissolve Cefditoren Pivoxil in a polar aprotic solvent like DMSO or DMF and cool the mixture to between -20°C and -5°C.
- Slowly add a stoichiometric amount of alkaline substance such as sodium hydroxide or potassium carbonate while maintaining low temperature.
- Warm the reaction to 10°C-20°C to complete the ring-opening, then extract with ethyl acetate and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders managing the supply chain of antibiotic production, the availability of high-quality impurity standards is a linchpin for maintaining continuous manufacturing operations. Without validated reference materials, batch release testing cannot be completed, potentially halting the distribution of life-saving medications. The adoption of this directed synthesis method offers substantial commercial advantages by decoupling the supply of reference standards from the variability of API production batches. This independence ensures that quality control laboratories have consistent access to the materials they need, thereby reducing the risk of supply chain disruptions caused by analytical bottlenecks.
- Cost Reduction in Manufacturing: The shift from isolation to synthesis eliminates the massive material loss associated with extracting trace impurities from crude products. By achieving high yields through a direct chemical route, the overall cost of goods for the reference standard is significantly lowered. Furthermore, the simplified workup procedure involving standard extraction and silica gel chromatography avoids the need for expensive preparative HPLC equipment or specialized resin columns, leading to substantial operational savings.
- Enhanced Supply Chain Reliability: Relying on isolation methods ties the availability of the standard to the production schedule of the API itself, creating a dependency that can delay quality testing. This synthetic route allows for the independent production of the impurity standard on demand. This flexibility ensures that reducing lead time for high-purity pharmaceutical intermediates becomes achievable, as the reference material can be manufactured in parallel with the API, ensuring that QC protocols are never stalled due to a lack of standards.
- Scalability and Environmental Compliance: The reaction conditions described utilize common industrial solvents and reagents that are readily available on a global scale, facilitating easy scale-up from gram to kilogram quantities. The process avoids the use of exotic catalysts or hazardous reagents that would complicate waste disposal. The straightforward aqueous workup and standard chromatographic purification align well with environmental health and safety guidelines, making the commercial scale-up of complex pharmaceutical intermediates both economically and environmentally sustainable.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the stability, application, and regulatory significance of the Cefditoren Pivoxil ring-opening product. These insights are derived directly from the patent specifications and are intended to assist technical teams in implementing this standard within their quality control frameworks.
Q: Why is the Cefditoren Pivoxil ring-opening product critical for quality control?
A: This specific degradation product serves as a vital reference standard for HPLC analysis. Regulatory bodies require precise quantification of impurities exceeding 0.1% to ensure patient safety and drug efficacy.
Q: How does this synthetic method improve upon traditional isolation techniques?
A: Traditional isolation from crude API batches yields low purity and poor recovery. This directed synthesis achieves over 95% content and 70% yield, providing a reliable source of reference material.
Q: What are the key parameters for controlling selectivity in this reaction?
A: Precise temperature control during the addition of the base (-20°C to -5°C) and careful selection of the solvent system are essential to prevent over-degradation and ensure the formation of the specific ring-opened structure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefditoren Pivoxil Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your final drug product depends on the precision of your analytical controls. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that even complex impurity standards like the Cefditoren Pivoxil ring-opening product are available in the quantities you need. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of reference material meets the highest international standards, providing you with the confidence to validate your methods effectively.
We invite you to collaborate with us to optimize your supply chain for antibiotic quality control. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific impurity profiling needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your regulatory compliance and production goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
