Advanced Synthesis of Cefditoren Pivoxil Dimer for High-Purity Quality Control Standards
Advanced Synthesis of Cefditoren Pivoxil Dimer for High-Purity Quality Control Standards
In the highly regulated landscape of pharmaceutical manufacturing, the integrity of Active Pharmaceutical Ingredients (APIs) is paramount, necessitating rigorous quality control measures that rely heavily on accurate impurity profiling. Patent CN108727418B introduces a groundbreaking preparation method for cefditoren pivoxil dimer, a critical related substance that serves as an essential reference standard for the third-generation cephalosporin antibiotic, cefditoren pivoxil. Traditionally, obtaining this specific dimeric impurity involved laborious separation and extraction from crude reaction mixtures, a process plagued by low recovery rates and insufficient purity levels that compromise analytical accuracy. This novel technical disclosure presents a direct, acid-catalyzed condensation strategy that transforms the parent API into the dimer with exceptional efficiency, addressing a significant bottleneck in the quality assurance workflows of global pharmaceutical producers. By shifting from extraction to synthesis, the industry gains access to a reliable source of high-purity reference materials, ensuring that safety and efficacy standards for this vital antibacterial agent are consistently met across all production batches.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of cefditoren pivoxil dimer for analytical calibration has been hindered by the inherent inefficiencies of isolating trace impurities from bulk drug substances. Conventional methodologies typically rely on preparative chromatography or repeated crystallization of crude cefditoren pivoxil, processes that are not only time-consuming but also economically draining due to the substantial loss of material. The structural similarity between the target dimer and other process-related impurities makes complete separation exceedingly difficult, often resulting in reference standards that contain co-eluting contaminants which skew HPLC quantification results. Furthermore, the variability of crude batches means that the availability of the dimer is inconsistent, creating supply chain vulnerabilities for quality control laboratories that require steady access to certified reference materials. These traditional extraction-based approaches fail to provide the scalability needed for industrial applications, forcing manufacturers to accept lower purity thresholds or invest disproportionately in purification infrastructure to meet regulatory specifications for impurity identification.
The Novel Approach
The methodology outlined in patent CN108727418B revolutionizes this landscape by employing a direct synthetic route where cefditoren pivoxil reacts with a formaldehyde solution under controlled acidic conditions to generate the dimer explicitly. This approach bypasses the need for complex isolation techniques, instead leveraging a straightforward condensation reaction that links two cephalosporin molecules via a methylene bridge, as illustrated in the reaction scheme below. 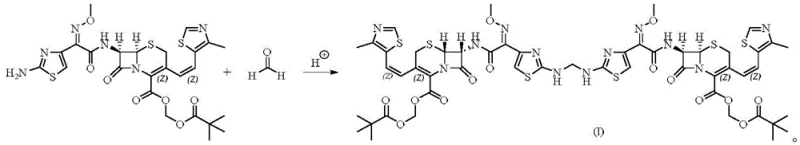 The process utilizes common organic solvents like acetonitrile and inexpensive catalysts such as sulfuric acid, operating at moderate temperatures between 50°C and 100°C to drive the reaction to completion with remarkable selectivity. Experimental data from the patent indicates that this method achieves yields exceeding 87% with purity levels surpassing 96%, a dramatic improvement over the meager recoveries associated with extraction methods. By controlling the stoichiometry of the formaldehyde feed and the acidity of the medium, the synthesis minimizes the formation of polymeric byproducts, ensuring that the resulting dimer is suitable for immediate use as a high-precision reference standard without extensive downstream processing.
The process utilizes common organic solvents like acetonitrile and inexpensive catalysts such as sulfuric acid, operating at moderate temperatures between 50°C and 100°C to drive the reaction to completion with remarkable selectivity. Experimental data from the patent indicates that this method achieves yields exceeding 87% with purity levels surpassing 96%, a dramatic improvement over the meager recoveries associated with extraction methods. By controlling the stoichiometry of the formaldehyde feed and the acidity of the medium, the synthesis minimizes the formation of polymeric byproducts, ensuring that the resulting dimer is suitable for immediate use as a high-precision reference standard without extensive downstream processing.
Mechanistic Insights into Acid-Catalyzed Condensation
The core chemical transformation driving this synthesis is an electrophilic substitution facilitated by the activation of formaldehyde in an acidic environment. In the presence of a proton donor like sulfuric acid, the carbonyl oxygen of formaldehyde becomes protonated, generating a highly reactive electrophilic species that attacks the electron-rich centers on the cefditoren pivoxil molecule. Specifically, the reaction likely targets the amino group on the thiazole ring or other nucleophilic sites within the cephalosporin nucleus, leading to the formation of a methylene-linked dimer. The choice of solvent plays a critical role in stabilizing the transition states and solubilizing the relatively polar cephalosporin substrate; acetonitrile is identified as the optimal medium due to its ability to dissolve both the organic API and the aqueous formaldehyde solution while maintaining thermal stability at the required reaction temperatures. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as the molar ratio of reactants and the strength of the acid catalyst, to maximize the formation of the desired dimer while suppressing competing side reactions that could lead to oligomerization or degradation of the sensitive beta-lactam ring.
Controlling the impurity profile is just as critical as maximizing yield, particularly when the product itself is intended to serve as a standard for impurity detection. The patent details a robust workup procedure involving liquid-liquid extraction with ethyl acetate and water, followed by washing with saturated saline and drying over anhydrous sodium sulfate, which effectively removes unreacted formaldehyde, residual acid catalyst, and polar byproducts.  Recrystallization from dichloromethane further refines the product, eliminating trace isomers and ensuring the final solid meets the stringent purity requirements necessary for analytical certification. This meticulous attention to purification mechanics ensures that the synthesized dimer does not introduce new variables into the quality control equation, providing a clean baseline for HPLC methods used to monitor the safety of the final antibiotic formulation. The ability to predict and manage these impurity pathways demonstrates a sophisticated grasp of cephalosporin chemistry, offering a reproducible protocol that can be validated across different manufacturing scales.
Recrystallization from dichloromethane further refines the product, eliminating trace isomers and ensuring the final solid meets the stringent purity requirements necessary for analytical certification. This meticulous attention to purification mechanics ensures that the synthesized dimer does not introduce new variables into the quality control equation, providing a clean baseline for HPLC methods used to monitor the safety of the final antibiotic formulation. The ability to predict and manage these impurity pathways demonstrates a sophisticated grasp of cephalosporin chemistry, offering a reproducible protocol that can be validated across different manufacturing scales.
How to Synthesize Cefditoren Pivoxil Dimer Efficiently
Implementing this synthesis route requires careful adherence to the optimized conditions established in the patent to ensure consistent batch-to-batch quality. The process begins with the dissolution of cefditoren pivoxil in a selected organic solvent, followed by the precise addition of formaldehyde and the acid catalyst, after which the mixture is heated to promote the condensation reaction. Once the reaction reaches completion, indicated by HPLC monitoring, the product is isolated through a series of extraction and washing steps designed to remove inorganic salts and unreacted starting materials before final purification via recrystallization. For a detailed breakdown of the specific reagent quantities, temperature profiles, and safety precautions required for this transformation, please refer to the standardized synthesis guide provided below.
- Dissolve cefditoren pivoxil in an organic solvent such as acetonitrile and add a formaldehyde solution along with an acid catalyst like sulfuric acid.
- Heat the reaction mixture to a temperature between 50°C and 100°C, preferably around 80°C, and maintain stirring for 1 to 4 hours until conversion is complete.
- Cool the reaction, perform liquid-liquid extraction with ethyl acetate and water, wash the organic phase, dry over sodium sulfate, and recrystallize the residue to obtain high-purity dimer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic method represents a strategic opportunity to optimize the cost structure and reliability of reference material sourcing. By replacing unpredictable extraction processes with a defined chemical synthesis, organizations can eliminate the variability associated with crude drug supplies and reduce the operational overhead linked to complex purification workflows. This shift not only streamlines the internal production of quality control standards but also opens avenues for external suppliers to offer these critical materials at more competitive price points due to the simplified manufacturing logic. The reliance on commodity chemicals like formaldehyde and sulfuric acid, rather than specialized reagents, further insulates the supply chain from raw material volatility, ensuring long-term stability in production planning and inventory management for pharmaceutical quality assurance departments.
- Cost Reduction in Manufacturing: The elimination of extensive chromatographic separation steps significantly lowers the operational costs associated with producing the dimer reference standard. Traditional extraction methods require large volumes of solvents and expensive stationary phases to achieve marginal purity, whereas this direct synthesis coupled with simple crystallization drastically reduces solvent consumption and waste disposal expenses. Furthermore, the high yield reported in the patent examples means that less starting API is wasted, maximizing the value derived from every kilogram of cefditoren pivoxil utilized in the process and contributing to substantial overall cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Dependence on the isolation of trace impurities from production batches creates a bottleneck where the availability of reference standards is tied to the schedule of API manufacturing runs. This independent synthetic route decouples the supply of the dimer from the main production line, allowing for dedicated campaigns that can be scheduled based on demand rather than production surplus. The use of stable, shelf-stable reagents ensures that production can proceed without interruption, mitigating the risk of stockouts that could delay critical quality release testing and potentially impact the time-to-market for the final antibiotic product.
- Scalability and Environmental Compliance: The reaction conditions described are inherently scalable, utilizing standard reactor equipment and moderate temperatures that do not require specialized high-pressure or cryogenic infrastructure. This ease of scale-up facilitates the transition from laboratory gram-scale synthesis to multi-kilogram commercial production, supporting the growing demand for high-purity impurity standards globally. Additionally, the simplified workup procedure generates less hazardous waste compared to repetitive chromatography, aligning with modern green chemistry initiatives and reducing the environmental footprint of the quality control supply chain, which is increasingly important for regulatory compliance and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of cefditoren pivoxil dimer, drawing directly from the experimental data and process descriptions found in the patent literature. These insights are designed to clarify the advantages of the new synthetic method for stakeholders involved in pharmaceutical development and quality assurance. Understanding these details is crucial for making informed decisions about sourcing strategies and process validation protocols.
Q: Why is synthetic cefditoren pivoxil dimer preferred over isolation from crude products?
A: Isolation from crude products often results in low yields and difficulty in separating structurally similar impurities. The synthetic method described in patent CN108727418B provides a direct route with significantly higher purity (over 95%) and yield, ensuring reliable quantification for quality control.
Q: What are the critical reaction parameters for maximizing dimer yield?
A: The patent identifies acetonitrile as the preferred solvent and sulfuric acid as the optimal catalyst. Maintaining a reaction temperature of approximately 80°C and a specific molar ratio of cefditoren pivoxil to formaldehyde (around 1:0.14 mass ratio) is crucial for achieving yields up to 87.6%.
Q: How does this synthesis method impact supply chain stability for reference standards?
A: By utilizing readily available starting materials like formaldehyde and the API itself, rather than relying on complex extraction processes, manufacturers can ensure a consistent and scalable supply of the reference standard, reducing lead times and dependency on variable crude batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefditoren Pivoxil Dimer Supplier
At NINGBO INNO PHARMCHEM, we recognize that the precision of your quality control data is only as good as the reference standards you employ, which is why we have integrated advanced synthetic methodologies like the one described in CN108727418B into our service portfolio. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients with consistency and speed. We operate state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of cefditoren pivoxil dimer we supply meets the highest international standards for analytical accuracy and reliability.
We invite you to collaborate with us to optimize your supply chain for critical impurity standards and leverage our technical expertise for your custom synthesis needs. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your operational efficiency and support your commitment to drug safety and quality.
