Scalable Synthesis of Myrtenal Dihydrazides: A Breakthrough in Agrochemical Manufacturing
Scalable Synthesis of Myrtenal Dihydrazides: A Breakthrough in Agrochemical Manufacturing
The agricultural chemical industry is currently witnessing a paradigm shift towards leveraging renewable natural products for the development of next-generation crop protection agents. Patent CN113121426B introduces a highly efficient synthetic methodology for a novel class of myrtenal-based diacylhydrazide compounds, which demonstrate potent antifungal activity. This technology represents a significant advancement by integrating the robust bicyclic skeleton of alpha-pinene, derived from abundant turpentine resources, with the biologically active diacylhydrazide pharmacophore. The strategic combination of these structural motifs results in molecules that not only possess enhanced biological efficacy against a broad spectrum of plant pathogenic fungi but also offer a sustainable alternative to traditional petrochemical-derived pesticides. For R&D directors and procurement specialists, this patent outlines a pathway to high-value agrochemical intermediates that align with modern green chemistry principles while maintaining economic viability.
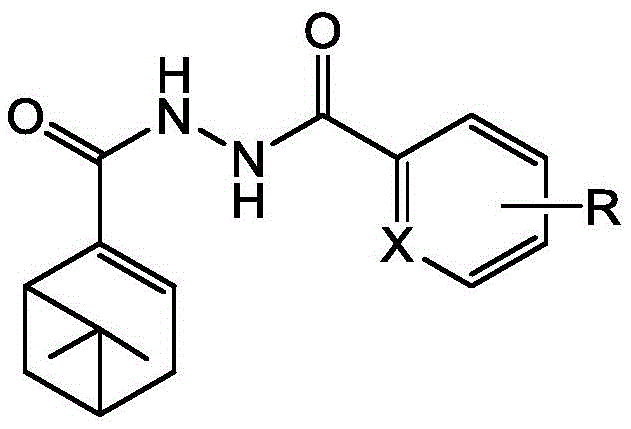
Furthermore, the structural versatility allowed by this synthetic route enables the rapid generation of diverse analogues through simple substitution on the aromatic or heteroaromatic rings. This modularity is crucial for lead optimization programs aimed at overcoming resistance issues in field applications. The core innovation lies in the preservation of the chiral integrity of the pinene skeleton throughout the multi-step transformation, ensuring that the final products retain the stereochemical features necessary for high-affinity binding to biological targets. As global regulatory pressures intensify regarding residue levels and environmental toxicity, the development of such bio-based fungicides provides a compelling value proposition for companies seeking to diversify their product portfolios with environmentally benign yet highly effective solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing complex diacylhydrazide fungicides often rely on fully synthetic routes starting from basic petrochemical feedstocks, which can be energetically intensive and generate significant waste streams. Many existing methods require harsh reaction conditions, such as extreme temperatures or the use of hazardous reagents that complicate downstream purification and waste management. Additionally, constructing the specific bicyclic terpene framework from scratch involves multiple steps with low overall atom economy, leading to inflated production costs and supply chain vulnerabilities associated with fluctuating raw material prices. The reliance on non-renewable resources also conflicts with the increasing corporate sustainability mandates that drive modern procurement strategies in the agrochemical sector. Consequently, there is a critical need for methodologies that can access these privileged scaffolds more directly and sustainably.
The Novel Approach
The methodology disclosed in the patent addresses these challenges by utilizing alpha-pinene, a readily available and inexpensive terpene, as the starting material for a streamlined semi-synthetic sequence. This approach capitalizes on the inherent structural complexity of the natural product, effectively bypassing the need for complex ring-forming reactions that typically plague total synthesis efforts. The process involves a logical progression of functional group transformations: selective allylic oxidation to install the aldehyde functionality, followed by oxidation to the carboxylic acid, activation to the acid chloride, and finally coupling with various hydrazides. This linear and convergent strategy minimizes the number of isolation steps and maximizes yield efficiency. By anchoring the synthesis on a renewable feedstock, the process inherently reduces the carbon footprint of the manufacturing operation while ensuring a stable supply of raw materials sourced from the forestry industry.
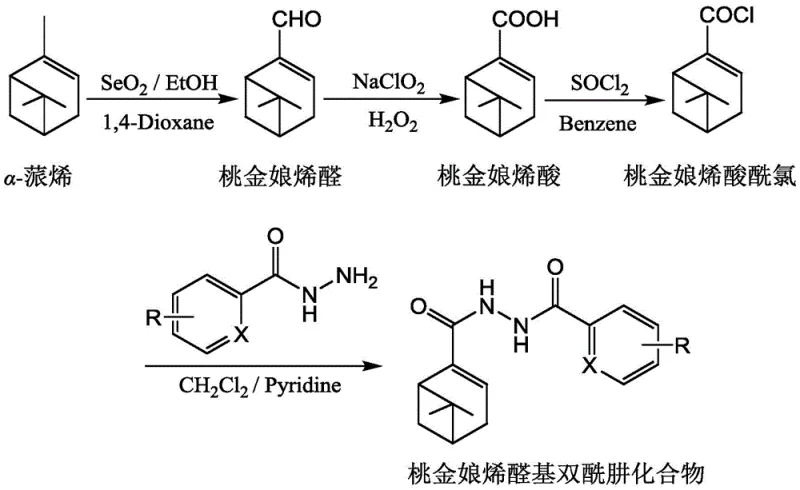
Moreover, the reaction conditions employed in this novel route are notably mild and compatible with standard industrial equipment. The use of selenium dioxide for the initial allylic oxidation is a well-established transformation that offers high regioselectivity for the allylic position, crucial for maintaining the integrity of the double bond within the ring system. Subsequent oxidation using sodium chlorite is performed under buffered aqueous conditions, which prevents over-oxidation or degradation of the sensitive terpene skeleton. The final acylation step proceeds smoothly at room temperature or with mild cooling, eliminating the need for energy-intensive heating protocols. This operational simplicity translates directly into reduced capital expenditure for reactor specifications and lower utility costs, making the process highly attractive for commercial scale-up in existing multipurpose facilities.
Mechanistic Insights into Selective Oxidation and Acylation
The chemical elegance of this synthesis is rooted in the precise control of oxidation states and the chemoselectivity of the reagents employed. The initial transformation of alpha-pinene to myrtenal utilizes selenium dioxide, which operates via a mechanism involving the formation of a selenite ester intermediate at the allylic position. This intermediate undergoes a syn-elimination to deliver the allylic alcohol, which is subsequently oxidized to the aldehyde under the reaction conditions. The presence of hydroquinone as an additive is critical, as it serves to inhibit polymerization of the reactive alkene moiety, thereby preserving the yield and purity of the myrtenal intermediate. This step is fundamental because any loss of the double bond or rearrangement of the bicyclic system would render the final product biologically inactive. The careful control of temperature and addition rates ensures that the exothermic nature of the oxidation does not lead to runaway reactions or the formation of tarry byproducts.
Following the formation of myrtenal, the conversion to myrtenic acid is achieved using sodium chlorite in the presence of a phosphate buffer and hydrogen peroxide. This variation of the Pinnick oxidation is particularly advantageous for substrates containing acid-sensitive functionalities or conjugated systems. The mechanism involves the generation of chlorous acid in situ, which selectively oxidizes the aldehyde to the carboxylic acid without affecting the electron-rich double bond of the pinene ring. This chemoselectivity is paramount, as alternative strong oxidants like potassium permanganate or chromic acid would likely cleave the double bond or degrade the bicyclic framework. The resulting myrtenic acid is then activated using thionyl chloride to form the corresponding acid chloride, a highly reactive electrophile that facilitates the final coupling reaction. The use of anhydrous conditions during this activation step prevents hydrolysis of the acid chloride, ensuring maximum efficiency in the subsequent nucleophilic attack by the hydrazide nitrogen.
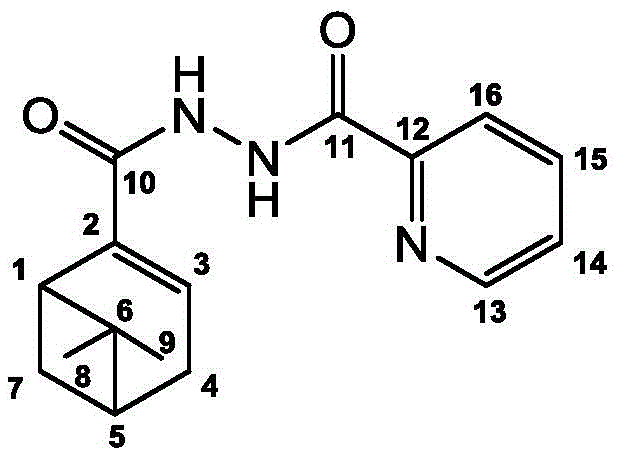
In the final coupling stage, the nucleophilic attack of the substituted hydrazide on the myrtenoyl chloride is facilitated by the presence of a base, such as pyridine, which scavenges the generated hydrochloric acid. This acid-base neutralization drives the equilibrium towards the formation of the diacylhydrazide bond. The structural integrity of the final products, such as Compound 1 shown above, is rigorously confirmed through spectroscopic analysis, including NMR and mass spectrometry, which verify the connectivity between the terpene moiety and the aromatic/heteroaromatic head group. The ability to tolerate various substituents (R groups) on the hydrazide component allows for fine-tuning of the physicochemical properties, such as lipophilicity and electronic distribution, which are key determinants of membrane permeability and target binding affinity in biological systems. This mechanistic understanding underscores the robustness of the synthetic design and its suitability for producing high-purity intermediates required for regulatory approval.
How to Synthesize Myrtenal Dihydrazide Efficiently
The practical execution of this synthesis requires attention to detail regarding reagent quality and moisture control, particularly during the acid chloride formation and coupling steps. The protocol outlined in the patent provides a reproducible framework that balances reaction kinetics with safety considerations, utilizing common solvents like dichloromethane and ethyl acetate that are easily recovered and recycled. Operators should be mindful of the exothermic nature of the oxidation steps and employ appropriate cooling measures to maintain selectivity. The workup procedures involving extraction and distillation are designed to remove inorganic salts and unreacted starting materials effectively, yielding products with high HPLC purity suitable for biological testing. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized guide below.
- Selective allylic oxidation of alpha-pinene using selenium dioxide to generate myrtenal.
- Oxidation of myrtenal to myrtenic acid using sodium chlorite under buffered conditions.
- Conversion of myrtenic acid to myrtenoyl chloride using thionyl chloride.
- Coupling of myrtenoyl chloride with substituted hydrazides to form the final diacylhydrazide product.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits centered around raw material security and process economics. By anchoring the production on alpha-pinene, a derivative of turpentine, manufacturers can decouple their supply chains from the volatility of the petrochemical market. Turpentine is a renewable resource with a stable global supply, primarily sourced from the paper and pulp industry, which ensures long-term availability and price stability for the key starting material. This resilience is critical for maintaining continuous production schedules and meeting the just-in-time delivery requirements of large-scale agrochemical formulators. Furthermore, the simplicity of the synthetic sequence reduces the dependency on specialized catalysts or exotic reagents that often suffer from supply bottlenecks or long lead times.
- Cost Reduction in Manufacturing: The streamlined nature of the synthesis significantly lowers the operational expenditure associated with producing these high-value intermediates. By reducing the total number of synthetic steps and avoiding the use of expensive transition metal catalysts, the process minimizes both material costs and waste disposal fees. The high atom economy of the coupling reaction ensures that the majority of the input mass is incorporated into the final product, reducing the burden on downstream purification units. Additionally, the ability to perform reactions at or near ambient temperatures reduces energy consumption for heating and cooling, contributing to a lower overall cost of goods sold. These efficiencies allow for competitive pricing strategies without compromising on the quality or purity of the final agrochemical active.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard unit operations enhances the robustness of the supply chain against external disruptions. Solvents such as dichloromethane, ethyl acetate, and benzene are widely available from multiple global suppliers, mitigating the risk of single-source dependency. The synthetic route does not require specialized high-pressure equipment or cryogenic conditions, meaning it can be manufactured in a wide range of existing chemical facilities without the need for significant capital investment in new infrastructure. This flexibility allows for geographic diversification of production sites, further securing the supply line against regional logistical challenges or regulatory changes. Consequently, customers can expect consistent lead times and reliable volume availability to support their own formulation and distribution networks.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram to multi-kilogram scales in the patent examples. The use of established separation techniques like distillation and liquid-liquid extraction facilitates easy scale-up to industrial tonnage without encountering the mixing or heat transfer issues often seen with novel flow chemistry or biocatalytic processes. From an environmental standpoint, the recovery of elemental selenium from the oxidation step and the potential for solvent recycling align with green chemistry metrics. The avoidance of heavy metal residues in the final product simplifies the regulatory registration process for the end-use fungicide, as impurity profiles are cleaner and easier to characterize. This compliance readiness accelerates time-to-market for new products derived from this technology platform.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is essential for R&D teams evaluating the feasibility of integrating these intermediates into their discovery pipelines, as well as for procurement officers assessing the risk profile of the supply chain. The answers are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance to real-world manufacturing scenarios.
Q: What is the primary starting material for this synthesis?
A: The synthesis utilizes alpha-pinene, a abundant and renewable component of turpentine, as the foundational chiral pool starting material.
Q: What is the antifungal spectrum of these compounds?
A: The synthesized diacylhydrazides exhibit broad-spectrum activity against various plant pathogens, including rice sheath blight and apple ring rot, often outperforming standard controls.
Q: Is this process suitable for large-scale production?
A: Yes, the methodology employs standard unit operations such as distillation and extraction, avoiding exotic catalysts, which facilitates straightforward commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Myrtenal Dihydrazide Supplier
As a leader in the custom synthesis and manufacturing of complex organic intermediates, NINGBO INNO PHARMCHEM is uniquely positioned to bring this innovative technology to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We understand the critical importance of stringent purity specifications in the agrochemical sector, and our rigorous QC labs are equipped to verify the identity and purity of every batch against the highest international standards. By leveraging our expertise in terpene chemistry and acylation reactions, we can guarantee a consistent supply of high-quality myrtenal dihydrazide derivatives that meet your specific formulation requirements.
We invite you to collaborate with us to explore the full potential of this patent-protected chemistry for your crop protection portfolio. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating how our optimized process can reduce your overall procurement costs. Please contact our technical procurement team today to request specific COA data for our available analogues and to discuss route feasibility assessments for your target molecules. Together, we can accelerate the development of next-generation fungicides that deliver superior performance and sustainability.
