Advanced Green Synthesis of Bicyclic Pyridone Intermediates for Pharmaceutical Applications
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable pathways to construct complex heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. Patent CN114507230A introduces a groundbreaking methodology for the synthesis of bicyclic pyridone derivatives, a structural motif prevalent in critical medications ranging from ACE inhibitors to anticancer agents like Camptothecin. This innovation addresses the longstanding challenges associated with traditional heterocycle synthesis, which often rely on hazardous reagents and energy-intensive processes. By leveraging citric acid—a ubiquitous, non-toxic organic molecule—as a key building block, this technology offers a paradigm shift towards greener chemistry without compromising on yield or purity. For R&D directors and procurement specialists, this represents a significant opportunity to streamline the supply chain for high-value pharmaceutical intermediates while adhering to increasingly stringent environmental regulations.
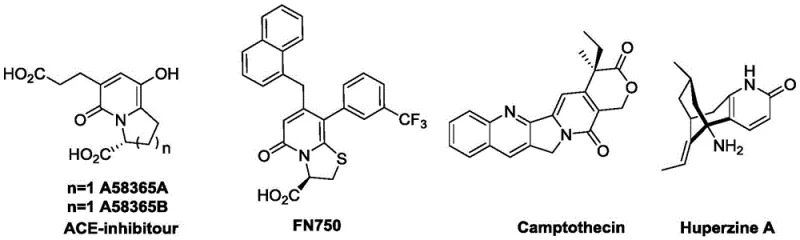
Bicyclic pyridone structures are foundational to a wide array of bioactive molecules, yet their construction has historically been fraught with chemical inefficiencies. Conventional methods typically depend on transition metal catalysts, strong acids or bases, and volatile organic solvents that pose significant safety risks and generate substantial toxic waste. These traditional routes often require harsh reaction conditions that can degrade sensitive functional groups, leading to complex impurity profiles that are difficult and costly to remove during downstream processing. Furthermore, the reliance on specialized metal catalysts introduces supply chain vulnerabilities and increases the overall cost of goods sold (COGS). The limitations of these legacy technologies have restricted the broader application of bicyclic pyridone compounds, particularly in contexts where cost-effectiveness and environmental compliance are paramount.
In stark contrast, the novel approach detailed in the patent utilizes a hydrothermal cyclization strategy driven by citric acid and simple amines. This method operates under solvent-free or aqueous conditions at moderate temperatures, typically around 140°C, effectively eliminating the need for flammable or toxic organic solvents. The reaction mechanism capitalizes on the multifunctional nature of citric acid, which acts simultaneously as a carbon source and a reaction medium, facilitating the formation of the fused ring system with high atom economy. This green chemistry protocol not only simplifies the operational workflow by removing solvent recovery steps but also drastically reduces the environmental footprint of the manufacturing process. The result is a cleaner reaction profile that yields high-purity intermediates suitable for direct use in subsequent synthetic transformations.
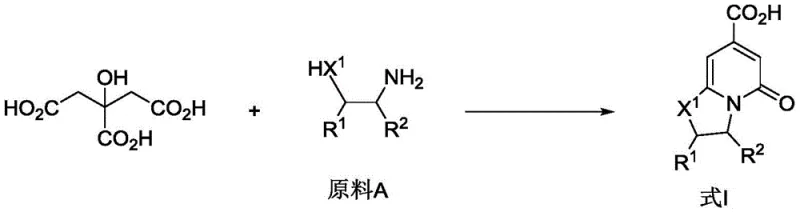
The mechanistic insights into this citric acid-mediated cyclization reveal a sophisticated interplay of thermal activation and molecular rearrangement. Under hydrothermal conditions, the carboxylic acid groups of citric acid interact with the amine functionalities of the substrate to initiate a condensation sequence. This is followed by an intramolecular cyclization that constructs the pyridone core, driven by the thermodynamic stability of the resulting aromatic system. The absence of external catalysts suggests that the reaction proceeds through a concerted mechanism where the reactants themselves provide the necessary acidic or basic environment to promote bond formation. This intrinsic catalytic behavior minimizes the introduction of extraneous impurities, such as metal residues, which are a major concern in pharmaceutical manufacturing. Consequently, the purification burden is significantly lowered, allowing for more efficient isolation of the target bicyclic scaffold.
Impurity control is a critical aspect of this synthesis, particularly given the potential for side reactions at elevated temperatures. However, the mild nature of the hydrothermal conditions (60-180°C) compared to traditional high-energy methods ensures that thermal degradation is minimized. The high chemical selectivity observed in this process means that competing reaction pathways are suppressed, leading to a dominant formation of the desired bicyclic product. For quality control teams, this translates to a more consistent product batch-to-batch, reducing the need for extensive chromatographic purification. The ability to tune the reaction by simply adjusting the molar ratio of citric acid to amine (optimally 1:1) provides an additional layer of process control, ensuring that the synthesis remains robust even when scaling up from laboratory to pilot plant volumes.
How to Synthesize Bicyclic Pyridone Derivatives Efficiently
The synthesis of these valuable intermediates is remarkably straightforward, requiring minimal equipment and standard laboratory safety protocols. The process begins with the precise weighing of citric acid and the chosen amine substrate, such as ethylenediamine or cysteine, which are then mixed thoroughly to ensure homogeneity. This mixture is transferred into a hydrothermal synthesis kettle, a standard piece of equipment in modern chemical processing, and heated to the optimal reaction temperature of 140°C. The reaction is allowed to proceed for a duration ranging from 0.5 to 12 hours, with 4 hours often being sufficient for complete conversion. Upon completion, the crude product, typically a solid, is isolated and subjected to recrystallization to achieve the high purity levels required for pharmaceutical applications. Detailed standardized synthesis steps are provided in the guide below.
- Mix citric acid and the selected amine原料 (such as ethylenediamine or cysteine) in a molar ratio of approximately 1: 1.
- Load the mixture into a hydrothermal synthesis kettle and heat to a temperature range of 60-180°C, optimally around 140°C.
- Maintain the solvent-free reaction for 0.5 to 12 hours, then purify the resulting solid product via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this citric acid-based synthesis route offers compelling economic and logistical benefits. The primary driver of cost reduction lies in the substitution of expensive, specialized reagents with commodity chemicals. Citric acid is produced on a massive global scale for the food and beverage industry, ensuring a stable and low-cost supply that is immune to the volatility often seen in the fine chemical sector. By eliminating the need for precious metal catalysts and toxic solvents, manufacturers can avoid the significant costs associated with catalyst recovery, solvent disposal, and environmental compliance reporting. This streamlined input profile directly translates to a lower cost of production, enhancing the margin potential for downstream API manufacturing.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and organic solvents removes entire unit operations from the production line, such as solvent recovery distillation and heavy metal scavenging. This simplification reduces both capital expenditure on equipment and operational expenditure on energy and waste management. Furthermore, the use of citric acid, a bulk chemical, stabilizes raw material costs against market fluctuations typical of niche synthetic reagents. The overall process efficiency is heightened by the solvent-free nature of the reaction, which concentrates the reactants and accelerates kinetics without the dilution effect of a solvent medium.
- Enhanced Supply Chain Reliability: Sourcing reliability is significantly improved because the key starting materials are globally available commodities rather than proprietary intermediates with single-source suppliers. Citric acid and simple diamines are produced by multiple manufacturers worldwide, mitigating the risk of supply disruptions due to geopolitical issues or production outages at a single facility. This diversification of the supply base ensures business continuity for pharmaceutical companies relying on these intermediates. Additionally, the simplified logistics of handling non-hazardous, solid starting materials reduce transportation costs and regulatory burdens associated with shipping dangerous goods.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on hydrothermal technology, which is well-established in the chemical industry for large-volume production. The absence of volatile organic compounds (VOCs) simplifies the permitting process for new manufacturing facilities and reduces the risk of workplace exposure incidents. From an environmental standpoint, the reduction in hazardous waste generation aligns with corporate sustainability goals and regulatory mandates like REACH. The energy efficiency of operating at moderate temperatures further contributes to a lower carbon footprint, making this technology attractive for companies aiming to achieve green chemistry certifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of using citric acid in this synthesis?
A: Citric acid serves as a low-toxicity, inexpensive, and readily available starting material that eliminates the need for hazardous transition metal catalysts and toxic organic solvents, significantly reducing environmental impact and production costs.
Q: Can this method be scaled for industrial production of API intermediates?
A: Yes, the process utilizes mild reaction conditions (approx. 140°C) and solvent-free hydrothermal techniques, which simplify reactor requirements and safety protocols, making it highly suitable for large-scale commercial manufacturing.
Q: What biological activities do these bicyclic pyridone derivatives exhibit?
A: The synthesized compounds demonstrate significant antibacterial activity against strains like Staphylococcus aureus and E. coli, alongside fluorescent properties, making them valuable for both therapeutic and diagnostic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bicyclic Pyridone Derivatives Supplier
As the demand for sustainable and cost-effective pharmaceutical intermediates grows, partnering with an experienced CDMO becomes essential for translating innovative patent technologies into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale hydrothermal synthesis to industrial manufacturing is seamless. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of bicyclic pyridone derivatives meets the exacting standards required for drug substance development. We understand the critical nature of supply chain continuity and are equipped to handle the complexities of green chemistry processes.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this green route can optimize your budget. Please contact us to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply of high-quality intermediates for your next generation of therapeutics.
