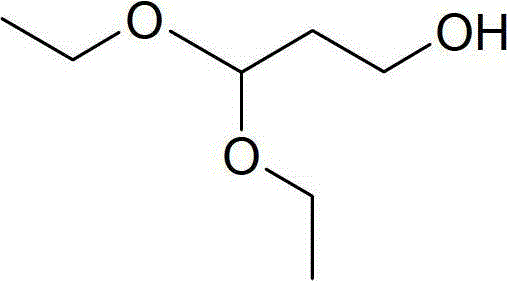
Advanced Manufacturing of 3,3-Diethoxy-1-Propanol: A Low-Pressure Route for Pharma Intermediates
Introduction to Advanced Synthetic Methodology
The pharmaceutical industry constantly seeks robust, scalable, and safe pathways for producing critical intermediates, and patent CN103724166A presents a significant breakthrough in the manufacturing of 3,3-diethoxy-1-propanol. This specific compound, characterized by its acetal functionality and terminal hydroxyl group, serves as a versatile building block in complex organic synthesis and medicinal chemistry. Traditionally, the production of such acetals has been plagued by harsh reaction conditions that pose significant engineering challenges. However, this innovative methodology shifts the paradigm by utilizing malonaldehyde and absolute ethanol as primary feedstocks, proceeding through a sophisticated four-step sequence involving aldolization, nucleophilic substitution, basic hydrolysis, and chemical reduction. The strategic design of this route eliminates the need for high-pressure reactors, offering a safer and more controllable alternative for industrial applications.
From a technical perspective, the transition from high-energy hydrolysis to this mild, multi-step catalytic process represents a major leap forward in process chemistry. The patent details a system where reaction temperatures are maintained between 60°C and 90°C for the initial steps, dropping even lower for the reduction phase, which stands in stark contrast to legacy methods requiring upwards of 300°C. This drastic reduction in thermal energy demand not only lowers the carbon footprint of the manufacturing process but also minimizes the formation of thermal degradation by-products, thereby ensuring a final product purity that consistently exceeds 98%. For R&D directors and process engineers, this level of control over the reaction environment is crucial for maintaining batch-to-batch consistency in GMP environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 3,3-diethoxy-1-propanol has relied heavily on the alkaline hydrolysis of 3-chloropropionaldehyde diethyl acetal. While chemically straightforward, this legacy approach imposes severe constraints on manufacturing infrastructure and operational safety. The prior art necessitates reaction temperatures ranging from 150°C to 300°C, coupled with system pressures between 0.25 MPa and 0.92 MPa. Such extreme conditions require specialized high-pressure autoclaves and rigorous safety protocols, significantly inflating capital expenditure (CAPEX) for production facilities. Furthermore, the harsh thermal environment often leads to side reactions, resulting in lower yields and a complex impurity profile that demands extensive downstream purification. The combination of high energy consumption, expensive equipment requirements, and suboptimal product quality makes this traditional route increasingly unviable for modern, cost-sensitive pharmaceutical supply chains.
The Novel Approach
In direct response to these inefficiencies, the novel synthetic route described in the patent introduces a温和 (mild) and highly efficient alternative that leverages readily available raw materials. By starting with malonaldehyde and employing a solid acid catalyst for the initial acetalization, the process avoids the corrosive and hazardous conditions associated with mineral acids and high heat. The subsequent steps utilize common reagents like acetyl chloride and sodium bicarbonate under ambient or near-ambient pressures, drastically simplifying the reactor requirements. A key innovation lies in the telescoping of the hydrolysis and reduction steps; rather than isolating the unstable aldehyde intermediate, the process reduces the crude hydrolysis mixture directly. This not only streamlines the workflow by removing a unit operation but also prevents the polymerization issues typically associated with free aldehydes, leading to superior overall yields and a much cleaner final product suitable for high-value applications.
Mechanistic Insights into Solid Acid Catalysis and Reduction
The core of this synthetic strategy relies on the precise manipulation of functional groups through a sequence of well-defined mechanistic steps. The initial aldolization and acetalization are driven by a solid acid catalyst, such as hydrated ferric sulfate or aluminum sulfate, which promotes the formation of 1,1,3,3-tetraethoxypropane from malonaldehyde and ethanol. The use of a heterogeneous or soluble solid acid allows for effective protonation of the carbonyl oxygen without the severe corrosion issues linked to liquid mineral acids. Following this, nucleophilic substitution with acetyl chloride introduces the chlorine atom at the C1 position, activating the molecule for the subsequent hydrolysis. The choice of acetyl chloride is critical here, as it acts as both a reagent and a dehydrating agent, driving the equilibrium towards the chloro-intermediate with high efficiency.
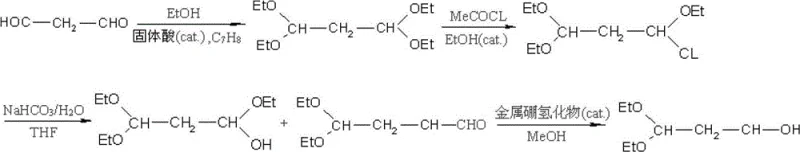
The final stages of the synthesis demonstrate exceptional chemical ingenuity regarding impurity control and process simplification. During the basic hydrolysis step using sodium bicarbonate in a THF-water system, the chloro-group is displaced to form a hydroxyl group, generating a mixture of 1-hydroxy-1,3,3-tetraethoxypropane and 3,3-diethoxy-1-propionaldehyde. Instead of attempting a difficult separation of these components, the process employs a metal borohydride (such as sodium borohydride or potassium borohydride) to reduce the entire mixture. This "one-pot" reduction strategy effectively converts both the hemiacetal and the free aldehyde into the desired 3,3-diethoxy-1-propanol. By bypassing the isolation of the aldehyde, the method circumvents the risk of aldol condensation or polymerization, which are common pitfalls in aldehyde chemistry. This mechanistic shortcut is the primary driver behind the reported high yields and the simplified workup procedure, making it highly attractive for commercial scale-up.
How to Synthesize 3,3-Diethoxy-1-Propanol Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control, particularly during the exothermic addition of acetyl chloride and the reduction phase. The patent outlines a robust protocol where solvents like toluene and THF are utilized not just for solubility but also for their ability to facilitate phase separation and recycling. The initial acetalization is performed under reflux with a Dean-Stark trap to remove water, driving the equilibrium towards the tetraethoxypropane product. Subsequent steps are conducted at controlled low temperatures (0°C to 30°C) to manage reaction rates and selectivity. For detailed operational parameters, including specific mass ratios of catalysts and solvents, operators should refer to the standardized guidelines derived from the patent examples.
- Perform aldolization of malonaldehyde with absolute ethanol using a solid acid catalyst in toluene to form 1,1,3,3-tetraethoxypropane.
- React the tetraethoxypropane intermediate with fresh acetyl chloride in ethanol to achieve nucleophilic substitution, yielding 1-chloro-1,3,3-tetraethoxypropane.
- Conduct basic hydrolysis using sodium bicarbonate in a THF-water system to convert the chloro-intermediate into a hydroxy-aldehyde mixture.
- Execute chemical reduction of the mixture using metal borohydride (such as sodium borohydride) in methanol to obtain the final high-purity alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The shift away from high-pressure, high-temperature processing fundamentally alters the cost structure of manufacturing 3,3-diethoxy-1-propanol. By eliminating the need for specialized high-pressure vessels and the intense energy input required to maintain 300°C reactions, the operational expenditure (OPEX) is significantly reduced. Furthermore, the ability to recycle key solvents like ethanol and THF creates a closed-loop system that minimizes raw material waste and disposal costs. This aligns perfectly with modern green chemistry initiatives and helps manufacturers meet increasingly stringent environmental regulations without sacrificing productivity.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven by the use of commodity chemicals such as malonaldehyde, ethanol, and sodium bicarbonate, which are abundant and inexpensive compared to specialized precursors. Additionally, the elimination of complex purification steps, such as the isolation of the unstable aldehyde intermediate, reduces labor hours and solvent consumption. The solid acid catalysts employed are also cost-effective and can often be recovered or replaced with cheap alternatives like iron salts, avoiding the need for expensive noble metal catalysts. These factors combine to create a leaner, more cost-efficient production model that enhances margin potential for the final API or intermediate.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the mild reaction conditions translate to higher equipment availability and reduced downtime. Traditional high-pressure reactions often require frequent maintenance and safety inspections, whereas this atmospheric or low-pressure process can run continuously with greater stability. The reliance on widely available raw materials also mitigates the risk of supply disruptions; unlike niche reagents that may have single-source suppliers, ethanol and basic inorganic salts are globally sourced commodities. This ensures a steady flow of production and reliable delivery schedules for downstream pharmaceutical customers who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this methodology is inherently designed for expansion. The absence of high-pressure hazards simplifies the engineering controls needed for larger reactors, allowing for a smoother transition from pilot plant to commercial tonnage. Moreover, the aqueous workup and solvent recovery systems minimize the generation of hazardous waste streams. The process avoids the use of heavy metals or toxic reagents that would require complex effluent treatment, making it easier for manufacturing sites to maintain compliance with local environmental protection laws and international sustainability standards.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common concerns regarding safety, purity, and scalability, drawing directly from the experimental data and technical specifications provided in the patent documentation. These insights are intended to clarify how this method outperforms legacy technologies in real-world manufacturing scenarios.
Q: What are the primary safety advantages of this new synthesis method compared to traditional hydrolysis?
A: Unlike conventional methods that require high temperatures (150°C-300°C) and elevated pressures (0.25MPa-0.92MPa), this novel route operates under mild conditions (20°C-90°C) and atmospheric or low pressure, significantly reducing equipment stress and operational hazards.
Q: How does the process handle impurity control during the reduction step?
A: The process utilizes a strategic shortcut where the hydrolysis mixture containing both 1-hydroxy-1,3,3-tetraethoxypropane and 3,3-diethoxy-1-propionaldehyde is directly reduced. This avoids the isolation of the unstable aldehyde, preventing polymerization by-products and simplifying purification.
Q: Is the solvent system designed for industrial scalability?
A: Yes, the protocol explicitly designs for solvent recovery. Ethanol and THF, which are used in large quantities, can be distilled and recycled after the reaction, enhancing the economic viability and environmental compliance of large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3-Diethoxy-1-Propanol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN103724166A are fully realized in practice. Our facilities are equipped with state-of-the-art reactors capable of handling mild catalytic processes with precision, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of 3,3-diethoxy-1-propanol meets the highest pharmaceutical standards. We are committed to delivering high-purity pharmaceutical intermediates that empower your drug development pipelines.
We invite you to leverage our technical proficiency to optimize your supply chain for this critical intermediate. Whether you are looking to validate this new low-pressure route or require a Customized Cost-Saving Analysis for your current procurement strategy, our team is ready to assist. Please contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project's unique volume and quality requirements.
