Advanced Engineered Penicillin G Acylase Variants For High Purity Insulin And Pharmaceutical Intermediates Production
The pharmaceutical and fine chemical industries are constantly seeking robust biocatalytic solutions to enhance the efficiency of complex molecule synthesis. Patent CN111051506A introduces a significant breakthrough in this domain by disclosing engineered Penicillin G Acylase (PGA) variants designed for superior performance in challenging deprotection reactions. Unlike traditional wild-type enzymes that often suffer from instability and require complex activation steps, these engineered polypeptides offer a streamlined pathway for producing high-purity pharmaceutical intermediates. Specifically, the patent details variants capable of efficiently removing phenyl acetate protecting groups from insulin dimers, a critical step in the manufacturing of modern biologic therapeutics. This technological advancement addresses long-standing pain points in enzymatic processing, offering a compelling value proposition for R&D directors focused on yield optimization and procurement managers seeking reliable supply chains for critical reagents.
Furthermore, the scope of this invention extends beyond mere sequence modifications; it encompasses the polynucleotides encoding these enzymes, the host cells for their expression, and the methods for their utilization in industrial settings. The engineered PGAs exhibit remarkable identity levels to specific reference sequences, such as SEQ ID NO: 6 and SEQ ID NO: 88, while incorporating strategic amino acid substitutions that enhance thermostability and solvent tolerance. For stakeholders in the pharmaceutical intermediates sector, this represents a shift towards more resilient biocatalysts that can withstand the rigorous conditions of large-scale fermentation and downstream processing. The ability to deploy these enzymes in processes requiring high concentrations of organic co-solvents, such as DMSO, without significant loss of activity is a testament to the robustness of the protein engineering strategies employed herein.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial application of Penicillin G Acylase has been constrained by the inherent biological limitations of the wild-type enzyme derived from organisms like Kluyvera citrophila. In its natural state, PGA is synthesized as an inactive precursor known as pre-pro-PGA, which contains an N-terminal signal peptide and a spacer region linking the alpha and beta subunits. For the enzyme to become catalytically active, it must undergo a series of self-processing steps to remove these intervening sequences. This requirement introduces significant variability and inefficiency into the manufacturing process, as the rate of auto-processing can be inconsistent and dependent on specific environmental conditions. Moreover, wild-type PGA often exhibits marginal stability when exposed to elevated temperatures or organic solvents, which are frequently necessary to solubilize hydrophobic substrates like protected insulin dimers. These factors collectively contribute to lower overall yields, increased batch-to-batch variability, and higher operational costs due to the need for precise control and extended reaction times.
The Novel Approach
The novel approach presented in this patent circumvents these biological bottlenecks through rational design and directed evolution. The engineered PGA variants are constructed to lack the spacer region entirely, meaning they are expressed directly as active heterodimers without the need for post-translational self-cleavage. This structural modification ensures that the enzyme is immediately functional upon expression, significantly simplifying the production workflow and reducing the risk of incomplete activation. Additionally, the introduction of specific amino acid substitutions at key positions within the protein structure enhances the enzyme's resilience against denaturation. The patent highlights variants that maintain high levels of residual activity even after exposure to stressful conditions, thereby enabling more flexible process parameters. This robustness allows manufacturers to operate at higher substrate loadings and potentially reduce the amount of enzyme required per batch, leading to substantial process intensification and cost efficiencies.
Mechanistic Insights into Engineered PGA Catalysis
The catalytic mechanism of Penicillin G Acylase involves the nucleophilic attack on the amide bond of the substrate, facilitated by the N-terminal serine residue of the beta subunit. In the context of the engineered variants described in the patent, this mechanism is preserved but optimized for specific steric and electronic environments presented by bulky protecting groups like phenyl acetate on insulin. The enzyme recognizes the hydrophobic phenyl moiety of the substrate, anchoring it within the active site pocket before executing the hydrolytic cleavage. This specificity is crucial for preventing off-target reactions that could compromise the integrity of the sensitive peptide backbone of insulin. The engineered variants achieve this through subtle alterations in the amino acid residues lining the binding pocket, which improve the affinity for the protected insulin dimer while excluding water molecules that might promote non-specific hydrolysis. Such precision is essential for maintaining the stereochemical purity of the final product, a critical quality attribute for regulatory approval.
Moreover, the stability of the enzyme-substrate complex is enhanced by the engineered mutations, which likely reinforce the tertiary structure of the protein against thermal agitation. The patent data suggests that these variants can operate effectively in the presence of significant concentrations of co-solvents, which is often a challenge for native enzymes. This solvent tolerance expands the range of substrates that can be processed, as many pharmaceutical intermediates have limited solubility in purely aqueous buffers. By enabling reactions in mixed solvent systems, the engineered PGA facilitates higher substrate concentrations, which directly translates to improved volumetric productivity. The mechanistic robustness of these variants ensures that the catalytic turnover rate remains high throughout the reaction duration, minimizing the formation of incomplete deprotection byproducts and simplifying the downstream purification requirements.
How to Synthesize Free Insulin Dimer Efficiently
The implementation of this technology involves a straightforward biocatalytic workflow that integrates seamlessly into existing fermentation infrastructure. The process begins with the transformation of a suitable host cell, such as E. coli, with a vector containing the codon-optimized polynucleotide sequence encoding the desired PGA variant. Following fermentation and cell lysis, the crude enzyme preparation can be directly utilized or further purified depending on the specific purity requirements of the application. The reaction is typically conducted in a buffered aqueous solution containing a controlled amount of organic co-solvent to ensure substrate solubility. Detailed standard operating procedures for the expression, purification, and application of these variants are critical for maximizing their potential in a commercial setting.
- Preparation of the engineered PGA variant expression system using host cells such as E. coli with codon-optimized polynucleotides.
- Culturing the transformed host cells under controlled fermentation conditions to express the variant PGA enzyme.
- Contacting the protected insulin dimer substrate with the engineered PGA variant under suitable reaction conditions to effect deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of these engineered PGA variants offers tangible benefits related to cost stability and operational reliability. The elimination of the self-processing step required by wild-type enzymes removes a source of process variability, leading to more predictable batch cycles and consistent product quality. This reliability is paramount for maintaining continuous supply lines in the highly regulated pharmaceutical industry. Furthermore, the enhanced stability of the engineered enzymes reduces the sensitivity of the process to minor fluctuations in temperature or pH, lowering the risk of batch failures and the associated financial losses. The ability to achieve high conversion rates with reduced enzyme loading also contributes to direct cost savings on biocatalyst consumption, which can be a significant portion of the variable costs in enzymatic manufacturing processes.
- Cost Reduction in Manufacturing: The structural optimization of the PGA variants eliminates the need for complex activation protocols, thereby reducing processing time and utility consumption. By removing the spacer region, the enzyme is produced in its active form, which streamlines the upstream production process and minimizes the loss of activity associated with maturation steps. This efficiency gain allows for a reduction in the overall enzyme dosage required to achieve complete substrate conversion, directly lowering the cost of goods sold. Additionally, the improved solvent stability enables the use of higher substrate concentrations, which increases the throughput of existing reactor volumes without the need for capital-intensive equipment upgrades.
- Enhanced Supply Chain Reliability: The robustness of the engineered enzymes ensures consistent performance across different production batches, mitigating the risk of supply disruptions caused by enzymatic underperformance. The ability of these variants to tolerate a wider range of reaction conditions provides a buffer against raw material variability, ensuring that the manufacturing process remains stable even if minor deviations occur in buffer preparation or substrate quality. This resilience supports a more agile supply chain capable of responding to fluctuating demand without compromising on product specifications. Long-term stability of the enzyme preparations also facilitates easier logistics and storage, reducing the complexity of inventory management.
- Scalability and Environmental Compliance: The high efficiency of the engineered PGA supports scalable manufacturing processes that align with green chemistry principles. By achieving high yields with minimal byproduct formation, the process reduces the burden on waste treatment facilities and lowers the environmental footprint of the manufacturing operation. The specificity of the enzyme minimizes the generation of hazardous waste streams associated with chemical deprotection methods, offering a more sustainable alternative for industrial applications. Furthermore, the compatibility of these variants with standard fermentation technologies ensures that scale-up from laboratory to commercial production can be achieved with minimal technical risk and investment.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of the engineered Penicillin G Acylase variants described in the patent. These insights are derived from the experimental data and embodiments disclosed, providing a clear understanding of the technology's capabilities and limitations. Understanding these aspects is crucial for technical teams evaluating the feasibility of integrating this biocatalytic solution into their existing workflows.
Q: What is the primary advantage of the engineered PGA variants over wild-type enzymes?
A: The engineered variants eliminate the need for self-processing steps by removing the spacer region found in wild-type precursors, leading to immediate enzymatic activity and improved stability.
Q: Can these enzymes be used for substrates other than insulin?
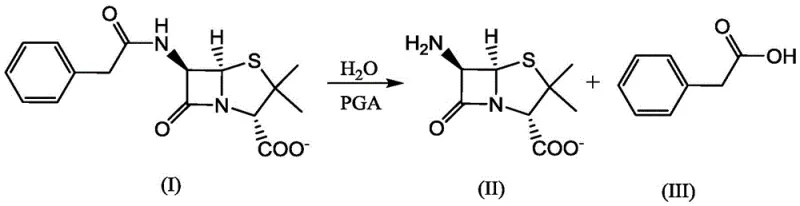
A: Yes, while optimized for insulin deprotection, the variants retain the ability to hydrolyze penicillin G into 6-APA and phenylacetic acid, serving dual purposes in antibiotic and biologics manufacturing.
Q: How does this technology impact the purity of the final insulin product?
A: The high chemoselectivity of the engineered PGA ensures exclusive cleavage of the phenyl acetate amide bond without affecting remaining peptide bonds, resulting in high-purity free insulin dimers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Engineered Penicillin G Acylase Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalysis in modern pharmaceutical manufacturing. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like these engineered PGA variants can be successfully translated from the lab to the plant. Our facility is equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of enzyme or intermediate meets the highest international standards. We understand the critical nature of supply continuity for key starting materials and are committed to providing a stable, high-quality supply of biocatalytic solutions tailored to your specific process needs.
We invite you to collaborate with our technical team to explore how these engineered enzymes can optimize your current manufacturing processes. Contact our technical procurement team today to request a Customized Cost-Saving Analysis specific to your production volume. We are ready to provide specific COA data and route feasibility assessments to demonstrate the tangible value our solutions can bring to your supply chain. Let us help you leverage cutting-edge enzyme technology to achieve greater efficiency and competitiveness in the global market.
