Advanced Synthesis and Commercial Scalability of Novel Spirobenzoxazine Piperidine Antifungal Intermediates
Introduction to Novel Antifungal Scaffold Technology
The escalating crisis of invasive fungal infections, characterized by high morbidity and mortality rates, necessitates the urgent development of therapeutic agents with novel mechanisms of action. Patent CN113264949B discloses a groundbreaking class of 4-(6-methoxy-2-oxo-1,2-dihydrospiro[benzo[d][1,3]oxazine-4,4'-piperidin]-1'-yl)-4-oxo-N-phenylbut-2-enamine derivatives. These molecules represent a significant leap forward in medicinal chemistry, specifically designed to target chitin synthase, an enzyme essential for fungal cell wall biosynthesis but absent in mammalian cells. This selectivity profile minimizes host toxicity while maximizing antifungal potency. The core structural motif integrates a rigid spirobenzoxazine piperidine scaffold with an alpha,beta-unsaturated ketone linker, creating a unique pharmacophore capable of penetrating fungal membranes effectively.
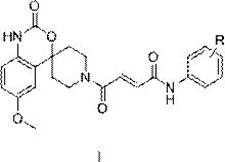
From a commercial manufacturing perspective, the accessibility of this chemical space is paramount. The general formula I encompasses a diverse library of substituents (R groups including -CH3, -CF3, -Cl, -OCH3), allowing for extensive Structure-Activity Relationship (SAR) optimization. For procurement managers and supply chain directors, understanding the synthetic feasibility of these pharmaceutical intermediates is critical. The disclosed technology offers a robust pathway that avoids exotic reagents, relying instead on established industrial building blocks like substituted anilines and maleic anhydride. This strategic design ensures that scaling from laboratory grams to metric tons is not only feasible but economically viable, positioning these compounds as reliable candidates for next-generation antifungal drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing complex amide-linked heterocycles often rely on aggressive activation strategies that introduce significant process hazards and purification bottlenecks. Classical methods frequently employ thionyl chloride or oxalyl chloride to convert carboxylic acids into acid chlorides prior to amidation. These reagents are corrosive, generate hazardous gaseous byproducts such as HCl and SO2, and require stringent moisture control, thereby increasing capital expenditure for specialized reactor lining and scrubbing systems. Furthermore, acid chloride intermediates are prone to hydrolysis and polymerization, leading to lower overall yields and complex impurity profiles that demand resource-intensive chromatographic purification. In the context of cost reduction in pharmaceutical intermediate manufacturing, these inefficiencies translate directly into higher production costs and extended lead times, making conventional routes less attractive for large-scale commercialization.
The Novel Approach
The methodology outlined in the patent data introduces a superior synthetic strategy utilizing 1-propylphosphonic anhydride (T3P) as a coupling mediator. This modern approach circumvents the formation of unstable acid chlorides, enabling direct activation of the cinnamic acid intermediates under mild conditions. The reaction proceeds efficiently in dichloromethane at temperatures ranging from ambient to 45°C, significantly reducing energy consumption compared to high-temperature reflux protocols. By eliminating the need for hazardous chlorinating agents, the process inherently improves workplace safety and environmental compliance. Moreover, the byproducts of T3P coupling are water-soluble phosphonic acids, which can be easily removed via aqueous washes, simplifying the isolation of the final spirobenzoxazine product. This streamlined workflow enhances the commercial scale-up of complex pharmaceutical intermediates by reducing unit operations and improving overall process mass intensity.
Mechanistic Insights into T3P-Mediated Amide Coupling
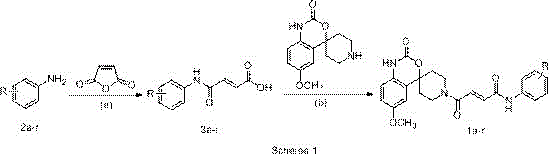
The chemical transformation at the heart of this synthesis involves the formation of a robust amide bond between a functionalized cinnamic acid derivative and the spiro-piperidine amine. Mechanistically, T3P acts as a dehydrating agent that activates the carboxylic acid group of the cinnamic acid intermediate (Compound 3a-r). Upon addition of triethylamine, a mixed anhydride species is generated in situ, which is highly electrophilic yet stable enough to prevent side reactions such as racemization or over-activation. The nucleophilic nitrogen of the 6-methoxy-spiro[benzo[d][1,3]oxazine-4,4'-piperidin]-2(1H)-one then attacks this activated carbonyl carbon. The presence of the alpha,beta-unsaturated system requires careful control to prevent Michael addition side reactions; however, the specific reaction conditions disclosed (45°C, 16 hours) favor the thermodynamic amide product. This precise control over reaction kinetics is essential for maintaining high-purity pharmaceutical intermediates suitable for downstream drug substance manufacturing.
Beyond the synthetic mechanism, the biological mechanism of action provides further value to R&D directors. These derivatives function as chitin synthase inhibitors. Chitin is a polysaccharide that provides structural rigidity to the fungal cell wall. By binding to the active site of chitin synthase enzymes, these spirobenzoxazine compounds block the polymerization of N-acetylglucosamine units. This inhibition leads to a weakened cell wall that cannot withstand internal osmotic pressure, resulting in cell lysis and death. Since mammals lack chitin biosynthetic pathways, this target offers a high therapeutic index. The structural rigidity imparted by the spiro-center likely enhances binding affinity by pre-organizing the molecule into the bioactive conformation required for enzyme interaction, thereby improving potency against resistant strains like Candida albicans and Aspergillus fumigatus.
How to Synthesize Spirobenzoxazine Piperidine Derivatives Efficiently
The synthesis of these valuable antifungal intermediates follows a logical two-step sequence that balances yield with operational simplicity. The process begins with the preparation of the cinnamic acid linker, followed by the key coupling event. Operators should note that maintaining anhydrous conditions during the T3P activation step is beneficial, although the protocol is more forgiving than acid chloride methods. The use of dichloromethane as a solvent ensures good solubility for both the polar spiro-ketone and the organic acid intermediates. Following the reaction, standard aqueous workup procedures effectively remove urea and phosphonic acid byproducts. For detailed operational parameters, stoichiometry, and specific purification techniques required to achieve GMP-grade quality, please refer to the standardized synthesis guide below.
- Condense substituted anilines with maleic anhydride in dichloromethane at room temperature to form cinnamic acid intermediates.
- Activate the carboxylic acid intermediate using 1-propylphosphonic anhydride (T3P) and triethylamine in dichloromethane.
- Couple the activated species with 6-methoxy-spiro[benzo[d][1,3]oxazine-4,4'-piperidin]-2(1H)-one at 45°C to yield the final derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers evaluating new supply chains, the economic and logistical attributes of a synthetic route are just as critical as the chemical yield. The disclosed technology offers substantial advantages in terms of raw material security and process robustness. The starting materials, including various substituted anilines and maleic anhydride, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. This diversification of the supply base ensures continuity of supply even during market fluctuations. Furthermore, the avoidance of specialized catalysts or cryogenic conditions means that the synthesis can be performed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants, eliminating the need for costly capital investment in new infrastructure.
- Cost Reduction in Manufacturing: The implementation of T3P-mediated coupling significantly lowers the cost of goods sold (COGS) by streamlining the purification process. Unlike traditional methods that may require column chromatography to remove toxic metal residues or chlorinated byproducts, this method allows for purification via crystallization or simple washing. The reduction in solvent usage and waste disposal costs contributes to a leaner manufacturing budget. Additionally, the mild reaction temperatures reduce energy overheads associated with heating and cooling cycles, further enhancing the economic viability of producing these pharmaceutical intermediates on a multi-ton scale.
- Enhanced Supply Chain Reliability: The synthetic route is designed with scalability in mind, ensuring that reducing lead time for high-purity intermediates is achievable. The reaction times are reasonable (approximately 16 hours for the coupling step), allowing for efficient batch turnover. The robustness of the chemistry means that batch-to-batch variability is minimized, providing procurement teams with predictable delivery schedules. The stability of the intermediates also allows for potential telescoping of steps or storage of key precursors, adding flexibility to inventory management strategies and buffering against upstream supply disruptions.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns with green chemistry principles. The elimination of thionyl chloride and the generation of water-soluble byproducts reduce the burden on wastewater treatment facilities. The process generates less hazardous waste, simplifying regulatory compliance and lowering disposal fees. This environmental profile is increasingly important for multinational corporations aiming to meet sustainability goals. The ability to scale this process from 100 kgs to 100 MT annually without encountering exothermic runaways or safety incidents makes it an ideal candidate for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common inquiries regarding the synthesis and application of these compounds. These insights are derived directly from the experimental data and technical specifications provided in the patent literature. Understanding these nuances helps in assessing the feasibility of integrating these intermediates into your existing drug discovery or process development workflows. The following section addresses key concerns regarding biological activity, synthetic scalability, and regulatory considerations.
Q: What is the primary biological target of these spirobenzoxazine derivatives?
A: These compounds function as potent chitin synthase inhibitors. By blocking chitin synthesis, they disrupt fungal cell wall integrity, leading to osmotic instability and growth arrest, offering a distinct mechanism from azole-based antifungals.
Q: Why is T3P preferred for the amide coupling step in this synthesis?
A: 1-Propylphosphonic anhydride (T3P) is utilized because it facilitates high-yield amide bond formation under mild conditions (45°C) without requiring harsh acid chlorides. This results in simpler workup procedures and reduced impurity profiles compared to traditional coupling methods.
Q: Do these intermediates show activity against drug-resistant strains?
A: Yes, specific derivatives such as compound 1q demonstrate significant inhibitory activity against Candida albicans, Aspergillus fumigatus, and Cryptococcus neoformans, suggesting potential efficacy against strains where conventional therapies face resistance challenges.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spirobenzoxazine Piperidine Supplier
The development of novel antifungal agents requires a partner who understands both the intricacies of complex organic synthesis and the demands of global supply chains. NINGBO INNO PHARMCHEM stands ready to support your projects with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications required for clinical trial materials. We recognize that consistency is key in pharmaceutical manufacturing, and our dedicated process chemistry team is prepared to optimize the T3P coupling protocol to maximize yield and minimize impurities according to your specific requirements.
We invite you to initiate a dialogue with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need specific COA data and route feasibility assessments for the spirobenzoxazine series, we are here to provide the data-driven insights necessary for your success. Contact us today to secure a reliable supply of these high-value antifungal intermediates and accelerate your path to market.
