Optimizing Cefozopran Side Chain Acid Production via Novel Alkaline Catalysis
The pharmaceutical landscape for fourth-generation cephalosporins continues to evolve, driven by the critical need for efficient synthesis of key intermediates like cefozopran side chain acid. Patent CN102127034B introduces a transformative preparation method for (Z)-2-(5-amino-1,2,4-thiadiazole-3-yl)-2-methoxyimino acetic acid, widely known as Compound IV. This molecule serves as the essential 7-position side chain for cefozopran, a broad-spectrum antibiotic effective against Pseudomonas aeruginosa and various Gram-positive and Gram-negative bacteria. The structural integrity of this side chain is paramount for the biological activity of the final drug substance. As a reliable cefozopran side chain acid supplier, understanding the nuances of this patented route is essential for ensuring consistent quality in API manufacturing. The innovation lies not just in the final product structure, shown below, but in the optimized pathway to reach it with superior efficiency.
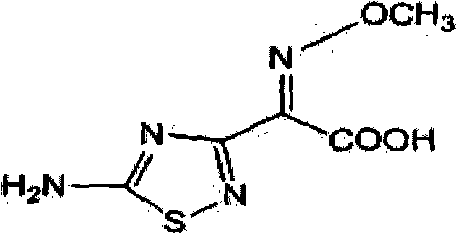
The strategic value of this intermediate cannot be overstated in the context of global antibiotic supply chains. Cefozopran represents a critical therapeutic option for severe infections, including septicemia and trauma wound infections. Consequently, the manufacturing process for its side chain must be robust, scalable, and economically viable. The patent data reveals a significant departure from traditional methods that often suffer from prolonged reaction times and suboptimal yields. By leveraging the specific catalytic conditions outlined in CN102127034B, manufacturers can achieve a level of process intensification that directly translates to cost reduction in pharmaceutical intermediates manufacturing. This report delves deep into the technical mechanisms and commercial implications of adopting this advanced synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in the patent data, the synthesis of this critical thiadiazole derivative faced substantial bottlenecks that hindered industrial scalability. Conventional routes often relied on starting materials like methoxycarbonyl carbonamidine hydrobromate, which presented significant supply chain vulnerabilities due to limited domestic availability of specific reagents like list methylthio group methyl-sulphoxide. Furthermore, historical literature indicates that uncatalyzed cyclization reactions between O-tosyl-2-carbamyl-2-methoxyimino acetyl amidoxime and potassium thiocyanate were notoriously sluggish. Without catalytic intervention, these reactions required incubation periods extending up to 48 hours to reach completion. Even after such extended durations, the isolated yields remained disappointingly low, typically hovering around 50-60%. Additionally, some prior art methods necessitated cumbersome purification steps, such as column chromatography, which are entirely impractical for multi-kilogram or ton-scale production facilities. These inefficiencies compounded to create a high-cost, low-throughput bottleneck for cefozopran production.
The Novel Approach
The breakthrough described in the patent centers on the introduction of alkaline metal catalysts to drive the ring-closure reaction, fundamentally altering the kinetics and thermodynamics of the process. By incorporating catalysts such as sodium methylate, sodium hydride, or potassium hydride into the reaction mixture, the transformation of Compound II into the cyclic intermediate (Compound III) is dramatically accelerated. The visual representation of this key cyclization step highlights the conversion of the linear amidoxime precursor into the stable thiadiazole ring system.
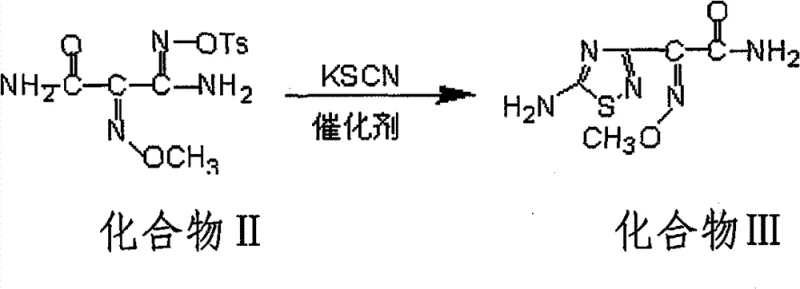
This novel approach reduces the reaction time from nearly two days to a mere 10-12 hours, representing a massive gain in reactor throughput. More importantly, the yield sees a marked improvement, consistently exceeding 70% in optimized embodiments compared to the roughly 60% baseline of non-catalytic methods. The process operates under mild conditions, typically at room temperature for the cyclization step, which minimizes energy consumption and reduces the formation of thermal degradation byproducts. This shift from a passive, slow reaction to an active, catalyzed process exemplifies how modern fine chemical engineering can revitalize established synthetic pathways to meet contemporary demands for speed and efficiency in high-purity pharmaceutical intermediate production.
Mechanistic Insights into Alkaline Metal Catalyzed Cyclization
To fully appreciate the technical superiority of this route, one must examine the mechanistic role of the alkaline catalyst in facilitating the heterocyclic ring closure. The reaction involves the nucleophilic attack of the thiocyanate ion on the electrophilic center of the amidoxime derivative. In the absence of a base, the protonation state of the reactants may not be optimal for rapid cyclization, leading to the observed kinetic lag. The addition of strong bases like sodium methylate or sodium hydride serves to deprotonate specific nitrogen centers within the substrate, thereby increasing electron density and enhancing nucleophilicity. This activation lowers the energy barrier for the intramolecular cyclization that forms the 1,2,4-thiadiazole ring. The catalyst effectively acts as a promoter, ensuring that the collision frequency between reactive species results in productive bond formation rather than reversible equilibrium states. This mechanistic understanding allows process chemists to fine-tune catalyst loading, typically between 2% and 8% by mass, to balance reaction rate against potential side reactions.
Impurity control is another critical aspect where this catalytic mechanism offers distinct advantages. In traditional long-duration reactions, the prolonged exposure of sensitive functional groups to reaction conditions often leads to hydrolysis or rearrangement byproducts that are difficult to separate. By compressing the reaction timeline to 10-12 hours, the window for secondary degradation reactions is significantly narrowed. Furthermore, the use of specific alkaline catalysts promotes a cleaner reaction profile, as evidenced by the high HPLC normalization content of over 98% reported in the patent examples. The subsequent hydrolysis step, converting the amide (Compound III) to the acid (Compound IV), is also carefully controlled. As shown in the reaction scheme below, treating the intermediate with aqueous sodium hydroxide at 50°C ensures complete conversion to the carboxylic acid without compromising the integrity of the methoxyimino group or the amino-thiadiazole moiety.
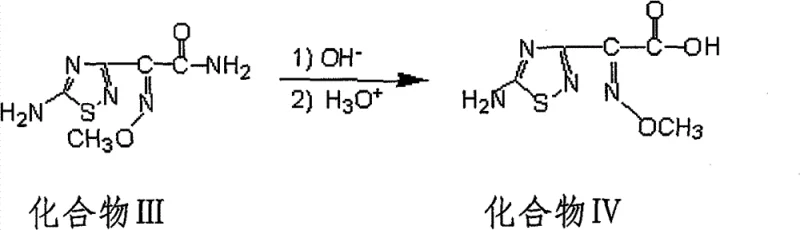
This precise control over the hydrolysis conditions prevents the formation of geometric isomers or hydrolysis of the oxime ether, which are common pitfalls in cephalosporin side chain synthesis. The final acidification step precipitates the product in high purity, ready for coupling with the cephem nucleus. Understanding these mechanistic subtleties is vital for R&D directors aiming to replicate this success in their own facilities, ensuring that the commercial scale-up of complex pharmaceutical intermediates maintains the fidelity of the laboratory results.
How to Synthesize Cefozopran Side Chain Acid Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters defined in the patent to maximize yield and purity. The process is divided into two distinct stages: the catalytic cyclization to form the thiadiazole amide, followed by hydrolysis to the free acid. The initial step involves dissolving potassium thiocyanate in methanol, followed by the addition of the tosylated precursor. The critical intervention is the timed addition of the alkaline catalyst under controlled stirring. Detailed standard operating procedures for temperature control, workup, and crystallization are essential to reproduce the >70% yields documented. For a comprehensive guide on executing this synthesis with industrial precision, please refer to the standardized protocol below.
- Dissolve potassium thiocyanate in methanol and add O-tosyl-2-carbamyl-2-methoxyimino acetamide oxime (Compound II) under stirring.
- Add an alkaline metal catalyst such as sodium methylate, sodium hydride, or potassium hydride to initiate rapid ring closure at room temperature for 10-12 hours.
- Hydrolyze the resulting intermediate (Compound III) with aqueous sodium hydroxide at 50°C, followed by acidification to isolate the final acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic methodology offers tangible benefits that extend beyond simple chemistry. The most immediate impact is the drastic reduction in cycle time. By shortening the reaction duration from 48 hours to roughly 10 hours, manufacturing facilities can significantly increase their asset utilization rates. This means that the same reactor volume can produce nearly four times the amount of product over a given month, effectively expanding capacity without capital expenditure on new equipment. This efficiency gain is a powerful driver for cost reduction in pharmaceutical intermediates manufacturing, allowing for more competitive pricing structures in a tight market. Furthermore, the elimination of column chromatography in favor of simple filtration and crystallization steps simplifies the downstream processing workflow, reducing solvent consumption and waste generation.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling when analyzed through the lens of operational efficiency. The primary cost driver in batch chemical manufacturing is often time; shorter cycles mean lower utility costs, reduced labor hours per kilogram, and decreased overhead allocation. By eliminating the need for expensive and time-consuming purification techniques like chromatography, the process relies on standard unit operations such as filtration and distillation. The use of commodity chemicals like potassium thiocyanate and common alkaline catalysts ensures that raw material costs remain stable and predictable. Additionally, the higher yield directly correlates to better atom economy, meaning less starting material is wasted, further driving down the cost of goods sold (COGS) for the final API.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability of raw materials and the robustness of the synthesis route. This method utilizes widely available reagents such as methanol, ethyl acetate, and potassium thiocyanate, which are produced at a global scale and are not subject to the same supply constraints as specialized proprietary reagents. The robustness of the reaction, indicated by its tolerance to standard workup procedures and high reproducibility across different catalysts (NaOMe, NaH, KH), reduces the risk of batch failures. Reducing lead time for high-purity pharmaceutical intermediates is critical for meeting the Just-In-Time delivery expectations of major pharmaceutical clients, ensuring that production schedules for the final cephalosporin antibiotic are not disrupted by intermediate shortages.
- Scalability and Environmental Compliance: Scaling a chemical process from the bench to the plant floor introduces challenges related to heat transfer and mixing, but this route is inherently designed for scalability. The reaction proceeds at moderate temperatures (room temperature to 50°C), avoiding the safety risks associated with highly exothermic or cryogenic processes. The simplified workup, involving precipitation and filtration, is easily adaptable to large-scale filter dryers. From an environmental perspective, the reduction in reaction time and the avoidance of silica gel for chromatography significantly lower the E-factor (mass of waste per mass of product). This aligns with modern green chemistry principles and helps manufacturers meet increasingly stringent environmental regulations regarding solvent discharge and solid waste disposal, making it a sustainable choice for long-term production.
Frequently Asked Questions (FAQ)
Navigating the technical specifics of a new manufacturing process often raises questions regarding optimization and quality control. The following inquiries address common concerns related to catalyst selection, reaction monitoring, and product specification. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a reliable foundation for process validation. Understanding these details helps bridge the gap between theoretical chemistry and practical industrial application.
Q: What is the primary advantage of using alkaline metal catalysts in this synthesis?
A: The addition of alkaline metal catalysts like sodium methylate significantly reduces reaction time from 48 hours to approximately 10-12 hours while increasing yield from roughly 60% to over 70%.
Q: Which catalysts are suitable for the cyclization of Compound II?
A: The patent specifies sodium methylate, sodium hydride, or potassium hydride as effective catalysts, typically used at 2%-8% of the mass of Compound II.
Q: How is the final purity of the cefozopran side chain acid ensured?
A: High purity (>98%) is achieved through controlled hydrolysis conditions at 50°C and subsequent recrystallization or precipitation steps using ethyl acetate and ether.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefozopran Side Chain Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies promised by patents like CN102127034B are fully realized in our manufacturing suites. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of cefozopran side chain acid meets the exacting standards required for GMP API synthesis. Our commitment to quality ensures that the impurity profiles of our intermediates are tightly controlled, safeguarding the safety and efficacy of the final medicinal product.
We invite pharmaceutical partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific supply chain needs. By optimizing the synthesis of key intermediates, we can help you reduce overall production costs and secure a stable supply of critical materials. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. Let us demonstrate how our advanced catalytic capabilities can become a strategic asset in your antibiotic manufacturing portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
