Revolutionizing Seratrodast Production: A High-Yield Commercial Scale-Up Strategy for Global Pharma
Revolutionizing Seratrodast Production: A High-Yield Commercial Scale-Up Strategy for Global Pharma
The global demand for effective asthma therapeutics continues to rise, driven by increasing environmental pollutants and respiratory health challenges worldwide. At the forefront of this therapeutic class is Seratrodast, a potent thromboxane A2 receptor antagonist that offers significant clinical benefits over traditional treatments. A pivotal advancement in the manufacturing of this critical active pharmaceutical ingredient is detailed in patent CN100999452B, which outlines a novel, high-efficiency synthetic route. This technical insight report analyzes the transformative potential of this methodology, specifically designed for R&D directors and procurement leaders seeking a reliable pharmaceutical intermediates supplier. By shifting away from legacy processes, manufacturers can achieve substantial improvements in yield and operational efficiency, ensuring a stable supply of high-purity Seratrodast for the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Seratrodast has relied heavily on routes utilizing monoethyl pimelate as the primary starting material. This conventional approach presents significant chemical engineering bottlenecks that hinder cost reduction in API manufacturing. The fundamental flaw lies in the necessity to protect one carboxyl group of pimelic acid via ethyl esterification, a reaction notorious for its poor selectivity. This lack of selectivity inevitably leads to the formation of diethyl pimelate by-products, which are chemically similar to the desired intermediate and extremely difficult to separate. Furthermore, the requirement for subsequent deprotection steps elongates the synthetic timeline, introducing additional unit operations that consume energy and solvents without adding value. Consequently, the total yield when starting from pimelic acid often struggles to reach 40%, creating a massive burden on raw material consumption and waste management systems.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN100999452B introduces a streamlined five-step sequence that bypasses these historical inefficiencies entirely. By utilizing cycloheptanone as the foundational building block, the new route eliminates the need for cumbersome protection and deprotection strategies. This strategic shift allows for a direct construction of the carbon skeleton through a Grignard reaction followed by dehydration and oxidative cleavage. The result is a robust process where individual step yields consistently range between 90% and 95%, culminating in an impressive total yield of 58% to 78%. This dramatic improvement in atomic economy not only reduces the cost of goods sold but also simplifies the downstream purification workflow, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into the Cycloheptanone-Based Synthesis
The core of this innovative synthesis lies in the precise manipulation of the seven-membered ring structure. The process initiates with a Grignard reaction where cycloheptanone reacts with a phenyl magnesium halide, such as phenylmagnesium bromide, at temperatures between 40°C and 120°C. This nucleophilic addition efficiently constructs the quaternary carbon center, yielding 1-phenylcycloheptanol. Subsequent dehydration using agents like acetic anhydride or polyphosphoric acid converts the alcohol into 1-phenylcycloheptene. The critical transformation occurs in the third step, where oxidative cleavage of the alkene double bond is performed using oxidants like chromic anhydride or potassium permanganate. This step effectively opens the ring to generate 6-benzoylhexanoic acid, establishing the linear chain required for the final drug structure while maintaining the integrity of the ketone functionality for later reduction.
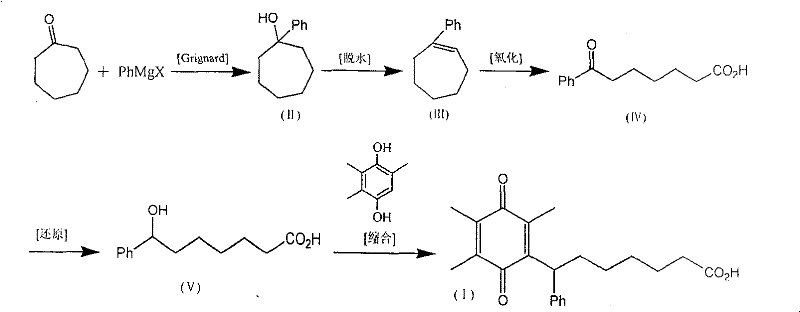
Following the ring opening, the ketone group undergoes selective reduction using mild reducing agents such as sodium borohydride or potassium borohydride at controlled temperatures of 0°C to 80°C. This step is crucial for generating the 7-hydroxy-7-phenylheptanoic acid intermediate without affecting the carboxylic acid moiety. The final stage involves a condensation reaction with trimethylhydroquinone catalyzed by boron trifluoride etherate, followed by oxidation with ferric chloride. This tandem process installs the quinone ring system characteristic of Seratrodast. The mechanistic elegance of this route ensures that impurities are minimized at every stage, providing R&D teams with a clear path to achieving high-purity OLED material or pharmaceutical grade standards depending on the application, though here specifically targeted for asthma therapy.
How to Synthesize Seratrodast Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly during the Grignard initiation and the oxidative cleavage phases. The protocol dictates strict control over molar ratios, such as maintaining the phenyl Grignard reagent to cycloheptanone ratio between 0.45 and 1.45 to maximize conversion while minimizing excess reagent waste. Workup procedures involve standard extraction and distillation techniques, with specific emphasis on removing unreacted bromobenzene via steam distillation to ensure product clarity. The detailed standardized synthetic steps, including exact solvent volumes, stirring rates, and crystallization protocols necessary for GMP compliance, are outlined in the technical guide below.
- Perform Grignard reaction between cycloheptanone and phenyl magnesium halide to form 1-phenylcycloheptanol.
- Dehydrate the alcohol intermediate using acidic agents to generate 1-phenylcycloheptene.
- Execute oxidative cleavage of the alkene to produce 6-benzoylhexanoic acid, followed by selective reduction.
- Condense the reduced acid with trimethylhydroquinone and treat with ferric chloride to finalize Seratrodast.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling economic and logistical benefits. The elimination of protection-deprotection cycles drastically simplifies the bill of materials, reducing the number of distinct raw materials that need to be sourced and qualified. This simplification directly translates to enhanced supply chain reliability, as the dependency on specialized protecting group reagents is removed. Furthermore, the mild reaction conditions, which mostly operate at atmospheric pressure and moderate temperatures, reduce the energy load on manufacturing facilities. This allows for the utilization of standard glass-lined or stainless steel reactors without the need for exotic high-pressure equipment, thereby lowering capital expenditure requirements for production scale-up.
- Cost Reduction in Manufacturing: The most significant financial advantage stems from the substantial increase in overall yield, which nearly doubles compared to traditional pimelic acid routes. By achieving total yields of up to 78%, the consumption of starting materials per kilogram of final product is significantly reduced. Additionally, the avoidance of difficult separation processes for diethyl ester by-products lowers solvent usage and waste disposal costs. The qualitative improvement in process efficiency means that labor hours and utility consumption are optimized, driving down the overall cost of production without compromising on quality standards.
- Enhanced Supply Chain Reliability: The starting material, cycloheptanone, is a commodity chemical with a stable global supply, unlike some specialized ester intermediates that may face availability fluctuations. The robustness of the reaction conditions ensures that production schedules are less likely to be disrupted by sensitive parameter deviations. This stability is critical for reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demand spikes. The simplified workflow also reduces the risk of batch failures, ensuring a consistent flow of material to downstream API synthesis units.
- Scalability and Environmental Compliance: From an environmental perspective, the shorter synthetic route inherently generates less chemical waste, aligning with green chemistry principles. The ability to perform reactions at lower temperatures reduces the carbon footprint associated with heating and cooling cycles. Moreover, the process avoids the use of highly hazardous reagents often associated with older chlorination methods, improving workplace safety and regulatory compliance. This makes the technology highly scalable, facilitating the transition from pilot plant batches to multi-ton commercial production with minimal technical barriers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Seratrodast synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for stakeholders evaluating the feasibility of adopting this route for their own manufacturing portfolios or sourcing strategies.
Q: What are the primary advantages of the cycloheptanone route over the monoethyl pimelate method?
A: The cycloheptanone route eliminates the need for complex protection and deprotection steps required in the monoethyl pimelate method. This significantly shortens the synthetic sequence, improves overall selectivity, and avoids the formation of difficult-to-remove diethyl ester by-products, leading to higher total yields ranging from 58% to 78%.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is highly amenable to commercial scale-up. The reaction conditions are mild, with most steps operating between 0°C and 120°C, and do not require extreme pressure or specialized anhydrous conditions beyond the initial Grignard step. The use of common reagents like acetic anhydride and sodium borohydride ensures supply chain stability.
Q: How does this process impact the impurity profile of the final API intermediate?
A: By avoiding the disproportionation reactions common in ester-based routes, this method produces a cleaner crude product. The selective reduction and specific oxidative cleavage steps minimize side reactions, resulting in a simplified purification process and a final product that meets stringent purity specifications for asthma medication manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Seratrodast Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthesis routes in maintaining a competitive edge in the pharmaceutical industry. Our team of expert chemists has thoroughly evaluated the pathway described in CN100999452B and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Seratrodast intermediate meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through this advanced technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can support your long-term strategic goals in the respiratory therapeutic market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
