Advanced Catalytic Cyclization for Industrial Scale 2-Phenylbenzoxazole Production
Advanced Catalytic Cyclization for Industrial Scale 2-Phenylbenzoxazole Production
The rapid expansion of the organic electronics and pharmaceutical sectors has intensified the demand for high-performance heterocyclic compounds, specifically 2-phenylbenzoxazole derivatives, which serve as critical building blocks for OLED materials, fluorescent probes, and bioactive agents. A groundbreaking preparation method detailed in patent CN108586374B introduces a robust, environmentally friendly synthetic pathway that addresses the longstanding inefficiencies of traditional manufacturing. This innovative approach leverages a telescoped condensation and acid-catalyzed cyclization strategy, utilizing readily available 2-aminophenol and benzoyl chloride precursors to achieve exceptional yields and purity. By shifting away from corrosive polyphosphoric acid systems, this technology offers a sustainable alternative that aligns with modern green chemistry principles while maintaining the rigorous quality standards required by global supply chains. As a reliable electronic chemical supplier, understanding these mechanistic advancements is crucial for securing long-term material availability.
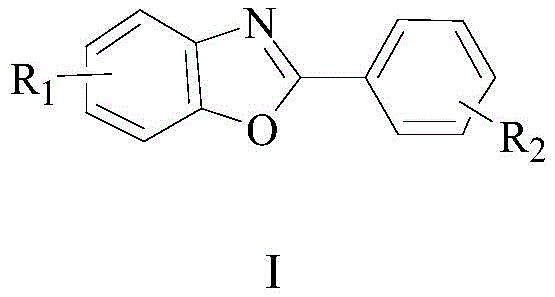
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-phenylbenzoxazole compounds has relied heavily on harsh reaction conditions involving polyphosphoric acid (PPA) dissolved in N-methylpyrrolidone (NMP). This legacy methodology presents severe operational and environmental challenges that hinder scalability and cost-efficiency. The process typically requires a massive excess of NMP solvent, often up to 24 times the mass of the reactants, alongside significant quantities of PPA, leading to extremely high raw material costs. Furthermore, the post-reaction workup is notoriously difficult; diluting the viscous reaction mass into water generates substantial volumes of phosphorus-containing wastewater that are complex and expensive to treat. These factors collectively create a bottleneck for manufacturers aiming to scale production, as the environmental burden and solvent recovery costs render the process economically unviable for large-volume applications in the competitive fine chemicals market.
The Novel Approach
In stark contrast, the novel preparation method disclosed in the patent data revolutionizes the synthesis landscape by employing a streamlined, acid-catalyzed cyclization in common organic solvents. This approach initiates with a condensation reaction between substituted 2-aminophenols and benzoyl chlorides in solvents such as xylene or toluene, forming an intermediate amide in situ. Subsequently, a catalytic amount of acid, such as p-toluenesulfonic acid (PTS), is introduced to drive the cyclization at moderate temperatures ranging from 135°C to 140°C. This strategic shift eliminates the need for stoichiometric amounts of dehydrating agents and toxic polar aprotic solvents. The resulting process not only simplifies the isolation of the final product through straightforward cooling and filtration but also drastically reduces the environmental footprint, making it an ideal solution for cost reduction in electronic chemical manufacturing.
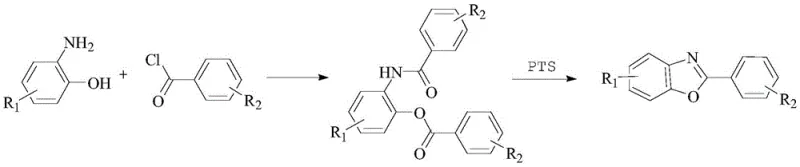
Mechanistic Insights into Acid-Catalyzed Cyclization
The core of this technological breakthrough lies in the efficient management of the dehydration cyclization step, which is traditionally the rate-limiting factor in benzoxazole formation. In the patented process, the initial nucleophilic attack of the amino group of the 2-aminophenol on the carbonyl carbon of the benzoyl chloride forms an N-(2-hydroxyphenyl)benzamide intermediate. Unlike traditional methods that force this equilibrium using massive excesses of reagents, the new method utilizes the thermal energy of the refluxing solvent combined with the proton-donating capability of the acid catalyst. The acid activates the carbonyl oxygen or the hydroxyl group, facilitating the intramolecular nucleophilic attack of the phenolic oxygen onto the amide carbon. This mechanism promotes the elimination of water and the subsequent aromatization of the oxazole ring under relatively mild conditions, ensuring high conversion rates without degrading sensitive functional groups on the aromatic rings.
Impurity control is another critical aspect where this mechanism excels, particularly for applications requiring high-purity OLED material precursors. The choice of p-toluenesulfonic acid as a catalyst, rather than inorganic strong acids, allows for better solubility control and minimizes side reactions such as hydrolysis of the acid chloride or polymerization of the phenol. Additionally, the final purification step involves washing the crude product with methanol, which effectively removes residual starting materials and soluble byproducts while retaining the highly crystalline benzoxazole product. This precise control over the reaction environment ensures that the final impurity profile is minimal, consistently achieving HPLC purities above 99.5%, which is essential for preventing quenching effects in luminescent devices and ensuring safety in pharmaceutical applications.
How to Synthesize 2-Phenylbenzoxazole Efficiently
The operational simplicity of this synthesis route makes it highly attractive for process chemists looking to transfer laboratory protocols to pilot and commercial scales. The procedure begins by dissolving the 2-aminophenol derivative in xylene, followed by the addition of the benzoyl chloride at elevated temperatures to ensure rapid condensation. Once the intermediate is formed, the cyclization catalyst is added directly to the same vessel, eliminating the need for intermediate isolation and reducing solvent consumption. The reaction mixture is then heated to promote ring closure, after which methanol is introduced to induce crystallization. This one-pot telescoped strategy significantly reduces processing time and equipment usage. For detailed standard operating procedures and specific parameter optimizations, please refer to the technical guide below.
- Perform condensation reaction between 2-aminophenol and benzoyl chloride in xylene at 115-120°C for 1 hour.
- Add p-toluenesulfonic acid catalyst directly to the mixture and heat to 135-140°C for cyclization over 3 hours.
- Cool the reaction, add methanol for precipitation, filter, and dry to obtain high-purity 2-phenylbenzoxazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers profound strategic benefits beyond mere technical superiority. The primary advantage lies in the drastic simplification of the supply chain for raw materials; by replacing specialized, high-cost solvents like NMP and corrosive polyphosphoric acid with commodity chemicals like xylene and p-toluenesulfonic acid, manufacturers can secure more stable pricing and availability. This shift mitigates the risk of supply disruptions caused by regulatory restrictions on hazardous solvents. Furthermore, the simplified workup procedure, which relies on filtration rather than complex aqueous extractions and distillations, translates to significantly reduced utility consumption and labor costs. These operational efficiencies compound to offer substantial cost savings in the overall manufacturing budget, allowing companies to remain competitive in price-sensitive markets while maintaining high margins.
- Cost Reduction in Manufacturing: The elimination of expensive N-methylpyrrolidone (NMP) and the reduction in solvent volume from 24 equivalents to roughly 24 parts by weight of cheaper xylene results in a direct decrease in raw material expenditure. Additionally, the ability to recycle the mother liquor and the avoidance of costly phosphorus waste treatment further enhance the economic viability of the process. By removing the need for stoichiometric amounts of polyphosphoric acid, the process also reduces the corrosion burden on reactor vessels, extending equipment lifespan and lowering maintenance overheads significantly.
- Enhanced Supply Chain Reliability: Utilizing widely available commodity solvents and catalysts ensures that production schedules are not held hostage by the limited availability of specialty reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and slight variations in stoichiometry, means that batch-to-batch consistency is high, reducing the rate of failed batches and ensuring a steady flow of finished goods. This reliability is critical for downstream customers in the electronics and pharma sectors who depend on just-in-time delivery models to maintain their own production lines without interruption.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, specifically avoiding the generation of phosphorus-rich wastewater that requires specialized treatment facilities. This environmental compatibility simplifies the permitting process for new production lines and ensures compliance with increasingly stringent global environmental regulations. The straightforward filtration and drying steps are easily adaptable to large-scale reactors, facilitating the commercial scale-up of complex benzoxazole derivatives from kilogram to multi-ton quantities without the need for extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear picture of what partners can expect when adopting this methodology. Understanding these nuances helps in making informed decisions regarding technology transfer and long-term sourcing strategies.
Q: How does this new method improve environmental compliance compared to traditional polyphosphoric acid routes?
A: The novel method eliminates the use of N-methylpyrrolidone (NMP) and massive amounts of polyphosphoric acid, thereby preventing the generation of difficult-to-treat phosphorus-containing wastewater and significantly reducing hazardous waste disposal costs.
Q: What purity levels can be achieved using this acid-catalyzed cyclization process?
A: Experimental data from the patent indicates that the process consistently yields products with HPLC purity exceeding 99.5%, meeting the stringent requirements for high-end luminescent materials and pharmaceutical intermediates without complex purification steps.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes common organic solvents like xylene and methanol, operates at manageable temperatures (115-140°C), and involves simple filtration workups, making it highly scalable from pilot plants to multi-ton production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Phenylbenzoxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced materials depends on more than just a promising patent; it requires a partner with the technical depth to execute complex chemistry at scale. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to factory floor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Whether you require custom derivatives for OLED applications or bulk intermediates for pharmaceutical synthesis, our infrastructure is designed to support your growth with reliability and precision.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your specific application needs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your current procurement volumes, demonstrating exactly how switching to our optimized process can improve your bottom line. Please contact our technical procurement team today to request specific COA data for our 2-phenylbenzoxazole portfolio and to discuss route feasibility assessments for your next project. Let us help you secure a sustainable, high-quality supply chain for your critical chemical intermediates.
