Revolutionizing Cefotetan Acid Production: A Deep Dive into Advanced Purification and Scalability
Revolutionizing Cefotetan Acid Production: A Deep Dive into Advanced Purification and Scalability
The pharmaceutical industry constantly seeks robust methodologies to enhance the purity and yield of critical antibiotic intermediates, particularly within the cephalosporin class. Patent CN101870704A introduces a groundbreaking purification technique specifically designed for cefotetan acid crude products, addressing the persistent challenge of isomer removal that has long plagued manufacturers. This innovation shifts the paradigm from energy-intensive cryogenic reactions to a highly efficient, room-temperature suspension washing protocol. By leveraging the distinct physicochemical properties between the target molecule and its structural isomers, this method ensures that isomer content is reduced to less than 1 percent, regardless of the initial impurity load. For R&D directors and process engineers, this represents a significant leap forward in operational simplicity and product quality control, offering a reliable pathway for producing high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of cefotetan has been bottlenecked by the rigorous conditions required for isomer transformation and removal. Traditional protocols necessitate maintaining reaction temperatures between -5°C and 0°C, a requirement that demands substantial energy investment in cooling infrastructure and precise thermal regulation. Furthermore, these processes rely on carbon dioxide to strictly regulate the pH value between 7.5 and 8.5, adding layers of complexity to the reactor control systems. Most critically, the reaction time to achieve an acceptable isomer content of less than 1 percent typically exceeds 72 hours. This extended duration not only ties up valuable reactor capacity but also increases the risk of product degradation over time, severely restricting the suitability for large-scale industrialized production of cefotetan raw materials.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a sequential physical washing strategy that eliminates the need for prolonged cryogenic reaction times. The process involves stirring and washing the crude cefotetan acid successively with ethanol, cyclohexane, and methanol at ambient temperatures. This method capitalizes on the solubility differences between the desired acid and its isomers, effectively stripping away impurities without altering the core molecular structure through harsh chemical treatments. 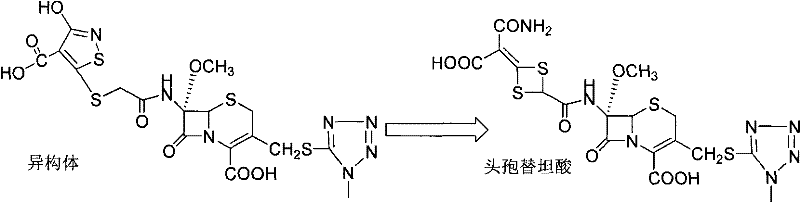 As illustrated in the reaction scheme, the conversion from isomer to the target cefotetan acid is the critical step; this new purification method ensures that even if the preceding conversion is incomplete, the final product quality remains uncompromised. The ability to operate at room temperature simplifies the equipment requirements and drastically reduces the operational window, making it an ideal solution for cost reduction in pharmaceutical intermediates manufacturing.
As illustrated in the reaction scheme, the conversion from isomer to the target cefotetan acid is the critical step; this new purification method ensures that even if the preceding conversion is incomplete, the final product quality remains uncompromised. The ability to operate at room temperature simplifies the equipment requirements and drastically reduces the operational window, making it an ideal solution for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Suspension Washing Purification
The core mechanism driving this purification success lies in the differential solubility and interaction of the solvents with the crystal lattice of the crude product. The process initiates with ethanol, which acts as a primary washing agent to dissolve surface impurities and loosely bound isomers without dissolving the bulk cefotetan acid. Following this, cyclohexane is introduced; as a non-polar solvent, it effectively removes organic residues and specific hydrophobic impurities that ethanol might leave behind, further refining the solid matrix. The final wash with methanol serves as a polishing step, leveraging its specific polarity to displace any remaining trace isomers. This tri-solvent system creates a gradient of purification that is far more effective than single-solvent recrystallization, ensuring that the isomer content drops precipitously with each stage.
Furthermore, the mechanical aspect of the agitation plays a crucial role in the efficacy of this method. The patent specifies an agitation treatment time of 0.5 to 1 hour for each step, which is essential to guarantee that agglomerated blocks of the crude product are fully smashed and exposed to the solvent. This physical disruption prevents the phenomenon of impurity entrapment, where isomers might remain hidden within the interior of large crystal clusters. By combining precise solvent selection with controlled mechanical agitation and rapid filtration techniques such as vacuum filtration or high-speed centrifugation, the process achieves a level of purity that was previously only attainable through much more arduous chemical means, providing a robust framework for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Cefotetan Acid Efficiently
The synthesis and subsequent purification of cefotetan acid via this patented route offer a streamlined alternative to legacy processes, focusing on operational ease and high throughput. The method is designed to be flexible, accommodating crude products with varying initial isomer contents by adjusting solvent volumes rather than changing the fundamental process parameters. This adaptability is key for manufacturing environments where feedstock quality may fluctuate. The following guide outlines the standardized operational procedure derived from the patent examples, ensuring consistent results across different batch sizes.
- Suspend the crude cefotetan acid in ethanol at 10-25°C, stir for 0.5-1 hour, and filter to remove initial impurities.
- Wash the resulting filter cake with cyclohexane under the same temperature conditions for 0.5-1 hour, followed by filtration.
- Perform a final wash on the filter cake using methanol for 0.5-1 hour at room temperature, then filter to obtain high-purity cefotetan acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this purification technology translates directly into tangible operational efficiencies and risk mitigation. The elimination of the 72-hour cryogenic reaction requirement fundamentally alters the production timeline, allowing for significantly faster turnover of manufacturing assets. This acceleration in cycle time means that facilities can produce more batches within the same timeframe, effectively increasing capacity without the need for capital expenditure on new reactors. Additionally, the room-temperature operation removes the dependency on extensive cooling systems, leading to substantial cost savings in terms of energy consumption and maintenance of specialized low-temperature equipment.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of energy-intensive cooling steps and the optimization of material usage. By operating at room temperature, the facility avoids the high utility costs associated with maintaining sub-zero conditions over multi-day periods. Moreover, the patent highlights that the mother liquor remaining after washing can be concentrated and recycled back into the isomerization reaction. This closed-loop approach ensures that valuable materials are not lost as waste, thereby improving the overall atom economy and reducing the cost of goods sold without compromising the total yield of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex processes that are sensitive to minor deviations. This purification method enhances reliability by simplifying the operational parameters; there is no need for precise pH control using carbon dioxide or strict thermal regulation. The robustness of the washing process against variations in crude isomer content (handling levels higher than 8 percent) means that upstream synthesis fluctuations do not necessarily halt downstream purification. This flexibility ensures a more steady flow of high-purity cefotetan acid, reducing lead time for high-purity pharmaceutical intermediates and safeguarding against production bottlenecks.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process is inherently greener and easier to expand. The use of common solvents like ethanol, cyclohexane, and methanol simplifies waste management and solvent recovery compared to exotic reagents. The ability to perform purification at room temperature reduces the thermal load on the plant, aligning with modern sustainability goals. Furthermore, the simplicity of the unit operations—suspension, agitation, and filtration—makes the technology highly scalable from pilot plants to multi-ton commercial production, ensuring that quality remains consistent regardless of batch size.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method outperforms traditional techniques in terms of purity, yield, and operational efficiency. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines.
Q: What is the maximum isomer content this purification method can handle effectively?
A: Unlike conventional methods that struggle with high impurity loads, this patented process is robust enough to purify crude products with isomer content exceeding 8%, consistently reducing it to below 1%.
Q: How does this method improve upon traditional isomer conversion processes?
A: Traditional methods require cryogenic conditions (-5 to 0°C) and over 72 hours of reaction time with strict pH control. This new approach operates at room temperature and utilizes physical washing, significantly shortening the production cycle.
Q: Can the solvents used in the washing process be recycled?
A: Yes, the mother liquor generated after the washing steps can be concentrated and recycled back into the isomerization reaction, which enhances material efficiency without negatively impacting the total yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefotetan Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced purification technologies to meet the stringent demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the room-temperature washing process for cefotetan acid are seamlessly translated into industrial reality. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical methods to verify isomer content and overall quality. Our capability to handle complex cephalosporin intermediates positions us as a strategic partner for companies seeking to optimize their supply chains.
We invite you to collaborate with us to leverage these technological advancements for your specific production needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your current manufacturing setup, demonstrating exactly how switching to our optimized processes can reduce your operational overhead. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a stable, high-quality supply of cefotetan acid that drives your business forward.
