Advanced Catalytic Hydrogenation Route for High-Purity 4-Amino-3-Methylphenol Manufacturing
Introduction to Advanced 4-Amino-3-Methylphenol Synthesis
The global demand for high-performance functional materials and pharmaceutical intermediates continues to drive innovation in synthetic chemistry, particularly for phenolic amines like 4-amino-3-methylphenol. A pivotal advancement in this field is documented in Chinese patent CN103508908A, which outlines a robust and environmentally superior preparation method utilizing m-cresol as the primary feedstock. This technology represents a significant departure from legacy manufacturing routes that have long plagued the industry with toxicity and waste management issues. By leveraging a controlled nitrosation followed by a selective catalytic hydrogenation, this method achieves exceptional product quality with minimal environmental footprint. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding the nuances of this patent is critical for securing supply chains that meet stringent regulatory and purity standards. The process not only simplifies post-treatment operations but also ensures the stability required for downstream applications in high-end resins and carbon fiber precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 4-amino-3-methylphenol has relied on several problematic methodologies that pose severe challenges for modern chemical manufacturing. One traditional approach involves the reduction of ortho-nitrotoluene derivatives, which often necessitates the use of toxic mercury salts in electrochemical methods or expensive precious metals like platinum and rhodium in acidic media, leading to prohibitively high operational costs and low yields. Another common route utilizes azo-compound reduction or 4-nitro-3-methylphenol reduction, but these suffer from the scarcity of raw materials and excessively long synthetic sequences that erode profit margins. Furthermore, methods employing hydrogen sulfide for reduction, while simple, introduce extreme safety hazards due to the toxicity of H2S gas, requiring costly safety infrastructure that diminishes economic viability. Perhaps the most widespread legacy method involves iron powder reduction in acetic acid; however, this generates massive quantities of iron oxide sludge that are difficult to filter and dispose of, creating a significant environmental burden and contaminating the final product with residual iron, rendering it unsuitable for high-end electronic or resin applications.
The Novel Approach
In stark contrast to these archaic techniques, the novel approach detailed in the patent data introduces a streamlined two-step synthesis starting from readily available m-cresol. This method replaces hazardous reducing agents with clean catalytic hydrogenation, fundamentally altering the impurity profile of the final product. By utilizing a nitrosation reaction followed by hydrogenation in an alcoholic solvent, the process eliminates the generation of heavy metal sludge and toxic gaseous byproducts entirely. The use of heterogeneous catalysts such as palladium on carbon or Raney nickel allows for easy separation and potential recycling, drastically simplifying the work-up procedure. This shift not only aligns with green chemistry principles but also directly addresses the pain points of cost reduction in fine chemical manufacturing by reducing waste disposal fees and raw material complexity. The resulting product exhibits superior stability and purity, making it an ideal candidate for sensitive applications where trace metal contamination is unacceptable, thereby opening new market opportunities for suppliers who can adopt this technology.
Mechanistic Insights into Nitrosation and Catalytic Hydrogenation
The core of this synthetic strategy lies in the precise control of electrophilic aromatic substitution during the nitrosation phase. In this step, m-cresol is dissolved in an aqueous sodium hydroxide solution to form the phenolate anion, which is significantly more nucleophilic than the neutral phenol. Upon the addition of sodium nitrite and hydrochloric acid, nitrous acid is generated in situ, which acts as the electrophile. Due to the strong activating effect of the hydroxyl group and the steric hindrance of the methyl group at the meta position, the nitrosation occurs selectively at the para-position relative to the hydroxyl group, yielding 4-nitroso-3-methylphenol. Maintaining the reaction temperature strictly between 3°C and 10°C is mechanistically crucial to suppress the formation of ortho-isomers and prevent oxidative degradation of the phenolic ring. This selectivity is the foundation of the high purity observed in the final product, as it minimizes the burden on downstream purification steps.
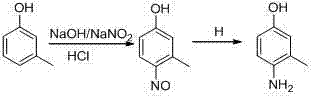
Following nitrosation, the transformation of the nitroso group to an amino group is achieved through catalytic hydrogenation, a mechanism that offers superior atom economy compared to stoichiometric reductions. In the presence of a promoter such as ammonia or organic amines, the catalyst surface (typically Pd/C or Raney Nickel) facilitates the adsorption of hydrogen gas and the nitroso substrate. The promoter plays a vital role in modulating the electronic environment of the catalyst, preventing over-reduction or hydrogenolysis of the phenolic hydroxyl group, which is a common side reaction in phenolic amine synthesis. The reaction proceeds smoothly at mild temperatures of 20°C to 40°C in alcoholic solvents like methanol or ethanol, which serve both as the reaction medium and as proton donors. This mild condition preserves the integrity of the aromatic ring and ensures that the only significant byproduct is water, further contributing to the cleanliness of the process and the ease of isolating high-purity 4-amino-3-methylphenol through simple crystallization.
How to Synthesize 4-Amino-3-Methylphenol Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and thermal management to ensure reproducibility and safety on a commercial scale. The process begins with the preparation of the nitroso intermediate, where m-cresol is reacted with sodium nitrite under acidic conditions at low temperatures, followed by isolation via filtration. This intermediate is then subjected to hydrogenation in a pressurized reactor with a selected catalyst and promoter system, where hydrogen uptake is monitored to determine the reaction endpoint. Finally, the crude amine is purified through a recrystallization process using low-boiling alcohols, which removes any remaining catalyst traces and unreacted starting materials. For detailed operational parameters, stoichiometry, and safety protocols required for GMP-compliant production, please refer to the standardized synthesis guide below.
- Perform nitrosation of m-cresol using sodium nitrite and hydrochloric acid in an alkaline aqueous solution at 3-10°C to form 4-nitroso-3-methylphenol.
- Conduct catalytic hydrogenation of the nitroso intermediate in an alcoholic solvent (methanol/ethanol) using Pd/C or Raney Nickel catalyst with ammonia promotion at 20-40°C.
- Purify the crude product through dissolution in low-boiling alcohol, cooling crystallization, filtration, and vacuum drying at 30-60°C to achieve >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic hydrogenation technology offers transformative advantages that extend far beyond simple chemical yield improvements. The elimination of iron powder and toxic sulfides from the process flow fundamentally reshapes the cost structure of manufacturing, removing the need for expensive hazardous waste treatment and complex filtration equipment designed to handle heavy metal sludge. This simplification of the production line translates directly into substantial cost savings, as the facility can operate with lower overheads regarding environmental compliance and waste disposal logistics. Furthermore, the reliance on commodity chemicals like m-cresol, methanol, and hydrogen ensures a stable and resilient supply chain, insulating buyers from the volatility associated with specialized or scarce reagents used in older synthetic routes. The ability to produce high-purity material consistently also reduces the risk of batch rejection by downstream customers, enhancing the overall reliability of the supply partnership.
- Cost Reduction in Manufacturing: The transition from stoichiometric reducing agents to catalytic hydrogenation eliminates the massive material costs associated with purchasing and disposing of tons of iron powder or zinc dust. Since the catalyst loading is minimal (typically 0.005 to 0.05 ratio) and the solvent can be recovered and recycled multiple times through distillation, the variable cost per kilogram of product is significantly lowered. Additionally, the simplified purification process reduces energy consumption and labor hours, contributing to a leaner and more competitive manufacturing model that can withstand market price fluctuations.
- Enhanced Supply Chain Reliability: By utilizing m-cresol as the starting material, which is a bulk petrochemical derivative with a mature global supply network, manufacturers can avoid the bottlenecks caused by niche intermediates. The robustness of the catalytic process means that production schedules are less prone to disruption from reagent shortages or quality variability. This stability is crucial for long-term contracts in the pharmaceutical and electronics sectors, where continuity of supply is often valued higher than marginal price differences, ensuring that partners can meet their own production deadlines without interruption.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from laboratory bench scales up to multi-ton reactors without loss of efficiency or selectivity. The absence of heavy metal waste streams simplifies the permitting process for new manufacturing sites and reduces the regulatory burden on existing facilities. This environmental compatibility not only future-proofs the supply chain against tightening global regulations but also aligns with the sustainability goals of major multinational corporations, making the product more attractive to eco-conscious buyers seeking green chemistry solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4-amino-3-methylphenol using this advanced methodology. These insights are derived directly from the experimental data and process descriptions found in the relevant patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions regarding specification requirements and process integration.
Q: How does this method improve product purity compared to traditional iron powder reduction?
A: Unlike traditional iron powder reduction which generates difficult-to-remove ferric oxide residues and heavy metal contaminants, this catalytic hydrogenation method uses clean heterogeneous catalysts (Pd/C or Raney Ni) that are easily filtered off, resulting in significantly higher purity (>99.5%) suitable for high-end resin applications.
Q: What are the critical temperature controls required for the nitrosation step?
A: The nitrosation reaction is highly exothermic and requires strict temperature control between 3°C and 10°C. Maintaining this low temperature range is essential to prevent side reactions and ensure the selective formation of the para-nitroso derivative over ortho-isomers or oxidation byproducts.
Q: Can the alcoholic solvent be recycled in this process to reduce costs?
A: Yes, the process utilizes low-boiling alcohols like methanol or ethanol which can be efficiently recovered via distillation under reduced pressure after the reaction. This solvent recovery loop significantly lowers raw material consumption and reduces waste disposal costs, enhancing overall process economics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Amino-3-Methylphenol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into tangible commercial reality through expert process engineering and rigorous quality control. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this catalytic hydrogenation route are fully realized in every batch we deliver. Our facilities are equipped with state-of-the-art hydrogenation reactors and stringent purity specifications are enforced through our rigorous QC labs, guaranteeing that the 4-amino-3-methylphenol supplied meets the exacting standards required for high-end resin and pharmaceutical applications. We bridge the gap between innovative academic research and industrial-scale reliability, offering a secure source for critical intermediates.
We invite global partners to engage with our technical team to explore how this optimized synthesis route can enhance your product portfolio and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to our high-purity grades. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments tailored to your specific volume requirements, ensuring a seamless integration of our materials into your supply chain.
