Advanced Synthesis of 7-Oxo-Abiraterone Acetate for Pharmaceutical Quality Control
Introduction to Advanced Impurity Synthesis
The pharmaceutical industry faces increasing regulatory pressure to fully characterize the impurity profiles of active pharmaceutical ingredients (APIs), particularly for potent oncology drugs like Abiraterone Acetate. Patent CN111253461B introduces a groundbreaking synthesis method for 7-oxo-abiraterone acetate, a specific oxidative degradation product that has historically been difficult to access in high purity. This technical breakthrough addresses a critical gap in the supply chain for reference standards and quality control materials. By enabling the directional synthesis of this specific impurity, manufacturers can now conduct rigorous safety, reliability, and stability studies that were previously hindered by the lack of authentic samples. The method described leverages a robust sequence of acetylation, selective oxidation, and palladium-catalyzed cross-coupling to deliver a product with exceptional chemical integrity.
For R&D directors and quality assurance teams, the availability of such high-purity impurities is not merely a convenience but a regulatory necessity. The ability to quantify trace degradants ensures that the final drug product meets stringent pharmacopoeial standards. This patent outlines a pathway that transforms a complex structural challenge into a manageable, reproducible industrial process. The strategic value lies in the method's capacity to generate substantial quantities of the target molecule with minimal byproduct formation, thereby streamlining the analytical validation processes required for global market approval of Abiraterone-based therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
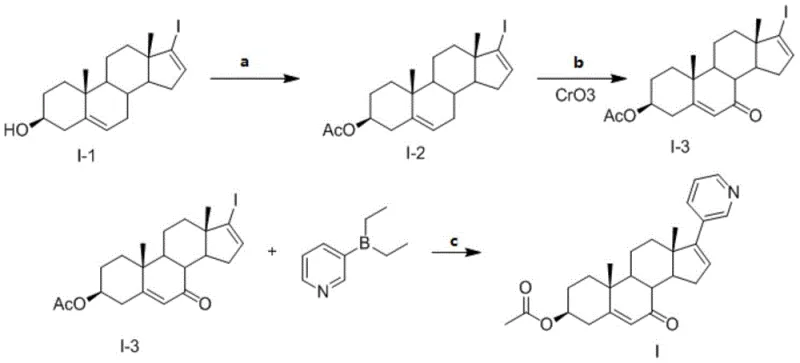
Traditionally, synthesizing specific oxidative impurities of steroidal APIs like Abiraterone has been fraught with challenges related to regioselectivity and yield. Conventional oxidation methods often lack the precision required to introduce a ketone functionality specifically at the C7 position without affecting other sensitive sites on the steroid nucleus, such as the C3 hydroxyl group or the C16-C17 double bond. Non-selective oxidants can lead to a complex mixture of over-oxidized products, requiring laborious and costly purification steps that drastically reduce overall throughput. Furthermore, earlier attempts to functionalize the C17 position with the pyridyl moiety prior to oxidation often resulted in decomposition of the heterocyclic ring under harsh oxidative conditions. These inefficiencies created a bottleneck in the supply of reliable reference standards, forcing quality control laboratories to rely on isolation from degradation mixtures, which is inherently unreliable and inconsistent.
The Novel Approach
The novel approach detailed in the patent circumvents these historical limitations through a cleverly designed linear synthesis that prioritizes chemoselectivity at every stage. By protecting the C3 hydroxyl group as an acetate ester prior to oxidation, the method effectively shields this position from unwanted side reactions, allowing the chromium trioxide reagent to target the C7 position exclusively. Following this precise oxidation, the strategy employs a Suzuki-Miyaura coupling reaction to install the 3-pyridyl group at the C17 position. This sequence ensures that the sensitive pyridine ring is introduced only after the harsh oxidative conditions have been completed, preserving its structural integrity. The result is a streamlined process that delivers the target 7-oxo-abiraterone acetate with purity exceeding 98%, eliminating the need for complex preparative HPLC purification.
This strategic reordering of synthetic steps represents a significant advancement in process chemistry for steroidal intermediates. It demonstrates how careful protection group strategy combined with modern transition metal catalysis can solve long-standing problems in impurity synthesis. For procurement managers, this translates to a more stable supply of critical reference materials, as the synthesis relies on readily available starting materials and robust reaction conditions that are easily transferable to large-scale manufacturing environments. The elimination of ambiguous isolation steps in favor of a defined synthetic route ensures batch-to-batch consistency, which is paramount for regulatory submissions.
Mechanistic Insights into Suzuki Coupling and Regioselective Oxidation
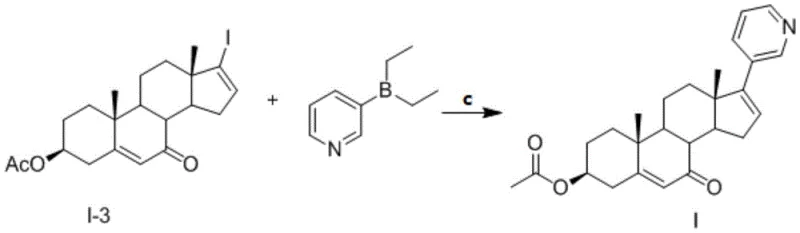
The core of this synthesis lies in the interplay between the regioselective oxidation of the steroid backbone and the subsequent palladium-catalyzed cross-coupling. The oxidation step utilizes chromium trioxide in a specialized solvent system comprising acetone and N-hydroxyphthalimide (NOP). This specific medium facilitates the activation of the allylic C-H bond at the C7 position while leaving the delta-5 double bond and the C3 acetate intact. The mechanism likely involves the formation of a chromate ester intermediate that undergoes elimination to generate the enone system. The presence of NOP may act as a radical mediator or stabilizer, enhancing the selectivity and preventing over-oxidation to carboxylic acids or cleavage of the steroid rings. This level of control is essential for maintaining the stereochemical integrity of the chiral centers at C8, C9, C10, and C13.
Following oxidation, the synthesis proceeds via a Suzuki coupling reaction, a powerful tool for forming carbon-carbon bonds between sp2 hybridized carbons. In this specific application, the 17-iodo-androstene derivative acts as the electrophile, reacting with diethyl(3-pyridyl)borane as the nucleophilic partner. The reaction is catalyzed by a palladium species, such as bis(triphenylphosphine)palladium chloride, in the presence of an inorganic base like potassium phosphate heptahydrate. The base activates the boron species to form a reactive boronate complex, which then undergoes transmetallation with the palladium center. Subsequent reductive elimination releases the final coupled product and regenerates the active palladium catalyst. This mechanism is highly tolerant of the ketone functionality introduced in the previous step, showcasing the orthogonality of the reaction conditions.
Understanding these mechanistic nuances is vital for scaling the process. The choice of solvent, specifically N-methylpyrrolidone (NMP) or dimethylformamide (DMF), is critical for solubilizing the polar steroid intermediate and facilitating the interaction between the inorganic base and the organoboron reagent. The temperature range of 80-100°C provides sufficient energy to overcome the activation barrier for the oxidative addition of the sterically hindered C17-iodide without promoting thermal decomposition of the product. This deep mechanistic understanding allows process chemists to fine-tune reaction parameters, ensuring that impurity levels remain below detection limits and that the final crystallization step yields a product of pharmaceutical grade quality.
How to Synthesize 7-Oxo-Abiraterone Acetate Efficiently
The synthesis of this high-value pharmaceutical intermediate is achieved through a logical four-step sequence that balances reaction efficiency with ease of operation. The process begins with the protection of the starting alcohol, followed by the critical oxidation and coupling steps, and concludes with a straightforward purification. Each step has been optimized to maximize yield and minimize the formation of difficult-to-remove byproducts. The detailed protocol provided in the patent offers a clear roadmap for laboratory and pilot plant execution, utilizing standard equipment and reagents that are accessible to most fine chemical manufacturers. This accessibility lowers the barrier to entry for producing this critical quality control material.
- Acetylate 17-iodoandrost-5,16-diene-3-beta-ol with acetic anhydride in pyridine at 40-60°C to form the 3-beta-acetate intermediate.
- Oxidize the 3-beta-acetate intermediate using chromium trioxide in acetone/NOP solvent system to introduce the 7-oxo ketone functionality.
- Perform a Suzuki coupling reaction between the 7-oxo-iodo-intermediate and diethyl(3-pyridyl)borane using a palladium catalyst and inorganic base.
- Purify the crude product via recrystallization in anhydrous ethanol to achieve purity levels exceeding 98%.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis method offers substantial advantages that directly impact the bottom line and supply chain resilience. The primary benefit stems from the drastic simplification of the purification workflow. Traditional methods for generating steroidal impurities often require multiple chromatographic separations, which are expensive, time-consuming, and difficult to scale. By achieving high crude purity through selective chemistry, this new route significantly reduces the consumption of silica gel and organic solvents associated with column chromatography. This reduction in downstream processing translates directly into lower manufacturing costs and a smaller environmental footprint, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the use of commodity chemicals and the elimination of exotic reagents. The acetylation step uses acetic anhydride and pyridine, which are inexpensive and widely available. Similarly, the oxidation utilizes chromium trioxide, a standard industrial oxidant, rather than costly enzymatic or electrochemical alternatives. Furthermore, the high selectivity of the Suzuki coupling minimizes the loss of valuable steroid intermediates, improving the overall atom economy of the process. These factors combine to create a cost structure that is significantly more favorable than legacy isolation methods, allowing for competitive pricing of the final reference standard.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness of the reaction conditions. The process operates at moderate temperatures and pressures, reducing the risk of equipment failure or safety incidents that could halt production. The starting materials, such as 17-iodoandrost-5,16-diene-3-beta-ol, are derived from abundant steroidal precursors, ensuring a stable upstream supply. Additionally, the final recrystallization step using anhydrous ethanol provides a reliable method for polishing the product to meet strict specifications, ensuring that every batch delivered to the customer is consistent. This reliability is crucial for pharmaceutical companies that depend on timely access to impurity standards for regulatory filings.
- Scalability and Environmental Compliance: The transition from laboratory to commercial scale is facilitated by the use of homogeneous reaction conditions and common solvents. The avoidance of cryogenic conditions or high-pressure hydrogenation simplifies the engineering requirements for large-scale reactors. Moreover, the process generates less hazardous waste compared to non-selective oxidation routes, as the byproduct profile is cleaner and easier to treat. The ability to recycle solvents like ethanol and acetone further enhances the sustainability profile of the manufacturing process. This scalability ensures that suppliers can meet surging demand for Abiraterone quality control materials without compromising on delivery timelines or product quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 7-oxo-abiraterone acetate. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details helps stakeholders make informed decisions about integrating this material into their quality control workflows. The clarity of the synthesis route ensures that potential risks are minimized and that the material performs consistently in analytical applications.
Q: What is the primary application of 7-oxo-abiraterone acetate?
A: It serves as a critical reference impurity for the quality control and stability testing of Abiraterone Acetate (Zytiga), ensuring medication safety and regulatory compliance.
Q: How does this synthesis method improve purity compared to traditional routes?
A: The patented method utilizes a directed Suzuki coupling and specific oxidation steps that minimize side reactions, achieving purity levels greater than 98% without complex chromatographic separations.
Q: Is this process scalable for commercial production?
A: Yes, the process uses standard industrial reagents like chromium trioxide and palladium catalysts under moderate temperatures (80-100°C), making it highly suitable for scale-up from kilogram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Oxo-Abiraterone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity impurities play in the development and lifecycle management of oncology drugs. Our team of expert process chemists has extensively evaluated the synthesis route described in patent CN111253461B and confirmed its viability for commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project phase. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every gram of 7-oxo-abiraterone acetate we supply meets the highest international standards for reference materials.
We invite you to collaborate with us to secure a stable and cost-effective supply of this essential intermediate. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific procurement needs, demonstrating how our optimized manufacturing process can reduce your overall quality control costs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain partner committed to excellence, innovation, and the relentless pursuit of quality in pharmaceutical intermediates.
