Revolutionizing Valsartan Intermediate Production: A Deep Dive into Novel Catalytic Efficiency
Revolutionizing Valsartan Intermediate Production: A Deep Dive into Novel Catalytic Efficiency
The global demand for high-quality antihypertensive medications continues to drive innovation in the synthesis of critical pharmaceutical intermediates. Patent CN115197096A introduces a transformative approach to manufacturing N-(2'-cyanobiphenyl-4-methylene)-L-valine methyl ester hydrochloride, a pivotal precursor in the production of Valsartan. This technology addresses long-standing challenges in purity and process efficiency that have plagued traditional synthetic routes. By leveraging a specialized cobalt-catalyzed system, the method achieves exceptional yields while completely avoiding the formation of problematic dimer impurities. For R&D directors and procurement strategists, this represents a significant opportunity to optimize supply chains for high-purity pharmaceutical intermediates. The following analysis dissects the technical merits and commercial implications of this breakthrough.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key valsartan intermediate has relied heavily on the reaction between 2'-cyano-4-bromomethylbiphenyl and L-valine methyl ester hydrochloride. As illustrated in prior art such as Indian patent 2011MU00371 and various Chinese patents, these conventional pathways suffer from inherent inefficiencies. The crude products obtained are frequently oily substances, which present significant logistical challenges regarding quantification, transfer, and storage. Furthermore, these methods typically achieve yields of only around 90% and necessitate rigorous purification steps, such as column chromatography, to remove impurities. Most critically, these routes inevitably generate dimer impurities that are notoriously difficult to separate, thereby compromising the overall quality and safety profile of the final active pharmaceutical ingredient.
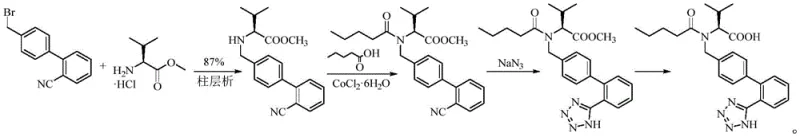
The Novel Approach
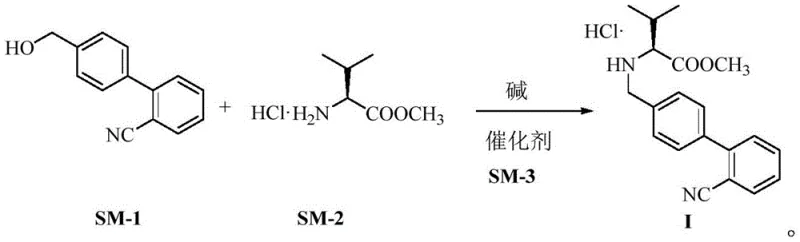
In stark contrast, the methodology disclosed in CN115197096A utilizes 4'-hydroxymethyl-2-cyanobiphenyl as the starting material, reacting it directly with L-valine methyl ester hydrochloride under the influence of a specific catalyst system. This strategic shift in starting materials and catalytic conditions fundamentally alters the reaction landscape. The process operates under mild conditions without the need for inert gas protection, significantly simplifying operational requirements. The result is a target product that isolates as a stable solid rather than an oil, with reported yields reaching as high as 96.8% and HPLC purity exceeding 99.89%. Crucially, analytical data confirms the complete absence of dimer impurities, marking a substantial improvement over legacy technologies.
Mechanistic Insights into Cobalt-Catalyzed N-Alkylation
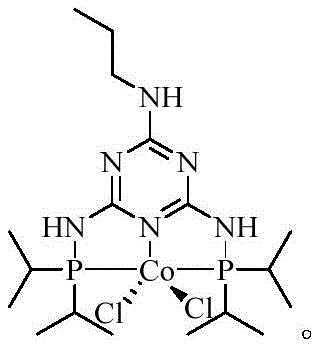
The core innovation of this patent lies in the utilization of a sophisticated cobalt-based catalyst, designated as SM-3. This catalyst features a complex coordination environment with phosphine ligands that modulate the electronic properties of the cobalt center. During the reaction, SM-3 likely facilitates the activation of the hydroxymethyl group on the biphenyl substrate, promoting a selective nucleophilic attack by the amine nitrogen of the valine ester. This mechanism proceeds with high regioselectivity and stereospecificity, ensuring that the chiral integrity of the L-valine moiety is preserved throughout the transformation. The mild thermal conditions (60-100°C) further support the stability of the intermediates, preventing thermal degradation or unwanted side reactions that are common in harsher alkylation protocols.
Impurity control is another critical aspect where this catalytic system excels. In traditional bromomethyl-based routes, the high reactivity of the benzylic bromide often leads to over-alkylation, resulting in the formation of secondary amine dimers. The SM-3 catalyst system appears to kinetically favor the mono-alkylation pathway while suppressing the reactivity of the newly formed secondary amine towards further substitution. Additionally, the use of a hydroxymethyl precursor instead of a bromomethyl one reduces the intrinsic electrophilicity of the substrate, requiring the catalyst to drive the reaction. This controlled activation prevents the runaway side reactions that generate dimers, leading to a much cleaner reaction profile and reducing the burden on downstream purification processes.
How to Synthesize N-(2'-cyanobiphenyl-4-methylene)-L-valine methyl ester hydrochloride Efficiently
The practical implementation of this synthesis involves a straightforward one-pot procedure that is highly amenable to scale-up. The process begins by combining the hydroxymethyl biphenyl substrate, the valine ester salt, the cobalt catalyst, and a base such as sodium methoxide in a toluene solvent system. The reaction is heated to a controlled temperature, typically optimized between 70-75°C, and monitored until completion. Following the reaction, a simple workup involving filtration, aqueous washing, and acidification allows for the direct isolation of the product as a high-purity solid. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Combine 4'-hydroxymethyl-2-cyanobiphenyl (SM-1) and L-valine methyl ester hydrochloride (SM-2) in toluene solvent with a specialized cobalt catalyst (SM-3) and sodium methoxide base.
- Heat the reaction mixture to a controlled temperature range of 60-100°C, specifically optimizing around 70-75°C, until conversion is complete without inert gas protection.
- Filter the reaction mixture, wash the filtrate with purified water, concentrate the organic phase, and treat with concentrated hydrochloric acid to precipitate the pure solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers compelling economic and operational benefits. The elimination of column chromatography and the avoidance of inert gas requirements drastically simplify the manufacturing infrastructure needed for production. This simplification translates directly into reduced capital expenditure and lower operating costs per kilogram of produced intermediate. Furthermore, the ability to isolate the product as a stable solid salt enhances storage stability and simplifies logistics, reducing the risks associated with transporting oily or unstable materials across the global supply chain.
- Cost Reduction in Manufacturing: The significant increase in yield, coupled with the removal of expensive purification steps like column chromatography, drives down the overall cost of goods sold. By avoiding the use of hazardous brominating agents and complex workups, the process also reduces waste disposal costs and raw material consumption. This leaner manufacturing approach allows for more competitive pricing strategies in the highly saturated market for antihypertensive drug intermediates.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance and avoiding production delays. The use of readily available starting materials and common solvents like toluene mitigates the risk of supply disruptions associated with specialty reagents. Consequently, manufacturers can maintain higher inventory turnover rates and respond more agilely to fluctuations in market demand for valsartan.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of heavy metal contaminants in the final product streamline the path to commercial scale-up. The process generates less hazardous waste compared to traditional methods, aligning with increasingly stringent environmental regulations. This environmental compatibility not only reduces regulatory hurdles but also enhances the sustainability profile of the supply chain, a key metric for modern pharmaceutical sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers evaluating this technology for potential adoption or licensing.
Q: How does this new catalytic method improve upon traditional bromomethyl-based synthesis routes?
A: Unlike conventional methods using 2'-cyano-4-bromomethylbiphenyl which often yield oily, hard-to-handle crude products with significant dimer impurities, this novel route utilizes 4'-hydroxymethyl-2-cyanobiphenyl. This shift results in a solid final product with purity exceeding 99.8% and eliminates the need for cumbersome column chromatography purification.
Q: What specific role does the Cobalt catalyst SM-3 play in preventing dimerization?
A: The specialized Cobalt catalyst SM-3 facilitates a highly selective nucleophilic substitution under mild conditions. Its unique ligand environment stabilizes the transition state, effectively suppressing the side reactions that typically lead to the formation of difficult-to-remove dimer impurities common in prior art syntheses.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is explicitly designed for industrial scalability. It operates at moderate temperatures (60-100°C), does not require expensive inert gas protection, and uses readily available solvents like toluene. The isolation of the product as a stable solid salt further simplifies downstream processing and logistics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(2'-cyanobiphenyl-4-methylene)-L-valine methyl ester hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and pure intermediate synthesis in the pharmaceutical value chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN115197096A can be successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for API manufacturing.
We invite you to collaborate with us to leverage this advanced catalytic technology for your valsartan production needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your strategic sourcing decisions.
