Advanced Catalytic Route for High-Purity 1,4,5,6-Tetrahydropyrimidine Derivatives Manufacturing
The pharmaceutical industry continuously seeks robust and efficient synthetic pathways for critical heterocyclic scaffolds, particularly those serving as core structures for bioactive molecules. Patent CN103664875A introduces a transformative methodology for the synthesis of 1,4,5,6-tetrahydropyrimidine derivatives, a class of compounds renowned for their diverse pharmacological activities ranging from antiparasitic to antidepressant properties. This novel approach leverages copper cinnamate as a highly effective catalyst to facilitate the cyclocondensation between substituted nitriles and 1,3-diaminopropane. By addressing the longstanding challenges of harsh reaction conditions and complex purification associated with traditional routes, this technology represents a significant leap forward in process chemistry. For R&D directors and procurement specialists alike, understanding the nuances of this catalytic system is essential for optimizing supply chains and reducing the cost of goods sold for key pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the tetrahydropyrimidine ring has relied heavily on multicomponent reactions involving carboxylic acids, aldehydes, esters, or thioamides reacting with 1,3-diamines. While chemically feasible, these conventional pathways are fraught with significant industrial drawbacks that hinder efficient manufacturing. Traditional catalysts employed in these reactions are often prohibitively expensive precious metal complexes or require stringent anhydrous conditions that escalate operational costs. Furthermore, many legacy processes suffer from poor atom economy and generate substantial quantities of hazardous waste, creating severe environmental compliance burdens. The reaction conditions frequently necessitate extreme temperatures or pressures, which not only pose safety risks but also limit the scope of compatible functional groups, leading to lower overall yields and difficult purification profiles that delay time-to-market for new drug candidates.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a readily accessible copper cinnamate catalyst to drive the reaction between nitriles and 1,3-diaminopropane with exceptional efficiency. This innovative route operates under remarkably mild thermal conditions, typically requiring temperatures between 70°C and 95°C in a common solvent like toluene. The simplicity of the protocol allows for a straightforward workup procedure where the solid catalyst is removed via simple filtration, eliminating the need for complex extraction or heavy metal scavenging steps. This streamlined process not only enhances the purity of the final product but also drastically reduces the consumption of auxiliary materials. By shifting the paradigm from complex multicomponent condensations to a direct nitrile-amine cyclization, manufacturers can achieve higher throughput with significantly reduced environmental footprint and operational complexity.
Mechanistic Insights into Copper Cinnamate-Catalyzed Cyclization
The efficacy of this synthetic route lies in the unique Lewis acidic properties of the copper cinnamate complex, which activates the nitrile group towards nucleophilic attack by the diamine. The copper center coordinates with the nitrogen atom of the nitrile, increasing the electrophilicity of the carbon atom and facilitating the initial addition of the amine. This activation lowers the energy barrier for the subsequent cyclization steps, allowing the reaction to proceed smoothly at moderate temperatures without the need for aggressive activating agents. The cinnamate ligand plays a crucial role in stabilizing the copper species and modulating its reactivity, ensuring that the catalytic cycle turns over efficiently without decomposing into inactive copper oxides or precipitates that would otherwise halt the reaction progress.
From an impurity control perspective, the mild nature of this catalytic system is paramount for maintaining high product quality. Harsh acidic or basic conditions often found in alternative syntheses can lead to the hydrolysis of the nitrile starting material or the polymerization of the diamine, generating difficult-to-remove byproducts. The copper cinnamate system avoids these pitfalls by providing a controlled reaction environment that favors the formation of the desired six-membered ring. Additionally, the ability to recover and reuse the catalyst, as demonstrated in the patent examples, suggests a stable catalytic species that resists degradation over multiple cycles. This stability is critical for commercial operations where batch-to-batch consistency and minimal metal contamination in the final API intermediate are non-negotiable requirements for regulatory approval.
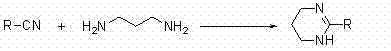
How to Synthesize 1,4,5,6-Tetrahydropyrimidine Derivatives Efficiently
The practical implementation of this synthesis involves a straightforward protocol that balances reaction kinetics with operational simplicity. The process begins with the preparation of the reaction mixture, where the specific substituted nitrile and 1,3-diaminopropane are combined in toluene with the copper cinnamate catalyst and a base such as sodium acetate. The detailed standardized synthesis steps, including precise molar ratios and specific workup parameters, are outlined in the guide below to ensure reproducibility and optimal yield for your specific substrate requirements.
- Prepare the reaction mixture by combining the substituted nitrile substrate and 1,3-diaminopropane in toluene solvent with copper cinnamate catalyst and sodium acetate.
- Heat the mixture to reflux at temperatures between 70°C and 95°C under agitation for a duration of 4 to 6 hours to ensure complete cyclization.
- Filter the reaction mixture to remove the solid catalyst, evaporate the solvent under reduced pressure, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed route offers tangible strategic benefits that extend beyond mere chemical yield. The shift towards a simpler, more robust manufacturing process directly translates to enhanced supply chain reliability and reduced vulnerability to raw material price fluctuations. By utilizing widely available starting materials like substituted benzonitriles and avoiding exotic reagents, companies can secure a more stable supply base. Furthermore, the elimination of expensive transition metal catalysts and the ability to recycle the copper cinnamate significantly lowers the direct material costs associated with production, providing a competitive edge in pricing negotiations with downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic impact of replacing costly catalysts with inexpensive copper cinnamate cannot be overstated in large-scale production scenarios. Since the catalyst can be recovered and reused through simple filtration, the consumption of metal salts per kilogram of product is drastically minimized, leading to substantial cost savings over the lifecycle of the product. Additionally, the use of toluene as a solvent, which is easily recovered and recycled via distillation, further reduces waste disposal costs and solvent purchase expenses. This lean manufacturing approach ensures that the cost of goods sold remains low even when scaling up to multi-ton quantities, making the process highly attractive for generic drug manufacturers seeking margin improvement.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as nitriles and diamines ensures that the supply chain is not dependent on single-source suppliers of specialized reagents. This diversification of raw material sources mitigates the risk of supply disruptions caused by geopolitical issues or production bottlenecks at specific vendor sites. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in utility supplies or environmental conditions, ensuring consistent output regardless of the manufacturing location. This reliability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical partners.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of hazardous reagents and the simplicity of the unit operations involved. The reaction does not require high-pressure equipment or cryogenic cooling, allowing it to be run in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. From an environmental standpoint, the reduced generation of heavy metal waste and the potential for solvent recycling align perfectly with modern green chemistry principles and increasingly strict environmental regulations. This compliance reduces the administrative burden of waste permitting and lowers the long-term liability associated with hazardous waste disposal, safeguarding the company's social license to operate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on the feasibility and advantages of adopting this technology for your specific manufacturing needs. Understanding these details is vital for making informed decisions about process development and technology transfer.
Q: What are the primary advantages of using copper cinnamate over traditional catalysts?
A: Copper cinnamate offers a significant advantage due to its low cost, ease of synthesis, and reusability. Unlike expensive transition metal complexes often used in conventional methods, this catalyst operates under milder conditions (70-95°C) and can be easily removed by simple filtration, reducing downstream processing costs and environmental impact.
Q: What is the typical yield range for this synthetic method?
A: According to the patented data, the method consistently achieves high yields ranging from 60% to 90%, with specific examples demonstrating yields up to 95% for certain substrates like isophthalodinitrile derivatives. This high efficiency minimizes raw material waste and improves overall process economics.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly suitable for scale-up. It utilizes common solvents like toluene, avoids strict anhydrous or inert atmosphere requirements, and features a simple workup procedure involving filtration and evaporation. These factors contribute to robust operational safety and ease of implementation in commercial manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4,5,6-Tetrahydropyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the competitive landscape of pharmaceutical intermediate manufacturing. Our team of expert chemists has extensively evaluated the copper cinnamate catalyzed pathway described in CN103664875A and confirmed its potential for delivering high-purity products at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale industrial output. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,4,5,6-tetrahydropyrimidine derivative meets the exacting standards required for drug substance synthesis.
We invite you to collaborate with us to leverage this advanced technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments that will demonstrate how our optimized manufacturing capabilities can enhance your supply chain efficiency and reduce your overall production costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
