Optimizing Bis(diphenylphosphino)alkane Production for Industrial Catalysis and Pharma Applications
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the need for safer, more efficient, and scalable synthetic routes for critical ligand systems. A pivotal advancement in this domain is detailed in patent CN102633836A, which discloses a robust method for synthesizing bis(diphenylphosphino)-alkane compounds. This technology represents a paradigm shift from traditional methodologies that rely on hazardous alkali metals, offering a streamlined pathway that utilizes lithium hydride as a key reagent under anhydrous and anoxybiotic conditions. For R&D directors and procurement specialists seeking a reliable catalyst ligand supplier, understanding the nuances of this innovation is crucial. The process involves enabling triphenylphosphine to react with lithium hydride to generate a lithium diphenylphosphide intermediate, which then directly reacts with dibromoalkane. This approach not only optimizes the reaction process but also ensures that the intermediate generated during the preparation has no adverse effects on the follow-up reaction, thereby enhancing the overall feasibility of the工艺 structure for commercial deployment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bis(diphenylphosphino)alkane compounds has been plagued by significant operational challenges and safety concerns inherent to conventional methodologies. Prior art, such as USP 4,618,720, describes methods utilizing diphenyl phosphine chloride and excess base sodium metal under inert solvent conditions. While chemically viable on a small scale, these traditional routes suffer from lower yields, particularly when preparing short-chain variants of the target molecule. Furthermore, the handling of unreacted basic metal poses severe safety risks and complicates the recovery and recycling processes, making the method relatively difficult to implement in actual industrial production environments. Other domestic literature reports have attempted to accelerate reaction speeds using ultrasonic irradiation with metal lithium sheets; however, these methods introduce new variables such as the wayward addition of tert-butyl chloride. Insufficient or excessive addition in these older protocols leads to the consumption of the system or the formation of unwanted by-products through side reactions with phenyl lithium, ultimately compromising the purity and economic viability of the final output.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach outlined in the reference patent leverages the unique reactivity of lithium hydride to overcome the bottlenecks of cost and control. By starting with triphenylphosphine and processing it with lithium hydride to prepare diphenylphosphine lithium, the method creates a highly reactive intermediate that can be directly utilized without tedious isolation steps. This direct reaction with dibromoalkane synthesizes the bis(diphenylphosphino)-alkane compound with remarkable efficiency. The optimization of the reaction process makes the entire sequence easier to control, significantly reducing the complexity of separation and purification stages. Consequently, the separation cost is reduced, and the method is deemed far more suitable for industrial production. This shift allows for cost reduction in fine chemical manufacturing by minimizing waste streams and maximizing the throughput of high-value ligand precursors essential for homogeneous catalysis in organic synthesis fields.
Mechanistic Insights into LiH-Mediated Phosphination
The core of this technological breakthrough lies in the mechanistic elegance of the lithium hydride-mediated phosphination. Under noble gas protection, typically argon, triphenylphosphine is dissolved in dry tetrahydrofuran (THF) and cooled via a water bath. The subsequent addition of the lithium hydride solution initiates the formation of the lithium diphenylphosphide species. This step is critical because the intermediate generated does not interfere with subsequent reactions, a common pitfall in other metallation strategies. The reaction mixture is stirred at room temperature overnight, ensuring complete conversion, which is rigorously monitored using High-Performance Liquid Chromatography (HPLC) to confirm the absence of residual triphenylphosphine. Once the intermediate is fully formed, the dibromoalkane is introduced, and the temperature is raised to reflux. This thermal activation drives the nucleophilic substitution, linking the phosphine units via the alkane chain. The precision of this mechanism ensures that the structural integrity of the ligand is maintained, providing the high-purity organophosphorus compounds required for sensitive catalytic applications.
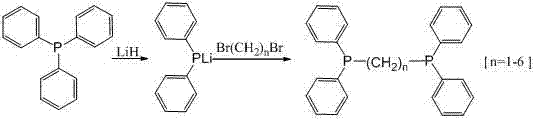
Following the reaction completion, the workup procedure is designed to maximize recovery while ensuring stringent purity specifications. The THF solvent is reclaimed under reduced pressure, leaving behind a sticky solid residue. This crude product undergoes a sequential washing process involving hydrochloric acid, water, and methanol to remove inorganic salts and unreacted starting materials. Finally, recrystallization from a chloroform and methanol mixed solvent yields the target bis(diphenylphosphino)alkane compounds as off-white solids. Examples from the patent data demonstrate the versatility of this mechanism across different chain lengths, from methylene bromide to 1,6-dibromo-hexane, consistently achieving contents of 98% by HPLC. This level of impurity control is vital for R&D teams focusing on the reproducibility of linked reactions in the synthesis of natural products, functional materials, and bioactive compounds, where trace impurities can poison expensive transition metal catalysts.
How to Synthesize Bis(diphenylphosphino)alkane Efficiently
Implementing this synthesis route requires strict adherence to anhydrous conditions and precise stoichiometric control to replicate the high yields reported in the patent literature. The process begins with the preparation of the reactor under an inert atmosphere, followed by the careful addition of solvents and reagents to manage the exothermic nature of the hydride reaction. Operators must monitor the reaction progress closely, utilizing HPLC detection to verify the complete consumption of triphenylphosphine before introducing the alkylating agent. This attention to detail ensures that the subsequent coupling reaction proceeds without competition from unreacted phosphine, which could lead to mono-substituted by-products. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational parameters.
- Under inert gas protection, react triphenylphosphine with lithium hydride in anhydrous THF to generate the lithium diphenylphosphide intermediate.
- Monitor the reaction via HPLC to ensure complete consumption of triphenylphosphine before proceeding to the next stage.
- Add dibromoalkane to the intermediate solution, reflux, and purify the final product through acid washing and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this LiH-mediated synthesis route offers substantial strategic benefits beyond mere chemical yield. The primary advantage lies in the drastic simplification of the raw material handling profile. By eliminating the need for pyrophoric sodium metal or complex ultrasonic setups involving metal lithium sheets, the process significantly reduces the safety infrastructure costs and insurance liabilities associated with manufacturing. This translates directly into cost reduction in manufacturing, as the facility requirements are less stringent, and the operational risk is lowered. Furthermore, the ability to reclaim solvents like THF under reduced pressure contributes to a more sustainable and economically efficient production cycle, aligning with modern green chemistry initiatives that many multinational corporations now mandate for their suppliers.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like sodium metal or the complex additives required for ultrasonic methods leads to a streamlined cost structure. The process avoids the need for specialized equipment to handle unreacted basic metals, which are difficult to reclaim and dispose of safely. Additionally, the high selectivity of the reaction minimizes the formation of by-products, reducing the load on downstream purification units and lowering the consumption of recrystallization solvents. These factors combine to create a leaner manufacturing process that offers substantial cost savings without compromising on the quality of the final ligand product.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method enhances supply chain reliability by reducing the dependency on hard-to-source or highly regulated reagents. Lithium hydride and triphenylphosphine are commercially available commodities with stable supply lines, unlike some specialized organometallic reagents that may face geopolitical or logistical bottlenecks. The simplicity of the reaction control also means that production schedules are more predictable, with fewer delays caused by failed batches or complex troubleshooting. This stability is crucial for reducing lead time for high-purity intermediates, ensuring that downstream pharmaceutical and agrochemical production lines remain uninterrupted.
- Scalability and Environmental Compliance: From a scalability perspective, the method is explicitly designed for suitability for industrialized production, addressing the limitations of lab-scale techniques that fail upon expansion. The washing and recrystallization steps are easily adaptable to large-scale filtration and drying equipment, facilitating the commercial scale-up of complex ligands. Moreover, the reduced generation of hazardous metallic waste simplifies environmental compliance and wastewater treatment processes. This alignment with environmental regulations ensures long-term operational continuity and mitigates the risk of regulatory shutdowns, making it a preferred choice for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the comparative data and beneficial effects outlined in the patent documentation, providing clarity on why this method is superior to legacy approaches. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing supply chains and R&D pipelines.
Q: Why is Lithium Hydride preferred over Sodium Metal for this synthesis?
A: Lithium Hydride eliminates the safety hazards associated with handling pyrophoric sodium metal and simplifies the post-reaction workup by avoiding difficult-to-remove metallic residues.
Q: What yields can be expected from this novel LiH-mediated pathway?
A: The patented method demonstrates consistently high yields, often exceeding 90%, with product purity reaching 98% as confirmed by HPLC analysis across various chain lengths.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process optimizes reaction control and reduces separation costs, making it significantly more suitable for industrial production compared to traditional ultrasonic or sodium-based methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis(diphenylphosphino)alkane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality ligands play in the success of modern catalytic processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to full-scale manufacturing is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of bis(diphenylphosphino)alkane meets the exacting standards required by the global pharmaceutical and fine chemical industries. Our infrastructure is designed to support the complex needs of clients who demand consistency, safety, and technical excellence in their raw material partners.
We invite you to collaborate with us to leverage these advanced synthetic capabilities for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce your overall production costs.
