Scalable Synthesis of Advanced UV-Absorbing Intermediates for High-Performance Cosmetics
The landscape of high-performance cosmetic ingredients is constantly evolving, driven by the demand for superior UV protection that does not compromise formulation stability or user experience. A significant breakthrough in this domain is detailed in patent CN115260127A, which discloses a highly efficient preparation method for octyl 5-pyrrolidinyl-2-phenylsulfonyl-2,4-pentadienoate. This compound serves as a critical functional ingredient, offering broad-spectrum ultraviolet absorption capabilities specifically tailored for keratin care and non-therapeutic cosmetic applications. Unlike traditional UV absorbers that struggle with solubility in fatty solvents or lack stability, this novel molecule demonstrates exceptional chemical and photostability, capable of dissolving completely in aqueous phases either molecularly or colloidally. The patent outlines a sophisticated synthetic route that overcomes previous limitations by introducing a unique salt metathesis strategy, fundamentally altering the reactivity profile of key intermediates to achieve superior yields and purity.
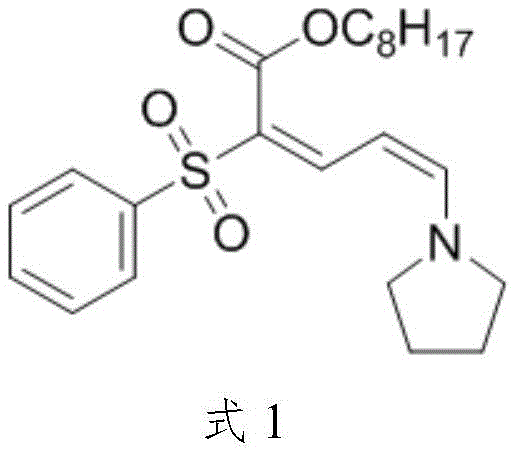
For procurement specialists and supply chain managers evaluating reliable functional active ingredients supplier options, understanding the technical nuances of this synthesis is paramount. The method described ensures that raw materials are easily accessible and reaction conditions remain mild, translating directly into economic practicability and reduced operational risk. By shifting from conventional acetate salts to tetrafluoroborate salts in the intermediate stage, the process enhances the electrophilicity of the reacting species, thereby streamlining the production workflow. This technical advancement lays a robust scientific foundation for the large-scale batch production of this high-value UV absorber, addressing the growing market need for effective sun protection agents that maintain the aesthetic quality of cosmetic products.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for similar conjugated diene structures often rely on less reactive intermediates that necessitate harsh reaction conditions to drive the coupling reactions to completion. In comparative studies, such as those referenced in the background of the patent, utilizing acetate salts as the leaving group or activating moiety frequently results in sluggish reaction kinetics. This inefficiency forces manufacturers to employ higher temperatures, extended reaction times, or excessive amounts of expensive catalysts to achieve acceptable conversion rates. Furthermore, the lower electrophilicity of acetate-based intermediates often leads to incomplete reactions, generating complex impurity profiles that are difficult and costly to remove during downstream purification. These factors collectively contribute to higher production costs, increased waste generation, and inconsistent batch-to-batch quality, posing significant challenges for cost reduction in personal care chemical manufacturing.
The Novel Approach
The innovative methodology presented in the patent data revolutionizes this process by substituting the acetate group with a tetrafluoroborate group in the critical intermediate stage. This strategic modification dramatically increases the electrophilicity of the N,N'-bis(1,4-butene)-1,5-1,5-diazapentadiene moiety. As a result, the subsequent electrophilic substitution reaction with 2-(benzenesulfonyl) octyl acetate can proceed under significantly milder conditions, typically ranging between 20°C and 40°C. This shift not only drastically shortens the required reaction time but also improves the overall synthesis efficiency and molar yield. By optimizing the electronic properties of the intermediate, the new approach minimizes side reactions and simplifies the workup procedure, establishing a more robust and economically viable pathway for the commercial scale-up of complex cosmetic intermediates.
Mechanistic Insights into Tetrafluoroborate-Mediated Electrophilic Substitution
The core mechanistic advantage of this synthesis lies in the enhanced reactivity imparted by the tetrafluoroborate anion ($BF_4^-$). In the second step of the process, the condensation of tetramethoxypropane and pyrrolidine initially yields an acetate salt. However, the subsequent displacement reaction with sodium tetrafluoroborate replaces the acetate anion, creating a more stable yet highly reactive tetrafluoroborate salt. The tetrafluoroborate group is a weakly coordinating anion that stabilizes the cationic character of the diazapentadiene system without hindering its nucleophilic attack capabilities in the reverse sense, or rather, it makes the adjacent carbons more susceptible to nucleophilic attack by the enolate formed in the next step. This increased electrophilicity is crucial for the success of Step S3, where the intermediate reacts with the sulfone ester.
Furthermore, the impurity control mechanism is inherently built into this high-selectivity pathway. The use of DBU (1,8-diazabicyclo[5.4.0]undec-7-ene) as a base in the final coupling step facilitates the rapid formation of the enolate from the sulfone ester, which then attacks the activated diazapentadiene salt. Because the reaction proceeds efficiently at lower temperatures, thermal degradation pathways and polymerization side reactions—common issues with conjugated dienes—are effectively suppressed. The final purification step, involving activated carbon treatment followed by controlled crystallization with n-hexane, leverages the high initial purity of the crude product to deliver a final substance with purity levels exceeding 99.6%. This rigorous control over the reaction environment ensures that the resulting high-purity UV absorber meets the stringent quality standards required for topical application.
How to Synthesize Octyl 5-Pyrrolidinyl-2-Phenylsulfonyl-2,4-Pentadienoate Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step guide for reproducing this high-value intermediate with consistent quality. The process begins with the preparation of the sulfone ester backbone, followed by the activation of the amine component, and concludes with the coupling and purification stages. Each step has been optimized to balance reaction rate with selectivity, ensuring that the process is not only chemically sound but also operationally simple for industrial implementation. The detailed standardized synthesis steps below provide the necessary technical framework for R&D teams to evaluate feasibility and initiate pilot trials.
- Prepare 2-(benzenesulfonyl) octyl acetate via nucleophilic substitution of 2-chloroacetic acid octyl ester with sodium benzenesulfinate in DMF at mild temperatures.
- Synthesize the key intermediate N,N'-bis(1,4-butene)-1,5-1,5-diazapentadiene tetrafluoroborate sodium salt by displacing acetate with tetrafluoroborate to increase electrophilicity.
- Execute the final electrophilic substitution reaction between the sulfone ester and the activated diazapentadiene salt using DBU catalyst, followed by recrystallization for high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers substantial advantages for organizations seeking to optimize their supply chain for cosmetic active ingredients. The primary benefit stems from the significant improvement in process efficiency, which directly correlates to reduced manufacturing costs and enhanced supply reliability. By eliminating the need for harsh reaction conditions and minimizing the formation of difficult-to-remove impurities, the process reduces the burden on downstream purification units. This streamlined workflow translates into shorter production cycles and lower energy consumption, providing a competitive edge in the fast-moving personal care market.
- Cost Reduction in Manufacturing: The transition to a tetrafluoroborate-mediated pathway eliminates the inefficiencies associated with traditional acetate-based methods. By achieving higher molar yields and reducing reaction times, the overall consumption of raw materials and solvents per kilogram of product is significantly decreased. Additionally, the ability to operate at near-ambient temperatures reduces the energy load required for heating and cooling, leading to substantial cost savings in utility expenses. The simplified purification process further lowers costs by reducing the volume of waste solvents and the need for extensive chromatographic separation.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, such as 2-chloroacetic acid octyl ester, sodium benzenesulfinate, and pyrrolidine, are commodity chemicals that are readily available from multiple global suppliers. This abundance mitigates the risk of supply disruptions that often plague processes relying on exotic or proprietary reagents. Furthermore, the robustness of the reaction conditions ensures high batch-to-batch consistency, allowing supply chain managers to forecast production timelines with greater accuracy and reduce lead time for high-purity cosmetic intermediates.
- Scalability and Environmental Compliance: The mild operating conditions and high selectivity of this process make it inherently safer and easier to scale from laboratory to commercial production. The reduction in hazardous waste generation and the use of standard solvents like ethanol and ethyl acetate align well with modern environmental, health, and safety (EHS) regulations. This compliance facilitates smoother regulatory approvals and supports sustainability goals, making the process attractive for long-term partnerships focused on green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this advanced UV absorber. These insights are derived directly from the patented technology and are intended to assist decision-makers in evaluating the suitability of this ingredient for their specific formulation needs. Understanding these details is crucial for integrating this high-performance molecule into next-generation skincare products.
Q: Why is the tetrafluoroborate salt preferred over the acetate salt in this synthesis?
A: The tetrafluoroborate anion significantly increases the electrophilicity of the diazapentadiene group compared to the acetate anion. This enhancement allows the subsequent electrophilic substitution reaction to proceed under much milder conditions with higher conversion rates and molar yields, reducing reaction time and energy consumption.
Q: What is the expected purity level of the final UV absorber intermediate?
A: Through the optimized purification process involving activated carbon decolorization and controlled crystallization with n-hexane, the final product achieves a purity exceeding 99.6%, ensuring excellent chemical and photostability for cosmetic formulations.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process utilizes readily available raw materials and operates at relatively low temperatures (20-40°C for key steps). The simplified operational steps and high molar yields make it highly economically practical and scalable for batch production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Octyl 5-Pyrrolidinyl-2-Phenylsulfonyl-2,4-Pentadienoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthesis route in the realm of advanced cosmetic ingredients. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of octyl 5-pyrrolidinyl-2-phenylsulfonyl-2,4-pentadienoate meets the highest industry standards for safety and efficacy.
We invite you to collaborate with us to optimize your supply chain and bring superior UV protection solutions to market faster. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can support your product development goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
