Advanced Electrochemical Manufacturing of 2-Carboxy-5-Nitrobenzenesulfonic Acid for Global Agrochemical Supply Chains
Advanced Electrochemical Manufacturing of 2-Carboxy-5-Nitrobenzenesulfonic Acid for Global Agrochemical Supply Chains
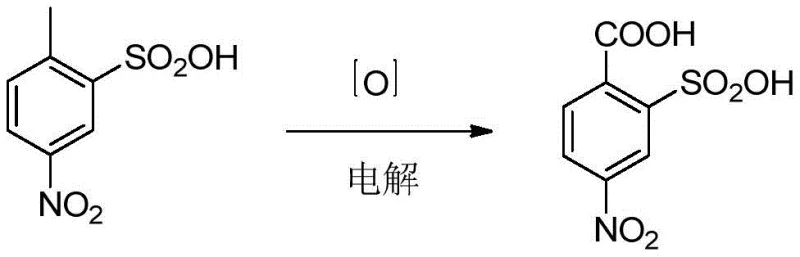
The global demand for high-performance herbicides continues to drive innovation in the synthesis of critical fine chemical intermediates, specifically 2-carboxy-5-nitrobenzenesulfonic acid (CAS 22952-26-7), a pivotal precursor for the widely used herbicide foramsulfuron. Recent intellectual property developments, notably patent CN113755867A filed in December 2021, have introduced a transformative electrochemical synthesis method that addresses long-standing inefficiencies in traditional manufacturing. This groundbreaking technology replaces hazardous chemical oxidants with electrons as the primary reagent, utilizing a continuous flow electrolytic system to achieve superior selectivity and environmental compliance. For R&D directors and procurement strategists, this shift represents a significant opportunity to optimize supply chains for reliable agrochemical intermediate supplier networks while drastically reducing the ecological footprint associated with nitro-aromatic oxidation processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 2-carboxy-5-nitrobenzenesulfonic acid has relied heavily on aggressive chemical oxidation protocols that pose severe economic and environmental challenges. Traditional routes often employ stoichiometric amounts of heavy metal salts, such as chromium or manganese oxidants, which generate substantial quantities of toxic metal-containing wastewater that requires complex and costly remediation before discharge. Alternative methods utilizing nitric acid as an oxidant in glacial acetic acid solvents present acute safety hazards due to the evolution of nitrogen oxides (NOx) and the risk of runaway exothermic reactions under high-temperature conditions. Furthermore, oxidation using hypochlorite has been documented to suffer from poor selectivity and low efficiency, making it unsuitable for the rigorous purity standards required in modern pharmaceutical and agrochemical applications. These legacy processes not only inflate the cost reduction in fine chemical manufacturing but also create significant supply chain vulnerabilities due to increasingly stringent environmental regulations on heavy metal and acidic waste disposal.
The Novel Approach
In stark contrast to these archaic methods, the electrochemical synthesis disclosed in patent CN113755867A offers a streamlined, green alternative that fundamentally alters the reaction thermodynamics and kinetics. By employing a diaphragm-free single-chamber electrolytic cell, the process utilizes sulfuric acid as a supporting electrolyte to create a strongly acidic environment where the substrate, p-nitrotoluene-o-sulfonic acid, undergoes direct anodic oxidation. This approach completely circumvents the need for external chemical oxidants or transition metal catalysts, thereby eliminating the generation of heavy metal sludge and simplifying the downstream purification workflow. The system operates under mild conditions, typically between 10-60 °C and at atmospheric pressure, which significantly reduces energy consumption and mitigates the safety risks associated with high-pressure reactors. Moreover, the implementation of a continuous feeding and continuous extraction mode ensures a steady-state operation that is inherently scalable, providing a robust foundation for commercial scale-up of complex agrochemical intermediates without the batch-to-batch variability seen in traditional kettle reactions.
Mechanistic Insights into Electrochemical Oxidation of Nitro-Aromatics
The core of this technological advancement lies in the precise control of electrode potential within the electrolytic cell, which dictates the selectivity of the methyl group oxidation to the carboxylic acid functionality. The patent specifies the use of anodes with high oxygen evolution overpotential, such as titanium-based lead dioxide (PbO2/Ti), pure lead, or titanium-based platinum, paired with cathodes like titanium, nickel, or lead to minimize side reactions. The reaction proceeds through the loss of electrons at the anode surface, where the methyl group of the p-nitrotoluene-o-sulfonic acid is sequentially oxidized, bypassing the formation of unstable aldehyde intermediates that often plague chemical oxidation routes. The presence of the strong electron-withdrawing nitro and sulfonic acid groups on the aromatic ring typically deactivates the ring towards oxidation, necessitating the high energy input provided by the controlled current density of 200-3000 A.m-2. This electrochemical driving force ensures that the reaction proceeds efficiently even with these deactivated substrates, achieving yields that consistently exceed 90% in optimized examples.
Impurity control is another critical aspect where this electrochemical mechanism excels, particularly for R&D teams focused on high-purity API intermediate specifications. By tuning the current density and maintaining the electrolyte composition within specific parameters—such as a p-nitrotoluene-o-sulfonic acid concentration of 5-30% in organic solvents like acetonitrile or acetone—the process minimizes over-oxidation or ring degradation. The patent data indicates that product purity can reach greater than 95%, with specific experimental runs demonstrating purity levels as high as 98.5% via liquid chromatography analysis. This high level of chemical fidelity is achieved because the electrode surface acts as a selective catalyst, where the potential is set just high enough to drive the desired methyl oxidation but low enough to prevent the degradation of the sensitive nitro group or the sulfonic acid moiety. Consequently, the resulting crude product requires less intensive purification, reducing the overall solvent usage and processing time required to meet the stringent quality standards of global agrochemical manufacturers.
How to Synthesize 2-Carboxy-5-Nitrobenzenesulfonic Acid Efficiently
To implement this cutting-edge synthesis route effectively, manufacturers must adhere to a precise protocol that balances electrolyte composition, electrode material selection, and hydrodynamic conditions within the reactor. The process begins with the preparation of a homogeneous electrolyte solution, where the organic substrate is dissolved in a compatible solvent and mixed with a diluted sulfuric acid solution to ensure adequate conductivity and proton availability. Following this, the solution is pumped through the electrolytic cell under a constant current regime, where the residence time and flow rate are critical parameters for maximizing conversion while minimizing energy waste. This structured approach ensures that the electrochemical oxidation proceeds with maximum efficiency, translating laboratory-scale success into reliable industrial production capabilities.
- Prepare electrolyte by mixing p-nitrotoluene-o-sulfonic acid in organic solvent (5-30% mass) with diluted sulfuric acid (1: 1 to 1:6 ratio).
- Perform constant current electrolysis in a diaphragm-free single-chamber cell at 200-3000 A.m-2 and 10-60°C using continuous feed mode.
- Extract the product using organic solvents like dichloroethane, followed by distillation and recrystallization to achieve >95% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical technology translates into tangible strategic benefits that extend far beyond simple chemical yield improvements. The elimination of expensive and regulated heavy metal oxidants removes a significant variable cost component from the manufacturing budget, while simultaneously alleviating the regulatory burden associated with hazardous waste disposal. The ability to recycle the aqueous phase after extraction further enhances the sustainability profile of the operation, aligning with the increasing corporate mandates for green chemistry and carbon footprint reduction across the specialty chemical sector. Furthermore, the continuous nature of the process allows for a more predictable production schedule, reducing the lead time for high-purity agrochemical intermediates and ensuring a steady flow of materials to downstream formulation plants.
- Cost Reduction in Manufacturing: The economic model of this electrochemical process is fundamentally superior to traditional methods due to the substitution of costly chemical reagents with electricity, which is often a cheaper and more controllable input. By avoiding the purchase of stoichiometric oxidants like potassium permanganate or chromic acid, manufacturers can achieve substantial cost savings on raw materials, while the simplified workup procedure reduces labor and utility costs associated with waste neutralization and treatment. Additionally, the longevity of the electrode materials and the simplicity of the diaphragm-free cell design lower the capital expenditure required for reactor maintenance and replacement, contributing to a lower total cost of ownership over the lifecycle of the production facility.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the continuous flow nature of this synthesis, which allows for on-demand production scaling without the long turnaround times typical of batch processing. The reliance on commodity chemicals like sulfuric acid and common organic solvents, rather than specialized oxidants that may face supply shortages, ensures a more robust and disruption-resistant supply line. This stability is crucial for maintaining the continuity of herbicide production, particularly during peak agricultural seasons when demand for foramsulfuron and related products surges globally.
- Scalability and Environmental Compliance: From an environmental compliance perspective, this technology offers a clear pathway to meeting increasingly strict international regulations on industrial effluents. The absence of heavy metal residues and nitrogen oxide emissions simplifies the permitting process for new production lines and reduces the risk of fines or shutdowns due to environmental violations. The process is inherently scalable, as increasing production capacity primarily involves adding more electrolytic cells in parallel or increasing the electrode surface area, rather than redesigning complex reaction vessels, making it an ideal solution for expanding manufacturing footprints in a sustainable manner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of 2-carboxy-5-nitrobenzenesulfonic acid, providing clarity for stakeholders evaluating this technology for integration into their supply chains. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is essential for leveraging the full potential of this green manufacturing route.
Q: What are the primary advantages of the electrochemical method over traditional chemical oxidation?
A: The electrochemical method eliminates the need for expensive and hazardous heavy metal oxidants or nitric acid, significantly reducing three-waste treatment costs and improving operational safety by avoiding high temperature and pressure conditions.
Q: What purity levels can be achieved with this synthesis route?
A: By precisely controlling electrode potential and current density, the process achieves product purity exceeding 95%, with specific examples demonstrating purity levels up to 98.5% as confirmed by liquid chromatography.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent specifies a continuous feeding and extraction mode which is inherently designed for industrial scale-up, allowing for consistent output and the recycling of the aqueous phase to meet green manufacturing standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Carboxy-5-Nitrobenzenesulfonic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global agrochemical industry, and we are committed to delivering 2-carboxy-5-nitrobenzenesulfonic acid that meets the most rigorous performance standards. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch conforms to the required chemical identity and impurity profiles, giving you confidence in the reliability of your raw material supply.
We invite you to collaborate with us to explore how this advanced electrochemical technology can optimize your specific manufacturing needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current production volumes. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can enhance your supply chain efficiency and support your long-term strategic goals in the agrochemical sector.
