Advanced Continuous Synthesis of p-Aminophenyl-Beta-Hydroxyethyl Sulfone Sulfate for Industrial Scale
Advanced Continuous Synthesis of p-Aminophenyl-Beta-Hydroxyethyl Sulfone Sulfate for Industrial Scale
The global demand for high-performance reactive dyes and specialized agrochemical intermediates necessitates a paradigm shift in manufacturing methodologies, moving away from batch-dependent, waste-intensive processes toward continuous, catalytic precision. Patent CN114369044A delineates a groundbreaking preparation method for p-aminophenyl-beta-hydroxyethyl sulfone and its sulfate derivative, addressing critical bottlenecks in selectivity and cost-efficiency. This technical disclosure introduces a proprietary modified skeletal nickel catalyst that effectively replaces expensive palladium-on-carbon systems, thereby mitigating sulfur poisoning risks that have historically plagued this specific chemical transformation. By integrating a continuous kneading esterification technique with optimized hydrogenation parameters, the process achieves yields exceeding 94% while substantially lowering the environmental burden associated with traditional sulfonation and reduction pathways. For procurement leaders and R&D directors, this represents a viable route to secure a reliable reactive dye intermediate supplier capable of delivering consistent quality at a fraction of the operational expenditure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of p-aminophenyl-beta-hydroxyethyl sulfone sulfate has relied on a cumbersome multi-step sequence involving p-acetanilide, chlorosulfonic acid, and ethylene oxide, a route fraught with significant inefficiencies and safety hazards. The traditional sulfonation procedure requires excessive chlorosulfonic acid and cryogenic conditions below -10°C to manage exothermic reactions, generating massive quantities of hazardous hydrogen chloride gas and waste hydrochloric acid that demand complex four-stage absorption systems. Furthermore, the subsequent reduction and condensation steps utilizing sodium pyrosulfite and ethylene oxide are typically conducted in batch reactors, leading to inconsistent thermal profiles and poor control over side reactions, ultimately capping yields at approximately 70%. The reliance on intermittent feeding production not only limits throughput capacity but also results in fluctuating product quality, while the generation of high-salinity wastewater and waste sulfuric acid creates a substantial liability for environmental compliance teams seeking to minimize their ecological footprint.
The Novel Approach
In stark contrast, the methodology disclosed in CN114369044A leverages a streamlined continuous production architecture that begins with the condensation of p-nitrochlorobenzene and mercaptoethanol, followed by a controlled oxidation to the sulfone prior to the critical hydrogenation step. 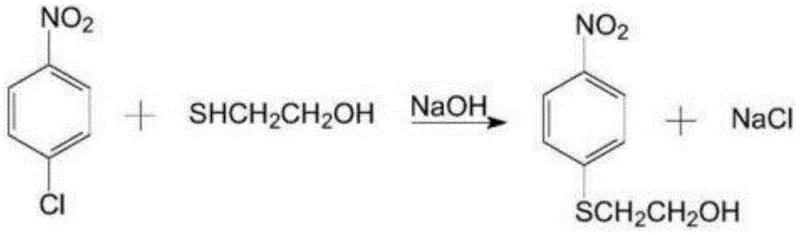
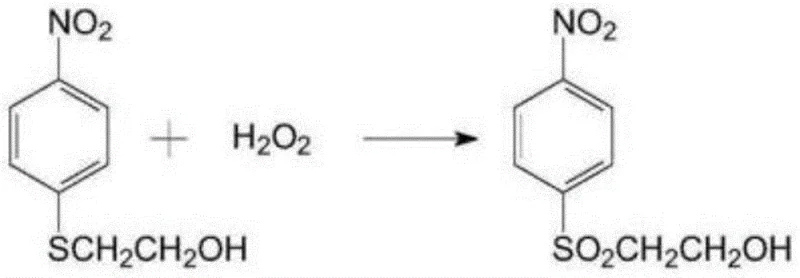 This novel route eliminates the need for hazardous chlorosulfonic acid sulfonation entirely, replacing it with a cleaner oxidation process using hydrogen peroxide and a tungstate catalyst that generates water as the primary byproduct. The transition to a continuous flow system for the esterification stage, utilizing a specialized kneader under vacuum conditions rather than a traditional pot, ensures precise temperature control between 80-160°C, preventing thermal degradation and enhancing the stability of the final sulfate ester. This architectural overhaul not only simplifies the process flow but also drastically reduces raw material consumption and waste generation, positioning it as a superior choice for cost reduction in fine chemical intermediate manufacturing.
This novel route eliminates the need for hazardous chlorosulfonic acid sulfonation entirely, replacing it with a cleaner oxidation process using hydrogen peroxide and a tungstate catalyst that generates water as the primary byproduct. The transition to a continuous flow system for the esterification stage, utilizing a specialized kneader under vacuum conditions rather than a traditional pot, ensures precise temperature control between 80-160°C, preventing thermal degradation and enhancing the stability of the final sulfate ester. This architectural overhaul not only simplifies the process flow but also drastically reduces raw material consumption and waste generation, positioning it as a superior choice for cost reduction in fine chemical intermediate manufacturing.
Mechanistic Insights into Modified Skeletal Nickel Catalyzed Hydrogenation
The cornerstone of this technological advancement lies in the deployment of a modified skeletal nickel catalyst, engineered with a specific composition of 75-95 parts Nickel, 3-15 parts Aluminum, and 2-10 parts metal additives such as Molybdenum, Copper, or Chromium. Unlike conventional palladium carbon catalysts which are highly susceptible to deactivation via sulfur poisoning—a common occurrence when reducing nitro groups in the presence of sulfone moieties—this modified alloy exhibits exceptional tolerance and longevity. The inclusion of metal promoters like Mo or Cu creates a synergistic electronic effect on the nickel surface, optimizing the adsorption energy of hydrogen and the nitro substrate while repelling sulfur-containing impurities that would otherwise block active sites. 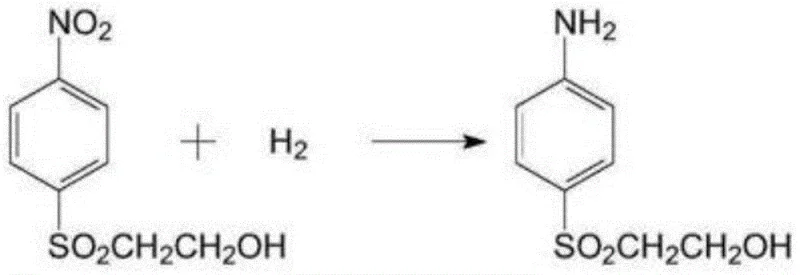 Operational parameters are tightly controlled, with hydrogenation occurring at 85-110°C under 1.5-4 MPa pressure, ensuring rapid conversion kinetics without compromising the integrity of the hydroxyethyl side chain. The rapid cooling rate during catalyst preparation, specified at 0.1-1 × 10^6 K/s, further refines the grain structure, preventing segregation and ensuring a uniform distribution of active phases that contribute to the observed selectivity improvements.
Operational parameters are tightly controlled, with hydrogenation occurring at 85-110°C under 1.5-4 MPa pressure, ensuring rapid conversion kinetics without compromising the integrity of the hydroxyethyl side chain. The rapid cooling rate during catalyst preparation, specified at 0.1-1 × 10^6 K/s, further refines the grain structure, preventing segregation and ensuring a uniform distribution of active phases that contribute to the observed selectivity improvements.
Impurity control is rigorously maintained through the integration of continuous counter-current washing and methanol crystallization steps immediately following the oxidation phase, which removes residual tungstate catalysts and unreacted sulfides before they enter the hydrogenation reactor. This pre-purification is vital because even trace amounts of oxidizing agents or sulfur impurities could degrade the performance of the skeletal nickel or lead to the formation of azo-byproducts. The subsequent separation of the catalyst from the reduction solution is facilitated by the robust physical structure of the alloy particles, which resist attrition under the high-shear stirring conditions of 200-1000 r/min. By eliminating the need for expensive noble metal scavengers and complex filtration setups required for palladium recovery, the process inherently simplifies the downstream purification train, resulting in a final product with a purity profile that meets the stringent specifications demanded by high-end textile and pharmaceutical applications.
How to Synthesize p-Aminophenyl-Beta-Hydroxyethyl Sulfone Efficiently
The synthesis protocol outlined in the patent provides a robust framework for scaling this chemistry from laboratory benchtop to multi-ton commercial production, emphasizing the importance of precise stoichiometric control and thermal management throughout the four distinct reaction stages. Operators must adhere strictly to the specified molar ratios, particularly during the initial condensation where a slight excess of mercaptoethanol ensures complete conversion of the chloronitrobenzene, and during the final esterification where the ratio of amine to sulfuric acid dictates the completeness of the sulfate formation. The transition from batch to continuous processing requires calibrated flow meters for reagent delivery and high-efficiency mixers to maintain homogeneity, especially during the exothermic oxidation phase where temperature spikes must be avoided to prevent runaway reactions. Detailed standardized operating procedures for catalyst activation, reactor pressurization, and vacuum kneading are essential to replicate the high yields and selectivity reported in the intellectual property documentation.
- Condense p-nitrochlorobenzene with mercaptoethanol using NaOH to form p-nitrophenyl-beta-hydroxyethyl sulfide.
- Oxidize the sulfide to p-nitrophenyl-beta-hydroxyethyl sulfone using hydrogen peroxide and a tungstate catalyst.
- Perform catalytic hydrogenation using modified skeletal nickel (Ni-Al-Mo/Cu/Cr) at 85-110°C and 1.5-4 MPa to obtain the amine.
- React the amine with concentrated sulfuric acid in a kneader under vacuum to produce the final sulfate ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates directly into enhanced operational resilience and significant structural cost advantages that extend beyond simple raw material pricing. The elimination of palladium carbon, a volatile commodity subject to geopolitical supply constraints and price fluctuations, in favor of a base metal nickel alloy stabilizes the bill of materials and insulates the production budget from precious metal market volatility. Furthermore, the continuous nature of the process allows for a smaller physical footprint and higher throughput per unit of time, effectively increasing asset utilization rates and reducing the capital expenditure required per kilogram of output. The reduction in hazardous waste streams, specifically the avoidance of large volumes of waste hydrochloric and sulfuric acids, lowers the overhead costs associated with waste treatment, disposal, and regulatory compliance, contributing to a more sustainable and economically viable manufacturing model.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts with modified skeletal nickel fundamentally alters the cost structure of the hydrogenation step, removing the need for costly catalyst recovery and regeneration units. By utilizing a continuous kneading process for esterification instead of traditional batch pots, energy consumption is optimized through better heat transfer efficiency, and labor costs are reduced due to the automated nature of the screw discharge system. The overall process design minimizes solvent usage and maximizes atom economy, ensuring that a higher proportion of input raw materials are converted into saleable product rather than lost as byproducts or waste.
- Enhanced Supply Chain Reliability: The reliance on widely available base metals like nickel and aluminum, rather than scarce platinum group metals, ensures a stable and secure supply of critical catalytic materials, reducing the risk of production stoppages due to vendor shortages. The continuous production capability allows for flexible scheduling and just-in-time manufacturing, enabling suppliers to respond more rapidly to fluctuations in market demand without the long lead times associated with cleaning and setting up large batch reactors. This agility is crucial for maintaining service levels to downstream customers in the reactive dye and agrochemical sectors who require consistent delivery schedules to manage their own inventory levels.
- Scalability and Environmental Compliance: The process is inherently scalable, as the continuous flow reactors and kneaders can be sized or numbered up to meet increasing volume requirements without the engineering challenges typical of scaling batch exothermic reactions. The significant reduction in the generation of high-salinity wastewater and corrosive waste acids simplifies the effluent treatment process, making it easier to meet increasingly strict environmental regulations in major manufacturing hubs. This environmental advantage not only reduces permitting risks but also enhances the brand reputation of the manufacturer as a responsible partner committed to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this continuous synthesis route, drawing directly from the experimental data and comparative examples provided in the patent literature. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer or licensing agreements for this specific intermediate. The answers reflect the consensus of the experimental results which demonstrate clear superiority over prior art methods in terms of both yield and selectivity metrics.
Q: Why is modified skeletal nickel preferred over palladium carbon for this synthesis?
A: Modified skeletal nickel offers superior resistance to sulfur poisoning, which is a critical failure mode for palladium catalysts in sulfone chemistry. Additionally, it significantly reduces raw material costs while maintaining high selectivity above 97%.
Q: How does the continuous kneading process improve product quality?
A: Replacing traditional batch esterification pots with a continuous kneader ensures uniform heat distribution and mixing under vacuum. This minimizes side reactions and degradation, leading to a more stable product with yields exceeding 94%.
Q: What are the environmental benefits of this new preparation method?
A: The process drastically reduces the generation of waste acid and high-salinity wastewater compared to conventional sulfonation routes. The continuous nature of the reaction also improves energy efficiency and lowers the overall environmental footprint.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Aminophenyl-Beta-Hydroxyethyl Sulfone Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced intermediates like p-aminophenyl-beta-hydroxyethyl sulfone sulfate requires more than just a patent; it demands deep process engineering expertise and a commitment to quality that spans from gram-scale development to full-scale production. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of continuous processing and modified catalysis are fully realized in our manufacturing facilities. We operate stringent purity specifications and maintain rigorous QC labs equipped to analyze complex impurity profiles, guaranteeing that every batch meets the exacting standards required for high-performance reactive dye applications and other specialty chemical uses.
We invite forward-thinking partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain visibility into the potential economic impact of switching to this greener, more efficient manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and sustainability across your entire product portfolio.
