Advanced Preparation of Vancomycin Impurity C: Enhancing Quality Control and Commercial Scalability
The pharmaceutical industry continuously demands higher standards for impurity profiling, particularly for critical antibiotics like Vancomycin Hydrochloride. Patent CN110903346B introduces a groundbreaking methodology for the preparation of Vancomycin Impurity C (ImpC), also known as Aglucovamycin B, addressing the critical need for high-purity reference standards in quality control laboratories. This innovation represents a significant leap forward from traditional isolation techniques, offering a streamlined synthetic pathway that leverages controlled acid hydrolysis to selectively cleave the glycosidic bonds of the parent molecule. For R&D directors and procurement specialists alike, this patent signifies a shift towards more efficient, cost-effective production of complex peptide intermediates. By establishing a reliable source for this specific impurity, manufacturers can ensure rigorous compliance with pharmacopoeial standards while optimizing their supply chain for essential analytical reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the isolation of Vancomycin related substances was fraught with inefficiencies and excessive operational complexity. Existing literature, such as CN106568620B and CN106565818A, describes laborious multi-step procedures involving the preparation of aqueous solutions, precise pH adjustments, extended heating in water baths, and subsequent separation via resin chromatography. These legacy methods often necessitate additional downstream processing steps like ultrafiltration and nanofiltration to remove salts and concentrate the target analytes before final purification by high-pressure liquid chromatography. Such extensive processing not only inflates the cost of goods sold due to high energy consumption and equipment usage but also introduces potential points of failure where yield loss can occur. Furthermore, the reliance on multiple filtration stages increases the turnaround time, creating bottlenecks for laboratories requiring rapid access to high-purity impurity standards for method validation and stability testing.
The Novel Approach
In stark contrast, the method disclosed in CN110903346B simplifies the entire workflow into a cohesive, chemically driven process that minimizes unit operations. The core innovation lies in the use of a strong acid system, specifically trifluoroacetic acid (TFA) combined with dichloromethane, to effectuate the hydrolysis of the glycopeptide. This approach bypasses the need for initial resin enrichment and complex membrane filtration steps. By directly treating Vancomycin with the acid mixture at controlled temperatures between 50°C and 60°C, the process efficiently generates the aglycone structure. The subsequent workup involves a straightforward dissolution in an ethyl acetate and methanol mixture, followed by pH neutralization and filtration. This reduction in procedural steps translates directly to lower solvent consumption, reduced waste generation, and a significantly shorter production cycle, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Acid-Catalyzed Hydrolysis of Glycopeptides
The chemical transformation at the heart of this patent is the selective cleavage of the glycosidic linkage connecting the disaccharide moiety to the heptapeptide aglycone backbone of Vancomycin. Under the acidic conditions provided by the TFA and hydrochloric acid mixture, the oxygen atom in the glycosidic bond becomes protonated, increasing its leaving group ability. This facilitates the heterolytic cleavage of the C-O bond, resulting in the formation of a carbocation intermediate on the sugar ring and the release of the free hydroxyl group on the peptide core. The presence of dichloromethane serves as a co-solvent that helps solubilize the increasingly hydrophobic aglycone product as the polar sugar groups are removed or modified, driving the equilibrium towards the desired ImpC formation. Careful control of the reaction temperature and time is paramount; too short a duration leads to incomplete hydrolysis, while excessive exposure can degrade the sensitive peptide backbone, generating unwanted byproducts.
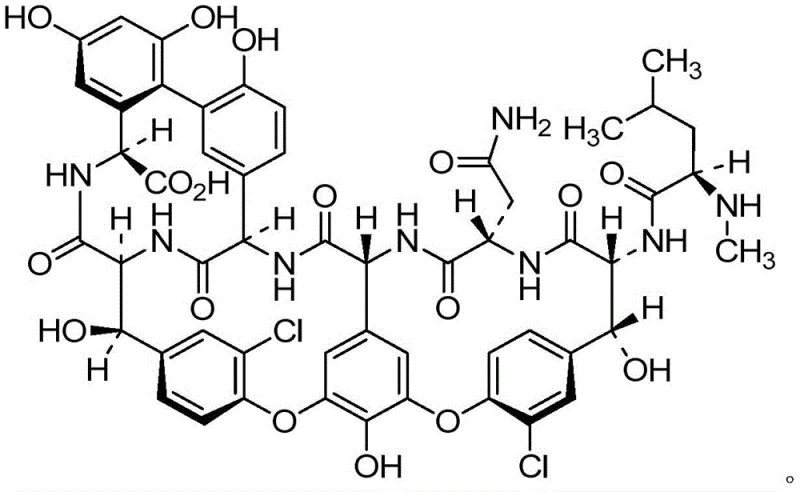
Understanding the structural nuances of the target molecule is crucial for effective purification. As illustrated in the chemical structure, Vancomycin Impurity C lacks the vancosamine and glucose sugars present in the parent antibiotic, resulting in a distinct polarity profile that can be exploited during chromatographic separation. The removal of these hydrophilic sugar units renders the molecule less polar, allowing for effective retention on reverse-phase C18 stationary phases. The patent specifies the use of a mobile phase system comprising acetonitrile and ammonium bicarbonate, which provides the necessary buffering capacity to maintain the ionization state of the amino and carboxylic acid groups on the peptide. This ensures sharp peak resolution and prevents tailing, which is essential for collecting fractions with purity levels exceeding 97%. Such mechanistic control over the separation process guarantees that the final product is suitable for use as a certified reference material in high-purity pharmaceutical intermediates applications.
How to Synthesize Vancomycin Impurity C Efficiently
The synthesis protocol outlined in the patent offers a robust framework for producing ImpC with high consistency. The process begins with the dissolution of Vancomycin hydrochloride in hydrochloric acid, followed by the addition of the TFA and dichloromethane mixture. The reaction is maintained at 50-60°C for a period ranging from 16 to 25 hours, a window determined to maximize yield while minimizing degradation. Following the reaction, the volatile acids are removed under reduced pressure, and the residue is taken up in an organic solvent system. Neutralization with sodium hydroxide precipitates salts and allows for the isolation of the crude product, which is then subjected to preparative HPLC. For a detailed, step-by-step guide on executing this synthesis with precision, please refer to the standardized operating procedure below.
- Mix vancomycin with hydrochloric acid, then add trifluoroacetic acid and dichloromethane, reacting at 50-60°C for 16-25 hours.
- Concentrate the reaction mixture under reduced pressure, dissolve the residue in an ethyl acetate and methanol mixture, and adjust pH to 7-8.
- Filter the solution, dry the filtrate to obtain crude ImpC, and purify using a C18 preparative column with acetonitrile and ammonium bicarbonate mobile phases.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route presents compelling economic and logistical benefits. The elimination of expensive resin columns and energy-intensive ultrafiltration equipment drastically reduces the capital expenditure required for production setup. Moreover, the simplified workflow decreases the dependency on specialized consumables that often have long lead times, thereby enhancing the overall resilience of the supply chain. By streamlining the process, manufacturers can respond more agilely to market demands for impurity standards, ensuring that quality control laboratories never face shortages of critical reference materials. This agility is a key factor in maintaining uninterrupted production schedules for the final drug product.
- Cost Reduction in Manufacturing: The substitution of complex multi-step purification sequences with a direct chemical hydrolysis and single-stage chromatographic purification significantly lowers operational expenses. By removing the need for resin chromatography enrichment and membrane filtration, the process reduces both material costs and labor hours associated with equipment setup and cleaning. The use of common industrial solvents like dichloromethane and ethyl acetate further ensures that raw material costs remain stable and predictable, avoiding the price volatility often seen with specialized chromatographic resins.
- Enhanced Supply Chain Reliability: The robustness of the acid hydrolysis method means that production is less susceptible to variations in raw material quality or minor fluctuations in process parameters. This reliability translates to consistent batch-to-batch quality, reducing the risk of failed production runs that could delay shipments. Furthermore, the shorter overall cycle time allows for more frequent production batches, enabling suppliers to maintain healthier inventory levels and reduce lead times for customers requiring urgent delivery of reliable pharmaceutical intermediates supplier products.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to kilogram-scale commercial production without the need for proportional increases in equipment footprint. The reduction in aqueous waste streams, typically generated by extensive washing and filtration steps in older methods, simplifies wastewater treatment requirements. This aligns with modern green chemistry principles and helps manufacturing facilities maintain strict environmental compliance, avoiding potential regulatory fines and enhancing the corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Vancomycin Impurity C. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for integrating this material into your quality assurance workflows effectively.
Q: What is the primary advantage of the new ImpC preparation method over prior art?
A: The new method utilizes a direct acid hydrolysis route using trifluoroacetic acid and dichloromethane, eliminating the need for complex multi-step processes like resin chromatography, ultrafiltration, and nanofiltration found in previous patents, thereby significantly simplifying the workflow and reducing operational costs.
Q: What purity levels can be achieved with this synthesis route?
A: By optimizing the reaction time between 16 to 25 hours and utilizing preparative HPLC with a C18 column, the method consistently yields Vancomycin Impurity C with a purity exceeding 96%, often reaching up to 97.1%, which is critical for accurate pharmacological and toxicological research.
Q: Why is high-purity ImpC essential for Vancomycin manufacturing?
A: High-purity ImpC samples are vital for establishing robust quality control standards and conducting comprehensive safety assessments. As a related substance, accurate quantification and identification of ImpC ensure the final Vancomycin Hydrochloride product meets stringent regulatory requirements for patient safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vancomycin Impurity C Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality impurity standards play in the development and manufacturing of safe pharmaceutical products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the scale. We are committed to delivering materials that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality ensures that every batch of Vancomycin Impurity C we supply is fully characterized and ready for immediate use in your validation studies.
We invite you to collaborate with us to optimize your supply chain for critical reference standards. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your R&D and production goals efficiently and reliably.
