Advanced Biocatalytic Deracemization for Industrial D-Amino Acid Production
Advanced Biocatalytic Deracemization for Industrial D-Amino Acid Production
The landscape of chiral intermediate manufacturing is undergoing a significant transformation driven by the need for greener, more efficient synthetic routes. A pivotal development in this sector is detailed in patent CN102586384B, which discloses a robust biological catalysis method for preparing D-amino acids through the deracemization of DL-amino acids. This technology leverages the specific enzymatic activity of L-amino acid oxidase found in permeabilized Alcaligenes faecalis cells to selectively oxidize L-enantiomers while preserving the desired D-enantiomers. For R&D directors and procurement specialists seeking a reliable D-amino acid supplier, this biocatalytic approach represents a paradigm shift away from traditional chemical resolution, offering a pathway to high optical purity without the burden of complex derivatization steps. The integration of co-expressed catalase further enhances the process stability by mitigating the accumulation of toxic byproducts, ensuring a consistent and scalable production environment suitable for the rigorous demands of the global pharmaceutical supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the industrial production of D-amino acids has relied heavily on chemical resolution methods or asymmetric synthesis, both of which present substantial operational and economic challenges for large-scale manufacturing. Chemical resolution typically involves the formation of diastereomeric salts using chiral resolving agents, a process that is inherently limited by a maximum theoretical yield of 50% for the desired enantiomer. Furthermore, these methods often necessitate multiple derivative and de-derivative steps, which not only increase the consumption of raw materials and solvents but also generate significant amounts of chemical waste that require costly disposal. Asymmetric synthesis, while potentially offering higher yields, frequently depends on expensive chiral catalysts or auxiliaries that are difficult to recover and recycle, leading to elevated production costs and potential metal contamination issues that complicate downstream purification for pharmaceutical applications.
The Novel Approach
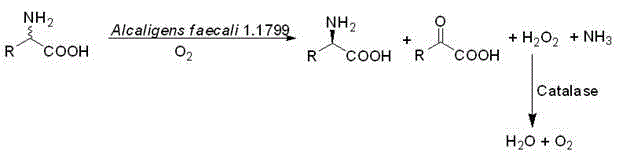
In stark contrast, the biocatalytic deracemization method described in the patent data offers a streamlined alternative that directly addresses the inefficiencies of conventional chemistry. By utilizing whole-cell biocatalysts containing L-amino acid oxidase, the process selectively converts the unwanted L-enantiomer of a DL-amino acid mixture into a corresponding keto acid, ammonia, and hydrogen peroxide, leaving the target D-amino acid intact in the reaction mixture. This selective oxidation eliminates the need for protecting group chemistry and derivatization, drastically simplifying the workflow. The resulting keto acid byproduct possesses significantly different physicochemical properties compared to the amino acid, facilitating straightforward separation through crystallization or extraction. This novel approach not only improves the overall atom economy but also aligns with modern green chemistry principles by operating under mild aqueous conditions, thereby reducing the environmental footprint associated with chiral intermediate manufacturing.
Mechanistic Insights into L-Amino Acid Oxidase Catalyzed Deracemization
The core of this technological breakthrough lies in the precise stereoselectivity of the L-amino acid oxidase enzyme housed within the Alcaligenes faecalis cellular matrix. Upon introduction of the DL-amino acid substrate, the enzyme binds specifically to the L-enantiomer, catalyzing an oxidative deamination reaction in the presence of molecular oxygen. This reaction mechanism involves the removal of the alpha-amino group, resulting in the formation of an alpha-keto acid, ammonia, and hydrogen peroxide as stoichiometric byproducts. Crucially, the D-enantiomer remains inert to this enzymatic activity due to the steric constraints of the enzyme's active site, effectively enriching the reaction mixture with the desired D-isomer. The use of permeabilized cells rather than isolated enzymes provides a protective microenvironment that enhances enzyme stability and allows for easier recovery of the biocatalyst via centrifugation, which is a critical factor for cost reduction in pharmaceutical intermediates manufacturing.
A critical component of this catalytic system is the concurrent activity of catalase, which is co-expressed within the same bacterial host. The oxidative deamination reaction inevitably generates hydrogen peroxide, a potent oxidizing agent that can irreversibly inactivate the L-amino acid oxidase and damage the cellular structure if allowed to accumulate. The endogenous catalase efficiently decomposes this hydrogen peroxide into water and oxygen, thereby maintaining the redox balance within the reaction vessel and prolonging the operational life of the biocatalyst. This self-regulating mechanism ensures that the reaction proceeds to completion without the need for external additives to scavenge peroxides, simplifying the formulation and reducing raw material costs. The synergy between the oxidase and catalase creates a robust catalytic cycle capable of handling high substrate loads while maintaining high enantiomeric excess values, often exceeding 99% ee as demonstrated in the experimental data.
How to Synthesize D-Amino Acid Efficiently
The implementation of this biocatalytic route requires careful control of cell culture conditions and permeabilization parameters to maximize enzyme expression and substrate accessibility. The process begins with the cultivation of Alcaligenes faecalis in a nutrient-rich medium where the specific L-amino acid acts as an inducer for enzyme production, followed by a permeabilization step using acetone to allow substrate diffusion into the cell. Once the biocatalyst is prepared, the deracemization reaction is conducted in an aqueous suspension at moderate temperatures, typically around 30°C, ensuring energy efficiency and safety. The detailed standardized synthesis steps, including specific media compositions, induction times, and downstream purification protocols involving acidification and propylene oxide treatment, are outlined below to guide process engineers in replicating this high-efficiency pathway.
- Cultivate Alcaligenes faecalis cells in a medium induced with L-amino acid enantiomers at 30°C for 24 hours to express L-amino acid oxidase.
- Perform cell permeability treatment using 30% acetone solution on wet biomass to facilitate substrate access while retaining enzyme activity.
- Conduct the enzymatic reaction in distilled water at 30°C, where L-enantiomers are oxidatively deaminated to keto acids, leaving D-enantiomers intact.
- Separate the D-amino acid by centrifugation, acidification, and purification using ethanol and propylene oxide to remove residual hydrochloric acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biocatalytic deracemization technology translates into tangible strategic benefits regarding cost structure and supply reliability. The elimination of chiral resolving agents and the reduction of synthetic steps directly correlate to a significant decrease in raw material expenditure and waste management costs. Unlike chemical processes that may fluctuate in price due to the volatility of petrochemical-derived reagents, the biological components of this process rely on fermentable sugars and standard nutrients, offering a more stable and predictable cost base. Furthermore, the simplicity of the downstream processing, which avoids complex chromatographic separations often required in asymmetric synthesis, reduces the capital investment needed for purification equipment and shortens the overall production cycle time, enhancing the agility of the supply chain.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of derivatization and de-derivatization stages that are characteristic of traditional resolution methods. By bypassing these steps, manufacturers save substantially on reagents, solvents, and labor hours associated with multi-step synthesis. Additionally, the use of whole-cell biocatalysts eliminates the need for expensive enzyme purification procedures, as the crude permeabilized cells serve as the effective catalyst. This streamlined approach minimizes the loss of product during intermediate isolation steps, thereby improving the overall mass balance and yield efficiency without requiring additional capital investment in specialized reactor hardware.
- Enhanced Supply Chain Reliability: The robustness of the Alcaligenes faecalis strain contributes to a more resilient supply chain by reducing the risk of batch failures associated with sensitive chemical catalysts. The biological system operates under mild physiological conditions, reducing the safety hazards related to high pressure or extreme temperatures, which in turn lowers insurance and compliance costs. Moreover, the versatility of the method allows for the production of a wide range of D-amino acids, from aliphatic to aromatic varieties, using a unified platform technology. This flexibility enables suppliers to respond rapidly to changing market demands for different chiral intermediates without the need for extensive process requalification or new equipment installation.
- Scalability and Environmental Compliance: Scaling this biocatalytic process from laboratory to commercial production is facilitated by the use of standard fermentation and bioreactor technologies that are well-established in the industry. The aqueous nature of the reaction medium significantly reduces the emission of volatile organic compounds (VOCs), aligning with increasingly stringent environmental regulations and sustainability goals. The decomposition of hydrogen peroxide into harmless water and oxygen further ensures that the effluent stream is less toxic and easier to treat, reducing the burden on wastewater treatment facilities. These environmental advantages not only mitigate regulatory risks but also enhance the corporate social responsibility profile of the manufacturing operation, which is increasingly valued by downstream pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this deracemization technology. These insights are derived directly from the patent specifications and experimental embodiments, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this biocatalytic route into their existing production portfolios for high-purity D-amino acids.
Q: What is the primary advantage of using Alcaligenes faecalis for D-amino acid synthesis?
A: The primary advantage is the ability to perform direct deracemization without chemical derivatization steps. The L-amino acid oxidase specifically targets the L-enantiomer for oxidative deamination, leaving the desired D-enantiomer untouched, which simplifies purification and avoids the 50% theoretical yield limit of traditional resolution.
Q: How is the toxicity of hydrogen peroxide managed in this biocatalytic process?
A: The process utilizes the co-expression of catalase within the Alcaligenes faecalis cells. This enzyme efficiently decomposes the hydrogen peroxide generated during the oxidative deamination of the L-enantiomer into water and oxygen, preventing enzyme inactivation and ensuring reaction stability.
Q: Can this method be applied to aromatic D-amino acids?
A: Yes, the patent data confirms the versatility of this method for both aliphatic and aromatic substrates. Successful preparation has been demonstrated for D-phenylalanine, D-tyrosine, and D-tryptophan with high enantiomeric excess (ee > 99%), indicating broad substrate specificity suitable for diverse pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-Amino Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced biocatalytic technologies to meet the evolving needs of the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the Alcaligenes faecalis deracemization method can be successfully transferred from the lab to the plant. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of D-amino acid meets the highest standards of optical purity and chemical quality required for API synthesis. Our commitment to technical excellence allows us to navigate the complexities of biocatalytic scale-up, delivering consistent supply continuity for our partners.
We invite you to collaborate with us to leverage these cutting-edge synthetic routes for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in chiral biocatalysis can drive value and efficiency in your supply chain.
