Scaling D-Glutamine Production: Advanced Chemoenzymatic Technology for Global Pharmaceutical Supply Chains
Scaling D-Glutamine Production: Advanced Chemoenzymatic Technology for Global Pharmaceutical Supply Chains
The pharmaceutical industry's demand for high-purity D-amino acids has surged, driven by their critical role in synthesizing beta-lactam antibiotics and physiologically active peptides. Patent CN110643670A introduces a groundbreaking chemoenzymatic methodology that fundamentally reshapes the production landscape for D-glutamine, a key medicinal amino acid used in treating gastric ulcers and leukemia. This technology addresses the longstanding challenges of low yield and high cost associated with traditional racemization and resolution techniques. By integrating a robust four-step chemical synthesis with a highly stereoselective enzymatic cleavage, the process achieves a total DL-glutamine yield of 67% and a D-glutamine recovery of 95% of the theoretical yield. For global procurement leaders, this represents a pivotal shift towards more sustainable and economically viable sourcing strategies for complex amino acid intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of D-amino acids has been plagued by inefficient resolution methods that struggle with scalability and cost-effectiveness. Traditional derivative resolution methods rely heavily on expensive chiral resolving agents such as strychnine, camphor sulfonic acid, or tartaric acid to separate enantiomers. These processes are not only capital intensive due to the high cost of the resolving agents but also operationally complex, requiring multiple cycles of recrystallization and hydrolysis to achieve acceptable purity. Furthermore, the non-target enantiomer often requires repeated racemization, leading to significant material loss and increased waste generation. Alternative methods like cyclic polyether resolution, while avoiding derivatization, are currently restricted to laboratory scales due to the prohibitive synthesis costs of optically active crown ethers. Additionally, direct chemical racemization of L-glutamine is fraught with difficulty because the gamma-amide bond is unstable under acidic, alkaline, or high-temperature conditions, often leading to decomposition into glutamic acid or lactonization into pyrrole carboxylic acid.
The Novel Approach
The chemoenzymatic route disclosed in the patent offers a decisive break from these limitations by utilizing low-cost DL-glutamic acid as the primary feedstock instead of unstable L-glutamine. The innovation lies in the strategic use of the phthaloyl group as a protective mask, which stabilizes the molecule during the critical amidation steps, preventing the degradation issues common in direct glutamine synthesis. This approach simplifies the workflow into four manageable chemical reactions followed by a single enzymatic resolution step. The process parameters have been meticulously optimized to enhance the yield of DL-glutamine from the historical baseline of 57% to an impressive 67%. Moreover, the enzymatic resolution step operates under mild conditions without the need for substrate modification, leveraging the natural stereoselectivity of aminopeptidase to cleave only the L-enantiomer. This results in a streamlined workflow that is inherently safer, more environmentally friendly, and far more conducive to large-scale industrial implementation compared to legacy chemical resolution techniques.
Mechanistic Insights into Phthaloyl-Protection and Aminopeptidase Resolution
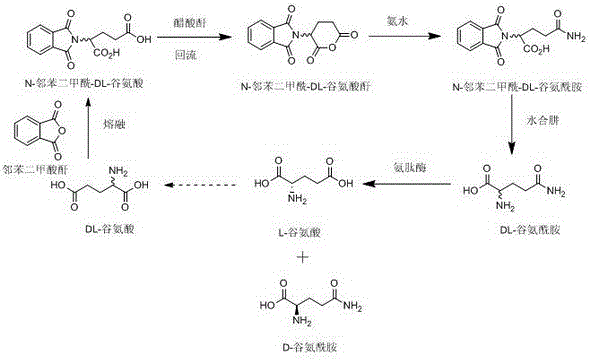
The core chemical ingenuity of this process revolves around the stability conferred by the N-phthaloyl protecting group. In the initial steps, DL-glutamic acid reacts with phthalic anhydride at elevated temperatures (150-200°C) to form N-phthaloyl-DL-glutamic acid. This imide formation effectively blocks the amino group, preventing unwanted side reactions during the subsequent activation of the carboxylic acid. The conversion to the mixed anhydride using acetic anhydride activates the gamma-carboxyl group for nucleophilic attack by ammonia. Crucially, the phthaloyl group remains stable throughout this amidation, ensuring that the gamma-amide bond of the glutamine skeleton is formed without premature hydrolysis. The final chemical deprotection utilizes hydrazine hydrate, which selectively cleaves the phthalimide ring to release the free amine, yielding DL-glutamine with high purity. This specific sequence avoids the harsh acidic or basic conditions that typically degrade free glutamine, thereby preserving the structural integrity of the target molecule.
Following the chemical synthesis, the resolution mechanism relies on the exquisite stereospecificity of aminopeptidase enzymes. Unlike chemical catalysts which often lack chiral discrimination, aminopeptidase possesses an active site geometry that strictly recognizes and hydrolyzes the peptide or amide bonds of L-amino acid derivatives. When introduced to the racemic DL-glutamine solution, the enzyme selectively cleaves the amide bond of the L-glutamine, converting it into L-glutamic acid and ammonia. The D-glutamine enantiomer, being the mirror image, does not fit into the enzyme's catalytic pocket and remains untouched in the solution. This biological discrimination allows for the separation of the two enantiomers based on their differing chemical properties post-reaction; the resulting L-glutamic acid can be crystallized out at its isoelectric point, while the D-glutamine remains in the mother liquor for further purification via ion exchange resin. This enzymatic precision eliminates the need for complex chromatographic separations typically required for racemate resolution.
How to Synthesize D-Glutamine Efficiently
Implementing this chemoenzymatic protocol requires precise control over reaction temperatures and pH levels to maximize the efficiency of both the chemical protection steps and the biological resolution phase. The synthesis begins with the melt condensation of DL-glutamic acid and phthalic anhydride, followed by reflux in acetic anhydride to generate the reactive anhydride intermediate. Subsequent ammonolysis and hydrazinolysis steps must be carefully monitored to ensure complete conversion while minimizing byproduct formation. Once the DL-glutamine racemate is secured, the process shifts to biocatalysis, where the cultivation of high-activity aminopeptidase strains becomes critical. The detailed standardized synthesis steps, including specific molar ratios, reaction times, and purification protocols necessary for GMP-compliant manufacturing, are outlined below.
- Synthesize N-phthaloyl-DL-glutamic acid by reacting DL-glutamic acid with phthalic anhydride at 150-200°C, followed by conversion to the anhydride using acetic anhydride.
- React the anhydride with ammonia water to form N-phthaloyl-DL-glutamine, then remove the phthaloyl protecting group using hydrazine hydrate to obtain DL-glutamine.
- Resolve the DL-glutamine racemate using cultured aminopeptidase at pH 7-10 and 20-45°C to selectively hydrolyze the L-enantiomer, isolating pure D-glutamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this chemoenzymatic methodology offers substantial strategic benefits beyond mere technical feasibility. The most immediate impact is seen in the drastic reduction of raw material costs. By shifting the feedstock from expensive, unstable L-glutamine or costly chiral resolving agents to commodity-grade DL-glutamic acid, the overall cost of goods sold (COGS) is significantly lowered. DL-glutamic acid is a bulk chemical produced at massive scales globally, ensuring a stable and resilient supply base that is less susceptible to the market volatility often seen with specialty fine chemicals. Furthermore, the elimination of expensive catalysts like crown ethers or strychnine salts removes a major variable cost driver, allowing for more predictable budgeting and long-term price stability for downstream pharmaceutical manufacturers.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the high atom economy and the recyclability of byproducts. A critical feature of this route is the ability to racemize the L-glutamic acid byproduct back into DL-glutamic acid, which can then be fed back into the start of the process. This circular approach minimizes raw material waste and maximizes the utility of every kilogram of input. Additionally, the simplicity of the purification steps—relying primarily on crystallization and filtration rather than energy-intensive distillation or preparative HPLC—reduces utility consumption and operational overhead. The removal of transition metal catalysts or heavy metal reagents also simplifies the purification burden, reducing the need for expensive scavenging resins and extensive testing for residual metals, which translates directly into lower quality control costs.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the chemical intermediates involved. The N-phthaloyl protected intermediates are crystalline solids with excellent stability, allowing them to be stored and transported without the stringent temperature controls required for free amino acids. This stability reduces the risk of spoilage during logistics and enables the maintenance of strategic inventory buffers. Moreover, the enzymatic step utilizes readily available microbial strains that can be cultured in-house or sourced from multiple established biotech suppliers, mitigating the risk of single-source dependency. The process's tolerance for variation in reaction conditions ensures consistent output even when scaling up from pilot plants to multi-ton commercial reactors, guaranteeing reliable delivery schedules for critical API intermediates.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process aligns perfectly with modern green chemistry principles. The avoidance of toxic organic solvents in favor of aqueous systems during the enzymatic resolution significantly reduces the volume of hazardous waste requiring treatment. The use of hydrazine hydrate, while requiring careful handling, is a well-understood reagent in industrial chemistry with established safety protocols, unlike some novel chiral ligands that lack long-term safety data. The high yield of the process means less waste per unit of product, lowering the carbon footprint of the manufacturing operation. This environmental efficiency not only reduces disposal costs but also facilitates easier regulatory approval in markets with strict environmental compliance standards, such as the EU and North America, making the supply chain more resilient to regulatory shocks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chemoenzymatic D-glutamine synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for R&D teams planning technology transfer and for procurement officers negotiating supply agreements.
Q: What are the primary advantages of this chemoenzymatic method over traditional chemical resolution?
A: Unlike traditional derivative resolution which requires expensive chiral resolving agents like strychnine or tartaric acid, this method utilizes low-cost DL-glutamic acid and a recyclable enzymatic step. It eliminates the need for complex recrystallization cycles and achieves a theoretical yield of 95% for the D-enantiomer, significantly lowering raw material costs.
Q: How does the process handle the instability of glutamine during synthesis?
A: The process employs a phthaloyl protecting group which stabilizes the gamma-amide bond against hydrolysis and lactonization during the harsh chemical steps. The protecting group is only removed under mild conditions using hydrazine hydrate, preventing the formation of pyroglutamic acid and ensuring high product integrity.
Q: Is the L-glutamic acid byproduct wasted in this process?
A: No, the process is designed for circular efficiency. The L-glutamic acid generated during the enzymatic resolution can be chemically racemized back into DL-glutamic acid, which serves as the starting material for the next batch, thereby maximizing atom economy and minimizing waste disposal costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-Glutamine Supplier
The chemoenzymatic synthesis of D-glutamine represents a mature and highly efficient pathway for producing this vital pharmaceutical intermediate. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate this patented methodology into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of D-glutamine meets the exacting standards required for medicinal applications, including low impurity profiles and consistent optical rotation.
We invite global partners to collaborate with us to leverage this cost-effective technology for their supply chains. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your procurement budget. We encourage you to contact our technical procurement team to request specific COA data from our pilot runs and comprehensive route feasibility assessments. By partnering with us, you secure a reliable source of high-purity D-glutamine that combines cutting-edge biocatalysis with robust chemical engineering.
