Advanced Metal-Free Synthesis Of Ruxolitinib Intermediate For Commercial Scale-Up
Advanced Metal-Free Synthesis Of Ruxolitinib Intermediate For Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust and scalable synthetic routes for critical kinase inhibitors, and the recent disclosure in patent CN115141180A offers a transformative approach to producing the key Ruxolitinib intermediate. This technology addresses long-standing challenges in the manufacturing of JAK inhibitors by introducing a streamlined, metal-free strategy that enhances both chemical efficiency and operational safety. By shifting away from traditional transition metal catalysis, this method not only mitigates the risk of heavy metal contamination but also simplifies the overall process flow, making it highly attractive for large-scale commercial production. For global procurement teams and R&D directors, understanding the nuances of this synthesis is crucial for securing a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials consistently. The following analysis dissects the technical advantages and commercial implications of this innovative pathway.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Ruxolitinib precursors, such as those disclosed in earlier patents like CN202080024598.1, often rely heavily on palladium-catalyzed cross-coupling reactions to construct the core heterocyclic framework. As illustrated in the prior art pathway, these methods typically involve the use of expensive catalysts such as tetrakis(triphenylphosphine)palladium, which significantly inflates the raw material costs and introduces complex purification requirements to meet regulatory limits on residual metals. Furthermore, these conventional processes frequently necessitate rigorous inert gas protection to prevent catalyst deactivation, adding layers of operational complexity and equipment costs to the manufacturing setup. The chemical selectivity in these older routes can also be problematic, with side reactions such as double substitution of chlorine atoms leading to bis-amino by-products that compromise the overall yield and quality of the final intermediate. These factors collectively create bottlenecks in cost reduction in API manufacturing and pose risks to supply chain continuity due to the reliance on specialized catalytic systems.
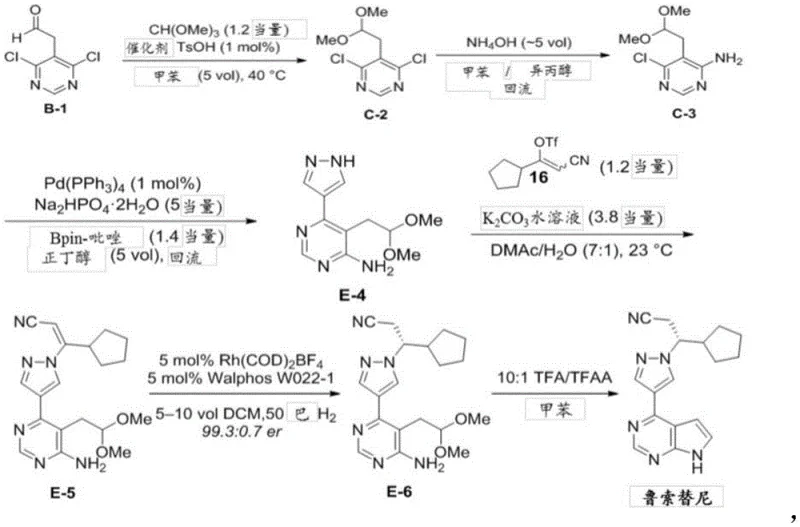
The Novel Approach
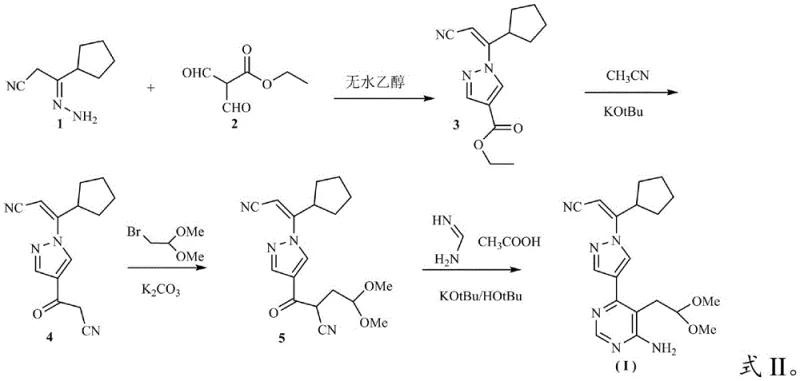
In stark contrast, the methodology presented in CN115141180A circumvents these issues by employing a sequence of cyclization, condensation, and substitution reactions that do not require precious metal catalysts. This new strategy utilizes readily available starting materials like 3-cyclopentyl-3-hydrazonopropionitrile and ethyl 2-formyl-3-oxopropionate to build the molecular architecture through efficient bond-forming steps. The elimination of palladium not only removes the burden of heavy metal scavenging but also allows for milder reaction conditions that are more forgiving in an industrial setting. By avoiding the need for inert atmosphere protection, the process becomes inherently safer and easier to scale, reducing the capital expenditure required for specialized reactor setups. This shift represents a significant advancement in the commercial scale-up of complex pharmaceutical intermediates, offering a pathway that is both economically viable and technically robust for high-volume production environments.
Mechanistic Insights into Metal-Free Pyrimidine Cyclization
The core of this innovative synthesis lies in its ability to construct the pyrazolo[1,5-a]pyrimidine scaffold through a carefully orchestrated series of nucleophilic attacks and cyclizations without metal mediation. The initial step involves the condensation of the hydrazonopropionitrile derivative with the formyl-oxopropionate ester, driven by thermal energy in an alcoholic solvent to form the pyrazole ring with high regioselectivity. Subsequent functionalization is achieved through a base-mediated substitution using potassium carbonate in polar aprotic solvents, which facilitates the introduction of the dimethoxyethyl side chain essential for the biological activity of the final drug. The final ring closure to form the pyrimidine moiety is executed using formamidine acetate under basic conditions, a transformation that proceeds cleanly to yield the target structure with minimal impurity formation. This mechanistic clarity ensures that the process is reproducible and controllable, key attributes for any high-purity Ruxolitinib intermediate intended for clinical or commercial use.
Impurity control is another critical aspect where this novel route excels, as the absence of transition metals eliminates a major source of trace contaminants that are difficult to remove during downstream processing. The reaction conditions are optimized to minimize side reactions, such as over-alkylation or hydrolysis, which are common pitfalls in the synthesis of multifunctional heterocycles. By maintaining strict control over temperature profiles and stoichiometric ratios, particularly during the substitution and cyclization phases, the process achieves a purity profile that often exceeds 99% without the need for extensive chromatographic purification. This level of chemical integrity is vital for R&D directors who must ensure that the intermediate meets the stringent impurity specifications required for regulatory filings. The robustness of the chemistry also means that variations in raw material quality can be accommodated more easily, further enhancing the reliability of the supply chain.
How to Synthesize Ruxolitinib Intermediate Efficiently
Implementing this synthesis requires precise adherence to the reaction parameters outlined in the patent to maximize yield and purity while maintaining safety standards. The process begins with the preparation of the pyrazole core, followed by sequential functionalization steps that build complexity without compromising stability. Operators must monitor reaction progress closely, particularly during the exothermic substitution phase, to ensure optimal conversion rates. While the general workflow is straightforward, the specific details regarding solvent choices, base equivalents, and workup procedures are critical for success. The detailed standardized synthesis steps see guide below.
- Cyclization of 3-cyclopentyl-3-hydrazonopropionitrile with ethyl 2-formyl-3-oxopropionate in ethanol at 70-75°C.
- Condensation with potassium tert-butoxide in acetonitrile at 45-50°C to form the pyrazole core.
- Substitution reaction with bromoacetaldehyde dimethyl acetal under basic conditions followed by pyrimidine ring closure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this metal-free synthesis route offers substantial benefits that extend beyond simple chemical efficiency, directly impacting the bottom line and operational resilience of pharmaceutical manufacturers. By eliminating the dependency on volatile and expensive palladium catalysts, companies can achieve significant cost reduction in manufacturing without sacrificing product quality or performance. The simplified process flow, which does not require specialized inert gas infrastructure, reduces both capital investment and ongoing operational expenses, making it an ideal solution for facilities looking to optimize their production capabilities. Additionally, the use of common, commercially available reagents ensures that sourcing remains stable and unaffected by the supply fluctuations often seen with specialized catalytic materials. This stability is crucial for reducing lead time for high-purity JAK inhibitors, allowing procurement managers to plan inventory more effectively and respond quickly to market demands.
- Cost Reduction in Manufacturing: The removal of precious metal catalysts from the synthetic route eliminates a major cost driver associated with both raw material procurement and waste disposal. Without the need for expensive ligands or metal scavengers, the overall cost of goods sold is drastically lowered, allowing for more competitive pricing strategies in the global market. Furthermore, the simplified purification process reduces solvent consumption and energy usage, contributing to additional savings in utility costs. These economic advantages make the process highly scalable and suitable for continuous manufacturing environments where margin optimization is a priority.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals rather than specialized catalysts insulates the production schedule from supply chain disruptions that frequently affect the availability of rare earth metals or complex organometallics. The robustness of the reaction conditions means that production can be maintained across different geographic locations without the need for highly specialized technical support or equipment. This flexibility ensures a consistent supply of materials, which is essential for maintaining uninterrupted API production schedules and meeting contractual obligations to downstream partners. It also simplifies the qualification process for new suppliers, as the raw materials are widely standardized and easily sourced.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metals align perfectly with modern environmental, health, and safety (EHS) standards, facilitating easier regulatory approval and permitting for large-scale plants. Waste streams are simpler to treat and dispose of, reducing the environmental footprint of the manufacturing process and lowering compliance costs associated with hazardous waste management. The process is inherently safer, with reduced risks of exothermic runaways or pressure buildup, which enhances operational safety for plant personnel. These factors combined make the technology highly attractive for long-term investment and capacity expansion in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity for stakeholders evaluating its potential for integration into their existing portfolios. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for assessing the feasibility of adopting this route for commercial production and for engaging in meaningful discussions with technical partners.
Q: Does this synthesis route require expensive palladium catalysts?
A: No, the patented method CN115141180A specifically eliminates the need for palladium catalysts like Pd(PPh3)4, thereby removing the risk of heavy metal residues and reducing raw material costs significantly.
Q: What is the expected purity of the intermediate using this method?
A: Experimental data indicates that the final product can achieve an HPLC purity of up to 99.5%, meeting stringent specifications required for downstream API manufacturing without extensive purification.
Q: Is inert gas protection required for this process?
A: Unlike conventional methods that require strict inert gas protection for sensitive catalytic steps, this novel approach operates under mild conditions without the need for complex atmospheric controls, simplifying operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ruxolitinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a robust and scalable supply chain for high-value pharmaceutical intermediates like the Ruxolitinib precursor. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical clients without compromising on quality. We are committed to maintaining stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our expertise in metal-free synthesis allows us to offer a product that is not only cost-effective but also environmentally sustainable, aligning with the growing demand for green chemistry solutions in the life sciences sector.
We invite you to collaborate with us to optimize your supply chain and secure a steady source of high-quality intermediates for your JAK inhibitor programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our process can enhance your operational efficiency. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our technical capabilities can support your long-term strategic goals in the pharmaceutical market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
