Advanced Hydantoinase Catalysis for Commercial Scale Glutathione Manufacturing
Advanced Hydantoinase Catalysis for Commercial Scale Glutathione Manufacturing
The global demand for high-purity glutathione (GSH), a critical tripeptide involved in cellular antioxidant defense and detoxification, continues to surge across pharmaceutical and nutraceutical sectors. Traditional production methods, including extraction from yeast and direct fermentation, often struggle with low yields, high impurity profiles, and significant environmental burdens. A groundbreaking solution is presented in patent CN101701239B, which details a novel hydantoinase method for preparing glutathione. This technology leverages a sophisticated chemo-enzymatic pathway that utilizes L-glutamic acid and cyanate to form a key hydantoin intermediate, subsequently coupled with cysteine and glycine derivatives. By employing wet bacterial cells containing L-hydantoinase and L-carbamoylase as biocatalysts, this method achieves exceptional regioselectivity and operational simplicity. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this patent represents a paradigm shift towards more efficient, scalable, and cost-effective manufacturing of complex peptide APIs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of glutathione has been plagued by significant technical and economic bottlenecks inherent to extraction and fermentation technologies. The extraction method, while classical, relies heavily on animal and plant tissues or yeast rich in GSH, necessitating the use of large volumes of organic solvents and complex enzymatic treatments with amylase and protease. This results in severe environmental pollution, low product purity, and prohibitively high costs due to poor recovery rates. Similarly, the fermentation method, although biological, faces challenges such as low transformation efficiency, extended fermentation cycles, and low intracellular GSH content, which demands expensive and complicated downstream separation technologies. Furthermore, purely chemical synthesis routes typically require step-by-step protection and deprotection of the alpha-amino and alpha-carboxyl groups of L-glutamic acid to ensure the gamma-carboxyl group reacts. These repeated protection cycles often lead to peptide bond rupture, racemization, and reduced optical purity, rendering the final product unsuitable for high-grade pharmaceutical applications without extensive purification.
The Novel Approach
The hydantoinase method described in the patent data offers a transformative alternative by fundamentally redesigning the synthetic logic to minimize protection steps and maximize enzymatic efficiency. Instead of relying on ATP-dependent GSH synthetases or struggling with low-yield fermentation, this approach utilizes a hydantoin ring to co-protect the alpha-amino and alpha-carboxyl groups of L-glutamic acid in a single step. This strategic masking leaves only the gamma-carboxyl group exposed, ensuring absolute position specificity during the subsequent peptide coupling reactions. The process avoids the repeated protection and deprotection cycles typical of standard chemical synthesis, thereby drastically reducing side reactions and operational complexity. Moreover, by utilizing a dual-enzyme system (L-hydantoinase and L-carbamoylase) contained within wet bacterial cells, the method circumvents the need for expensive ATP regeneration systems required by other enzymatic routes, offering a streamlined path to cost reduction in pharmaceutical intermediates manufacturing.
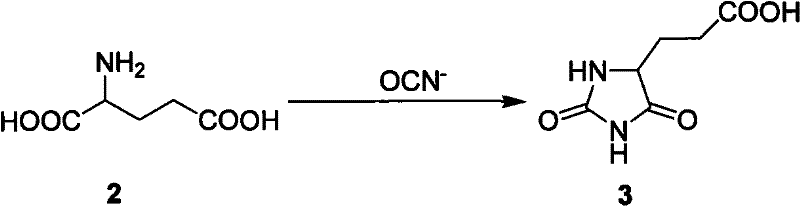
Mechanistic Insights into Hydantoinase-Catalyzed Ring Opening
The core innovation of this technology lies in the precise mechanistic control exerted by the hydantoin ring and the subsequent enzymatic hydrolysis. The process initiates with the reaction of L-glutamic acid and cyanate under alkaline conditions (pH 8.5-10.0) to form N-carbamoyl-L-glutamic acid, which undergoes acidification and cyclization at elevated temperatures (90-120°C) to yield 5-carboxyethyl hydantoin. This intermediate is crucial as it locks the stereochemistry and reactivity of the glutamic acid backbone. Following this, standard peptide coupling reagents such as DCC/HOBt or DIC/HOBt are employed in DMF to sequentially attach S-protected L-cysteine and glycine. The resulting N-hydantoinylpropionyl-S-R-L-cysteinyl-glycine serves as the substrate for the biocatalytic step. The use of wet cells from Bacillus fordii MH602, which naturally express both L-hydantoinase and L-carbamoylase, allows for the simultaneous hydrolysis of the hydantoin ring and the removal of the carbamoyl group. This dual-action enzymatic cleavage regenerates the free alpha-amino and alpha-carboxyl groups of the glutamic acid residue without affecting the newly formed peptide bonds, a feat difficult to achieve with chemical hydrolysis alone.
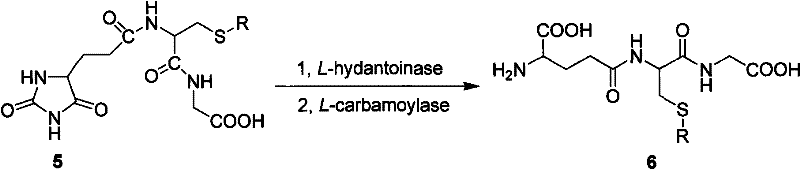
Impurity control in this synthesis is inherently robust due to the high specificity of the enzymatic catalysts and the protective nature of the hydantoin scaffold. In conventional chemical synthesis, the risk of racemization at the chiral center of glutamic acid is a persistent threat during activation and coupling steps. However, the hydantoin formation stabilizes the chiral center, and the mild enzymatic hydrolysis conditions (pH 7.5-9.5, 35-45°C) preserve optical integrity. The patent data indicates that the wet cell catalyst effectively converts the hydantoin precursor into S-protected glutathione with high conversion rates, minimizing the formation of hydrolyzed byproducts or rotation peptides that often plague gamma-glutamyl transpeptidase (GGT) methods. Furthermore, the final deprotection of the sulfur group (S-R) can be achieved using standard hydrogenation or acidolysis methods, yielding the final reduced glutathione with high purity. This rigorous control over the reaction pathway ensures that the high-purity pharmaceutical intermediates produced meet the stringent quality specifications required for injectable formulations and clinical applications.
How to Synthesize Glutathione Efficiently
The synthesis of glutathione via this hydantoinase route involves a sequence of well-defined chemical and enzymatic transformations that balance efficiency with scalability. The process begins with the cyclization of commodity chemicals like L-glutamic acid and cyanate, followed by solution-phase peptide coupling in polar aprotic solvents. The critical biocatalytic step utilizes inexpensive wet biomass rather than purified enzymes, significantly lowering input costs. For detailed laboratory protocols and specific reaction parameters regarding temperature, pH, and molar ratios, please refer to the standardized synthesis guide below.
- Cyclize L-glutamic acid with cyanate under alkaline conditions to form 5-carboxyethyl hydantoin, protecting the alpha-amino and alpha-carboxyl groups.
- Perform sequential peptide coupling reactions with S-protected L-cysteine and glycine using standard coupling reagents like DCC/HOBt in DMF.
- Hydrolyze the hydantoin ring using wet cells containing L-hydantoinase and L-carbamoylase, followed by removal of the sulfur protecting group to yield final glutathione.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the hydantoinase method offers compelling strategic advantages over legacy production technologies. The primary value driver is the substantial simplification of the manufacturing workflow, which directly translates to improved margin structures and supply reliability. By eliminating the need for complex ATP regeneration systems and avoiding the low yields associated with fermentation, manufacturers can achieve a more predictable and consistent output. The reliance on readily available raw materials such as L-glutamic acid, cyanate, and glycine ensures that the supply chain is not vulnerable to the volatility often seen with specialized biological feedstocks. Additionally, the use of wet cell catalysts removes the capital expenditure and operational complexity associated with enzyme purification facilities, allowing for a more agile production setup that can be scaled rapidly to meet market demand fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the elimination of expensive reagents and energy-intensive steps found in alternative methods. Unlike GSH synthetase pathways that require continuous ATP supplementation, this hydantoinase route operates without external energy cofactors, drastically reducing utility and reagent costs. Furthermore, the co-protection strategy of the hydantoin ring removes the need for multiple discrete protection and deprotection stages, which typically consume significant amounts of solvents and coupling agents. This streamlining of the synthetic route leads to a leaner material bill and reduced waste disposal costs, providing a clear pathway for cost reduction in pharmaceutical intermediates manufacturing without compromising product quality.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the catalytic system and the stability of the intermediates. The wet cell catalyst, derived from Bacillus fordii, is stable and can be produced in bulk, ensuring that the biocatalytic step is never a bottleneck. The chemical intermediates, such as the hydantoin derivatives, are solid compounds that can be isolated, stored, and quality-controlled before proceeding to the enzymatic step, adding a layer of flexibility to the production schedule. This modularity allows manufacturers to stockpile key intermediates during periods of low demand and ramp up final conversion quickly, thereby reducing lead time for high-purity pharmaceutical intermediates and ensuring consistent availability for downstream API synthesis.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this method aligns well with modern green chemistry principles. The avoidance of heavy metal catalysts in the main synthetic sequence and the use of aqueous enzymatic steps reduce the toxic load of the effluent. The process operates under mild thermal and pH conditions, which lowers energy consumption for heating and cooling compared to harsh chemical hydrolysis methods. As production scales from pilot batches to multi-ton quantities, the simplicity of the unit operations—primarily stirring, filtration, and crystallization—facilitates the commercial scale-up of complex pharmaceutical intermediates. This ease of scale-up, combined with lower solvent intensity, ensures that the manufacturing process remains compliant with increasingly stringent environmental regulations globally.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydantoinase technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and strategic benefits for potential partners. Understanding these nuances is essential for evaluating the fit of this technology within your existing supply chain infrastructure.
Q: What is the primary advantage of the hydantoinase method over traditional fermentation for glutathione production?
A: Unlike fermentation, which suffers from low transformation efficiency and complex downstream processing, the hydantoinase method utilizes a chemo-enzymatic approach that ensures high regioselectivity and avoids the need for expensive ATP regeneration systems required by GSH synthetase pathways.
Q: How does the hydantoin ring protect the glutamic acid molecule during synthesis?
A: The hydantoin ring formation simultaneously protects both the alpha-amino group and the alpha-carboxyl group of L-glutamic acid. This unique co-protection strategy leaves only the gamma-carboxyl group exposed, guaranteeing that peptide bonding occurs exclusively at the correct position without needing separate protection and deprotection steps.
Q: Is the enzymatic hydrolysis step scalable for industrial production?
A: Yes, the process utilizes wet bacterial cells containing the necessary enzymes (L-hydantoinase and L-carbamoylase), which eliminates the high cost of enzyme purification. The reaction operates under mild conditions (pH 7.5-9.5, 35-45°C), making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glutathione Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the hydantoinase method for producing high-value peptides like glutathione. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial realities. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of glutathione meets the highest international standards for pharmaceutical and food-grade applications. We are committed to leveraging advanced chemo-enzymatic technologies to deliver superior products that drive value for our global clientele.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your supply chain and reduce overall production costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us demonstrate how our expertise can support your long-term strategic goals in the competitive landscape of fine chemicals and API intermediates.
