Advanced Manufacturing of High-Purity Vigabatrin Intermediates for Global Epilepsy Therapy Supply Chains
The global pharmaceutical landscape for antiepileptic drugs continues to demand higher standards of purity and supply chain reliability, particularly for critical therapies like Vigabatrin. As detailed in the recent patent CN110713440B, a significant technological breakthrough has been achieved in the synthesis of 4-amino-5-hexenoic acid, the key active pharmaceutical ingredient (API) for Vigabatrin. This patent outlines a sophisticated multi-step synthetic pathway that addresses the longstanding issue of impurity profiles associated with earlier manufacturing methods. By leveraging a strategic silyl-protection strategy combined with a novel ternary solvent recrystallization system, this new methodology promises to deliver Vigabatrin intermediates with HPLC purity exceeding 99.9%. For R&D directors and procurement specialists in the neurology sector, understanding the nuances of this process is essential for securing a reliable API intermediate supplier capable of meeting stringent regulatory specifications while optimizing production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Vigabatrin has been plagued by challenges related to impurity control and yield optimization. Prior art, such as the methods disclosed in US4178463, often relied on direct cyclization or hydrolysis routes that generated complex mixtures of stereoisomers and polymeric byproducts. These conventional pathways frequently resulted in crude products that required extensive and costly downstream purification efforts, often involving multiple chromatographic separations which are not ideal for industrial scale-up. The presence of trace impurities in the final API can lead to significant regulatory hurdles and potential safety concerns for patients suffering from refractory epilepsy. Furthermore, older synthetic routes sometimes utilized harsh conditions that compromised the stability of the vinyl group, leading to reduced overall yields and inconsistent batch-to-batch quality. As illustrated in the reaction schemes of previous patents, the lack of a robust protecting group strategy often left the reactive amine functionality vulnerable to side reactions during the esterification and hydrolysis stages.
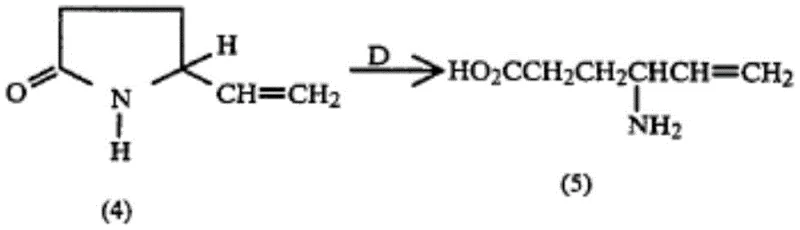
The Novel Approach
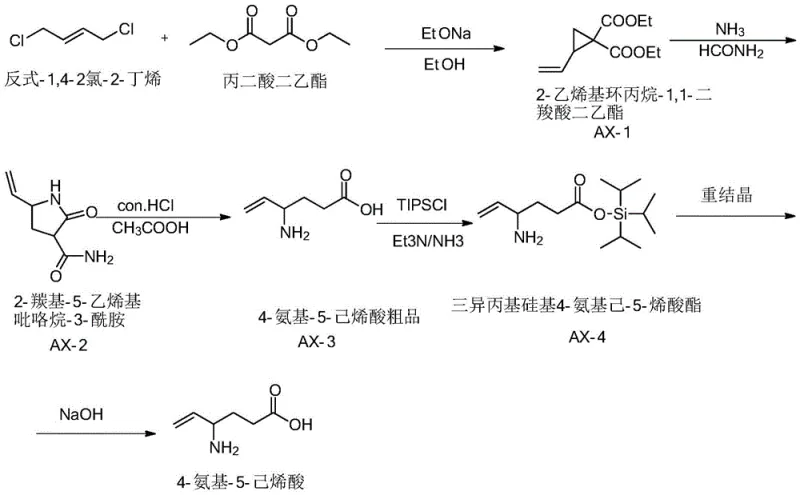
In stark contrast to these legacy methods, the process described in CN110713440B introduces a refined approach centered on the intermediate 2-carbonyl-5-vinyl-pyrrolidine-3-amide. This novel pathway begins with the efficient construction of the cyclopropane ring using diethyl malonate and trans-1,4-dichloro-2-butene, followed by a high-pressure ammonolysis to form the lactam core. The true innovation, however, lies in the subsequent steps where the crude amino acid is converted into a triisopropylsilyl (TIPS) protected ester. This protection step is not merely a defensive measure; it fundamentally changes the physical properties of the molecule, enabling a highly selective recrystallization that acts as a 'molecular filter.' By converting the polar, difficult-to-crystallize amino acid into a lipophilic silyl ester, the process facilitates the removal of non-polar impurities and structural analogs that co-precipitate in traditional methods. This strategic pivot allows manufacturers to achieve commercial grade purity through simple crystallization rather than expensive chromatography, representing a paradigm shift in cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Silyl-Protection and Recrystallization
The core of this technological advancement rests on the precise manipulation of the amine functionality using triisopropylchlorosilane (TIPSCl). In the presence of triethylamine and ammonia, the primary amine of the 4-amino-5-hexenoic acid intermediate is selectively silylated. This transformation serves a dual purpose: firstly, it masks the nucleophilic character of the nitrogen, preventing intermolecular condensation or polymerization during subsequent handling and concentration steps. Secondly, and perhaps more critically for purification, the bulky triisopropyl group significantly alters the solubility profile of the molecule. The resulting triisopropylsilyl 4-aminohex-5-enoate exhibits distinct solubility characteristics in organic solvents compared to its unprotected counterpart and common impurities. This differential solubility is exploited in the recrystallization step, where a specific mixture of chloroform and methanol (1:7 v/v) is used as the primary solvent, followed by the anti-solvent addition of cyclohexane. This ternary system creates a supersaturation environment that favors the nucleation of the desired product while keeping impurities in solution, effectively acting as a thermodynamic purification engine.
Furthermore, the control of crystallization kinetics plays a pivotal role in the final quality of the high-purity API intermediate. The patent specifies a controlled cooling rate of 10-15°C/min down to 40°C, followed by slow stirring at low speeds (20-30 rpm). This careful management of the metastable zone width ensures the growth of large, well-defined crystals with minimal solvent inclusion and defect sites. Rapid cooling or aggressive agitation could trap mother liquor containing impurities within the crystal lattice, thereby defeating the purpose of the recrystallization. By adhering to these specific kinetic parameters, the process ensures that the crystal lattice forms with high selectivity, excluding structurally similar byproducts that might have survived the initial chemical synthesis. This level of control over the solid-state chemistry is what enables the reported HPLC purity levels of over 99.9%, setting a new benchmark for the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 4-Amino-5-hexenoic Acid Efficiently
The synthesis of this critical epilepsy drug intermediate requires strict adherence to the optimized reaction conditions and purification protocols outlined in the patent data. The process integrates standard organic transformations with specialized crystallization techniques to ensure maximum yield and purity. Operators must pay close attention to the stoichiometry of the silylating agent and the precise temperature gradients during the recrystallization phase to replicate the high-quality results demonstrated in the examples. For a detailed breakdown of the operational parameters, reagents, and workup procedures required to implement this technology in a pilot or production plant, please refer to the standardized synthesis guide below.
- Condense diethyl malonate with trans-1,4-dichloro-2-butene using sodium ethoxide to form diethyl 2-vinylcyclopropane-1,1-dicarboxylate.
- React the cyclopropane derivative with ammonia and formamide under pressure to generate the pyrrolidine-3-amide intermediate.
- Hydrolyze the amide under acidic conditions to obtain crude 4-amino-5-hexenoic acid, then protect with TIPSCl.
- Purify the silyl-protected intermediate via recrystallization in a chloroform/methanol/cyclohexane system before final hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification train. By achieving high purity through crystallization rather than column chromatography or multiple distillation steps, the process significantly reduces the consumption of silica gel, solvents, and energy. This reduction in processing complexity translates directly into lower manufacturing costs and a smaller environmental footprint, aligning with modern green chemistry initiatives. Additionally, the use of commodity starting materials like diethyl malonate and trans-1,4-dichloro-2-butene ensures a stable and cost-effective raw material supply base, mitigating the risks associated with sourcing exotic or volatile precursors. The robustness of the process also implies a higher success rate in production batches, reducing the incidence of out-of-specification material that can disrupt inventory planning.
- Cost Reduction in Manufacturing: The elimination of complex purification steps such as preparative HPLC or repeated recrystallizations of the free acid significantly lowers the operational expenditure per kilogram of product. The ability to purify the intermediate at the silyl-ester stage, where solubility differences are maximized, avoids the losses typically associated with purifying zwitterionic amino acids. This efficiency gain allows for a more competitive pricing structure for the final API, providing a strategic advantage in cost-sensitive markets. Furthermore, the recovery and recycling of solvents like chloroform and methanol in this closed-loop crystallization process further enhance the economic viability of the route.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals for the initial cyclopropanation step ensures that production is not bottlenecked by the availability of niche reagents. The process conditions, involving standard reflux and pressure reactions, are compatible with existing multipurpose reactor infrastructure found in most fine chemical plants. This compatibility reduces the need for capital-intensive equipment upgrades, allowing for rapid deployment and scaling of production capacity to meet surging demand for antiepileptic medications. The stability of the silyl-protected intermediate also offers logistical flexibility, allowing for safer storage and transport between different manufacturing sites if a distributed production model is required.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations that are easily transferred from laboratory to kiloton scale without significant re-optimization. The waste stream is primarily composed of aqueous salts and recoverable organic solvents, which are easier to treat and dispose of compared to the heavy metal waste or complex organic sludge generated by some transition-metal catalyzed alternatives. This cleaner waste profile simplifies regulatory compliance and reduces the costs associated with environmental remediation. The high atom economy of the initial cyclopropanation and the high yield of the recrystallization step contribute to an overall sustainable manufacturing process that meets the rigorous ESG criteria of modern pharmaceutical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Vigabatrin synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and advantages of the method. Understanding these details is crucial for technical teams evaluating the integration of this process into their existing manufacturing portfolios.
Q: How does the new silyl-protection method improve Vigabatrin purity compared to prior art?
A: The introduction of a triisopropylsilyl (TIPS) protecting group allows for a highly specific recrystallization step that removes structural isomers and oligomeric impurities which are difficult to eliminate in conventional acid-hydrolysis-only routes.
Q: What are the critical process parameters for the recrystallization step?
A: The patent specifies a precise solvent system of chloroform and methanol at a 1:7 volume ratio, followed by the controlled addition of cyclohexane. Temperature control during cooling (10-15°C/min) is also vital for crystal habit and purity.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route utilizes commodity chemicals like diethyl malonate and avoids exotic catalysts. The unit operations (reflux, crystallization, filtration) are standard in fine chemical manufacturing, ensuring excellent scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vigabatrin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-purity intermediates for life-saving neurological treatments. Our team of expert chemists has thoroughly analyzed the pathway described in CN110713440B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art reactors capable of handling the pressure and temperature requirements of the ammonolysis and cyclopropanation steps, alongside advanced crystallization suites designed to replicate the precise solvent gradients necessary for achieving 99.9% purity. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize HPLC and NMR to validate every batch against the highest international pharmacopeia standards.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced synthesis route for your Vigabatrin supply needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and regional logistics. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of chemical excellence and reliability.
