Revolutionizing Norspeltol Production: A Safe, Scalable Route for Pharmaceutical Intermediates
Revolutionizing Norspeltol Production: A Safe, Scalable Route for Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust manufacturing processes that balance high purity with operational safety, particularly for complex small molecule activators like Norspeltol (also known as Rosiptor). Patent CN111056962A, filed in April 2020, introduces a groundbreaking preparation method for Norspeltol and its acetate salt that fundamentally reengineers the synthetic pathway to eliminate severe safety hazards. This technical disclosure addresses the critical limitations of previous methodologies by replacing explosive reagents with safer alternatives and removing tedious purification bottlenecks. For R&D directors and procurement specialists, this patent represents a pivotal shift towards a more sustainable and economically viable supply chain for this potent SHIP1 inhibitor used in treating bladder pain syndrome. The following analysis dissects the chemical innovations and their profound implications for industrial scale-up.
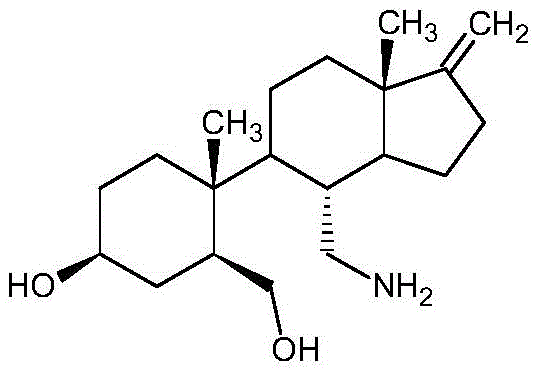
Norspeltol, chemically defined as 5-(1β-methyl-4β-hydroxy-2β-hydroxymethylcyclohexyl)-4α-aminomethyl-7aβ-methyl-1-methyleneoctahydroindan, serves as a critical active pharmaceutical ingredient precursor. Its therapeutic potential in modulating inflammatory responses necessitates a supply chain capable of delivering high-purity material consistently. The structural complexity, featuring multiple chiral centers and sensitive functional groups, traditionally demanded aggressive reagents that compromised process safety. The new methodology outlined in the patent data offers a streamlined approach that maintains stereochemical integrity while drastically simplifying the operational workflow, making it an ideal candidate for reliable pharmaceutical intermediate supplier partnerships focused on long-term stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis routes, such as those disclosed in earlier patents like CN102001916, relied heavily on hazardous chemistry that is increasingly untenable for modern GMP manufacturing environments. As illustrated in the historical reaction scheme, the conventional pathway utilized sodium azide for the introduction of the nitrogen functionality, a reagent notorious for its toxicity and potential to form explosive metal azides upon contact with heavy metals in piping systems. Furthermore, the reduction of the resulting azide intermediate required lithium aluminum hydride (LiAlH4), a pyrophoric substance that demands rigorous exclusion of moisture and specialized handling equipment, significantly inflating capital expenditure and operational risk. Beyond safety concerns, these legacy methods suffered from poor scalability due to a reliance on column chromatography for purification at multiple stages, creating a bottleneck that limits throughput and generates excessive solvent waste, thereby hindering cost reduction in API manufacturing.
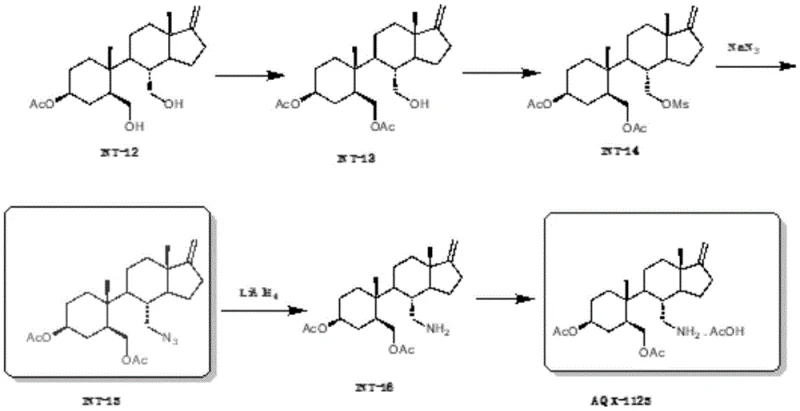
The Novel Approach
The innovative process detailed in CN111056962A circumvents these pitfalls through a strategic redesign of the functional group interconversions. Instead of the dangerous azide displacement, the new route employs a mesylate or tosylate leaving group which undergoes direct nucleophilic substitution with ammonia. This transformation is executed using either concentrated ammonia water or pressurized ammonia gas, reagents that are far more manageable on a multi-ton scale. Crucially, the patent emphasizes that the entire synthesis avoids column chromatography, relying instead on crystallization and filtration to achieve high purity. This shift not only enhances the safety profile by removing explosive and pyrophoric reagents but also dramatically improves the mass balance and throughput of the production line. The result is a process that is inherently safer, cleaner, and significantly more aligned with the principles of green chemistry and industrial feasibility.
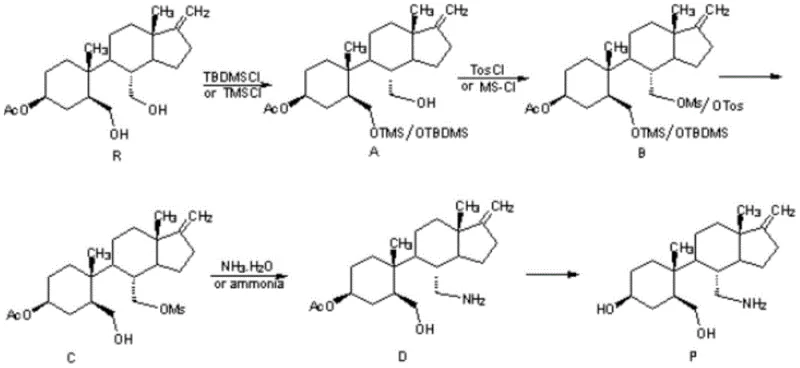
Mechanistic Insights into Ammonolysis and Protecting Group Strategy
The success of this optimized synthesis hinges on a sophisticated protecting group strategy that allows for selective functionalization without compromising the delicate stereochemistry of the carbocyclic framework. The process initiates with the selective protection of the 3-position hydroxyl group on the starting material (Compound R) using silyl chlorides such as tert-butyldimethylsilyl chloride (TBDMSCl) or trimethylchlorosilane (TMSCl). This step is critical because it masks the secondary alcohol, preventing it from competing in the subsequent activation step. Following this, the primary hydroxyl group at the 4-position is activated by conversion into a sulfonate ester, specifically a mesylate or tosylate, using methanesulfonyl chloride or p-toluenesulfonyl chloride in the presence of a base like triethylamine. This activation transforms a poor leaving group (hydroxyl) into an excellent one, priming the molecule for the pivotal amination reaction.
The core mechanistic breakthrough occurs during the ammonolysis step, where the activated sulfonate intermediate (Compound C) reacts with ammonia to install the primary amine. Unlike the SN2 displacement with azide which requires a subsequent energetic reduction, this direct substitution yields the amine (Compound D) in a single operation. The patent specifies that this reaction can be driven to completion using pressurized ammonia gas (0.2-5 MPa) or concentrated aqueous ammonia at elevated temperatures (30-100°C). Following amination, the silyl protecting group is removed, and any remaining esters are hydrolyzed under basic conditions (pH 12-14) using sodium hydroxide to reveal the final free base. This sequence ensures that the final product, Norspeltol, is obtained with high optical purity and minimal impurities, as evidenced by the HPLC profiles showing purity levels exceeding 99% after simple recrystallization.
How to Synthesize Norspeltol Efficiently
The synthesis of Norspeltol via this patented route involves a logical sequence of protection, activation, substitution, and deprotection steps that are amenable to standard reactor configurations. The process begins with the silylation of the starting diol, followed by selective mesylation of the primary alcohol. The subsequent ammonolysis is the key step that differentiates this route from legacy methods, offering a safer alternative to azide chemistry. Finally, global deprotection and hydrolysis yield the target molecule, which can be isolated as a free base or converted to its acetate salt for improved stability. For a detailed breakdown of the specific reaction conditions, stoichiometry, and workup procedures required to execute this synthesis in a pilot or production plant, please refer to the standardized technical guide below.
- Protect the 3-position hydroxyl of the starting material (Compound R) using tert-butyldimethylsilyl chloride or trimethylchlorosilane to form Compound A.
- React the 4-position hydroxyl of Compound A with p-toluenesulfonyl chloride or methanesulfonyl chloride to generate the leaving group in Compound B.
- Remove the 3-hydroxyl protecting group from Compound B to obtain Compound C, preparing it for amination.
- Perform nucleophilic substitution on Compound C using concentrated ammonia water or ammonia gas to generate the amine intermediate Compound D.
- Hydrolyze Compound D under alkaline conditions to yield the final Norspeltol free base, optionally converting it to the acetate salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers tangible benefits that extend beyond mere chemical elegance. The elimination of column chromatography is perhaps the most significant economic driver, as chromatographic separations are notoriously resource-intensive, requiring vast quantities of silica gel and organic solvents, along with significant downtime for column packing and elution. By replacing this with crystallization-based purification, the process achieves a substantial reduction in raw material costs and waste disposal fees. Furthermore, the removal of lithium aluminum hydride and sodium azide simplifies the regulatory compliance landscape, reducing the need for specialized hazardous waste permits and expensive safety infrastructure, which translates directly into lower overheads for the manufacturing facility and potentially lower pricing for the downstream buyer.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the simplification of unit operations. By avoiding the use of expensive and hazardous reagents like lithium aluminum hydride, the process eliminates the need for specialized quenching protocols and the associated safety monitoring systems. Additionally, the ability to telescope reactions—such as proceeding from the protection step directly to activation without intermediate isolation—reduces the number of discrete processing steps. This streamlining minimizes labor hours and equipment occupancy time, leading to a more efficient utilization of plant assets. The cumulative effect is a manufacturing process that delivers high-purity pharmaceutical intermediates at a significantly reduced cost basis compared to traditional azide-based routes.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of specialized or regulated reagents. Sodium azide, for instance, is subject to strict controls in many jurisdictions due to its toxicity, which can lead to procurement delays and supply disruptions. By substituting this with commodity chemicals like ammonia and sulfonyl chlorides, the new route insulates the supply chain from regulatory bottlenecks. Moreover, the robustness of the crystallization steps ensures consistent product quality batch-to-batch, reducing the risk of failed campaigns that could interrupt the supply of critical API precursors. This reliability is essential for maintaining the production schedules of downstream drug manufacturers who depend on just-in-time delivery of high-quality intermediates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process represents a major advancement. The absence of azides removes the risk of forming explosive heavy metal salts in wastewater streams, a common concern in pharmaceutical manufacturing. Similarly, avoiding pyrophoric reagents reduces the fire hazard profile of the plant. The process generates less hazardous waste, aligning with increasingly stringent global environmental regulations. This 'green' profile not only facilitates easier permitting for new production lines but also enhances the corporate sustainability metrics of the supply chain partners. The scalability is further supported by the use of common solvents like methyl tert-butyl ether and dichloromethane, which are readily available in bulk quantities globally.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Norspeltol using this optimized methodology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders assess the feasibility of integrating this intermediate into their broader drug development pipelines.
Q: Why is the new synthesis route for Norspeltol considered safer than conventional methods?
A: The novel process described in patent CN111056962A eliminates the use of sodium azide and lithium aluminum hydride, which are highly hazardous reagents posing significant explosion and handling risks in traditional synthetic pathways.
Q: How does this process improve cost efficiency for pharmaceutical manufacturers?
A: By removing the need for column chromatography purification at every step and utilizing simple crystallization and filtration techniques, the process drastically reduces solvent consumption, operational time, and labor costs associated with complex purification.
Q: What represents the key chemical transformation in this optimized route?
A: The critical innovation is the direct installation of the aminomethyl group via ammonolysis of a mesylate or tosylate intermediate, replacing the dangerous azide substitution and subsequent reduction steps found in prior art.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Norspeltol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of novel therapeutics depends on a partner who can translate complex patent chemistry into reliable commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to delivering high-purity Norspeltol and its acetate salt that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our infrastructure is designed to handle the specific solvent systems and reaction conditions required by this patented route, guaranteeing a consistent supply of this critical pharmaceutical intermediate.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By leveraging our expertise, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to request specific COA data and route feasibility assessments to validate the superior quality and economic advantages of our manufacturing process. Let us collaborate to bring safer, more effective anti-inflammatory treatments to the market faster.
