Advanced Biocatalytic Route for High-Purity Chiral Cis-Beta-Aryl-Beta-Hydroxy-Alpha-Amino Esters
Advanced Biocatalytic Route for High-Purity Chiral Cis-Beta-Aryl-Beta-Hydroxy-Alpha-Amino Esters
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable pathways for constructing complex chiral scaffolds essential for active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in Chinese Patent CN113249417B, which discloses a highly efficient preparation method for chiral cis-β-aryl-β-hydroxy-α-amino ester compounds. These compounds serve as critical structural motifs in a wide array of therapeutic agents, including antibiotics like thiamphenicol and florfenicol, as well as other bioactive molecules such as droxidopa and eliglustat. The core innovation lies in the utilization of a specific carbonyl reductase, designated as KRED-Bt, derived from Bacillus thuringiensis, which catalyzes the asymmetric reduction of β-aryl-β-oxo-α-aminopropionate precursors. This biocatalytic approach addresses long-standing challenges in stereoselective synthesis, offering a pathway that is not only environmentally benign but also capable of operating at high substrate concentrations, making it exceptionally attractive for industrial adoption by a reliable pharmaceutical intermediate supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral cis-β-aryl-β-hydroxy-α-amino esters has relied heavily on chemical catalysis, often employing transition metals such as ruthenium or rhodium to facilitate dynamic kinetic resolution. While effective in laboratory settings, these chemical methodologies present substantial drawbacks for large-scale manufacturing. The primary concern is the inevitable introduction of heavy metal residues, which necessitates rigorous and costly purification processes to meet stringent regulatory limits for pharmaceutical products. Furthermore, these metal-catalyzed reactions often require harsh conditions, including high pressures or temperatures, and may involve hazardous solvents, complicating waste management and increasing the overall environmental footprint. Although earlier biocatalytic attempts, such as the Merck-reported CDX-018 enzyme system shown in Reaction B, demonstrated high selectivity, there remained a critical need for enzymes with broader substrate tolerance and higher operational stability to support diverse synthetic routes without compromising yield or purity.
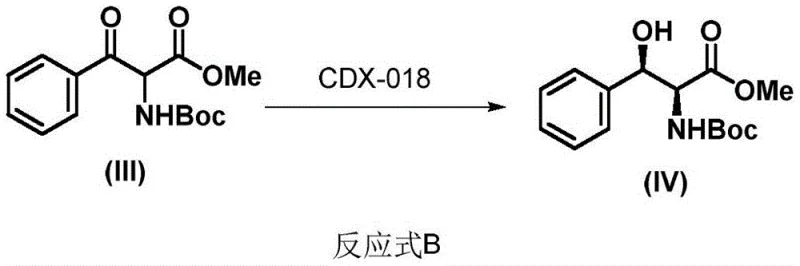
The Novel Approach
The novel methodology described in the patent overcomes these limitations by leveraging the unique catalytic properties of KRED-Bt. This enzyme exhibits remarkable promiscuity, successfully reducing a wide spectrum of β-keto ester substrates bearing various electron-withdrawing and electron-donating groups, such as methylsulfonyl, nitro, halogens, and trifluoromethyl moieties. As illustrated in the specific transformation of substrate IIa to product Ia, the process achieves outstanding yields and stereocontrol under mild aqueous conditions. The use of whole-cell supernatants or resting cells simplifies the catalyst preparation, avoiding the need for complex enzyme purification while maintaining high activity. This shift from precious metal catalysis to biocatalysis represents a paradigm shift in cost reduction in API manufacturing, as it replaces expensive metal ligands with renewable biological systems and utilizes glucose as a cheap hydrogen source, significantly lowering the raw material costs associated with cofactor regeneration.
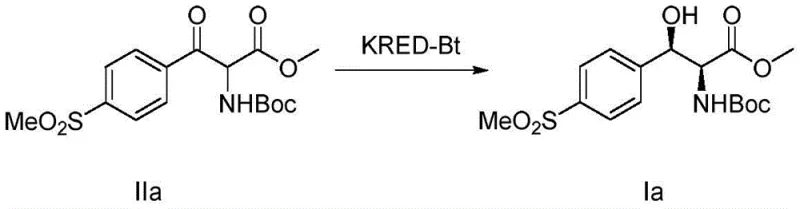
Mechanistic Insights into KRED-Bt Catalyzed Asymmetric Reduction
The heart of this technology is a sophisticated dual-enzyme cascade system that ensures both high conversion and perfect stereochemical fidelity. The primary catalyst, carbonyl reductase KRED-Bt, specifically recognizes the prochiral ketone substrate and delivers a hydride ion from the reduced cofactor NADPH to the carbonyl carbon. This step is highly stereoselective, favoring the formation of the syn-cis configuration with ee values consistently exceeding 99%. Crucially, the reaction does not consume stoichiometric amounts of the expensive cofactor. Instead, it employs a coupled enzyme system where glucose dehydrogenase (GDH) continuously regenerates NADPH from NADP+ by oxidizing glucose to gluconolactone. This creates a closed catalytic loop, as depicted in the reaction mechanism diagram, allowing the reaction to proceed efficiently with only catalytic amounts of the cofactor. This mechanism not only drives the thermodynamic equilibrium towards the product but also ensures that the process remains economically viable even at very large scales.
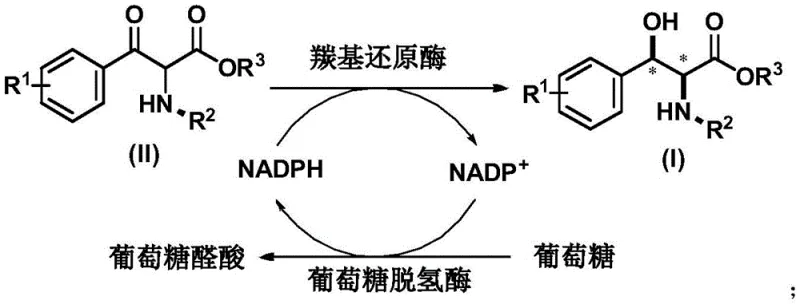
From an impurity control perspective, the enzymatic nature of the reaction provides inherent advantages over chemical reduction. Chemical reductants like sodium borohydride often lack the finesse to distinguish between similar functional groups, potentially leading to over-reduction or side reactions with other sensitive moieties on the aromatic ring. In contrast, the active site of KRED-Bt acts as a molecular sieve, sterically excluding unwanted reaction pathways and ensuring that only the target ketone is reduced. This high chemoselectivity minimizes the formation of by-products, resulting in a cleaner crude reaction profile. Consequently, downstream processing is simplified, requiring fewer chromatographic steps to achieve the high-purity chiral amino esters demanded by regulatory agencies. The ability to maintain such high purity profiles across diverse substrates, including those with bulky protecting groups like Boc or Cbz, underscores the robustness of this biocatalytic platform for producing high-purity OLED material precursors or pharmaceutical intermediates.
How to Synthesize Chiral Cis-Beta-Aryl-Beta-Hydroxy-Alpha-Amino Esters Efficiently
Implementing this biocatalytic route requires precise control over fermentation and reaction parameters to maximize the efficiency of the engineered biocatalysts. The process begins with the cultivation of recombinant E. coli strains harboring the specific genes for KRED-Bt and GDH, followed by induction to express the enzymes. The subsequent steps involve harvesting the biomass, preparing the enzyme sources (either as resting cells or cell-free supernatants), and setting up the reduction reaction in a buffered aqueous-organic solvent system. The patent details specific conditions regarding pH, temperature, and substrate loading that are critical for success. For R&D teams looking to replicate or adapt this chemistry, adhering to the standardized protocol is essential to achieve the reported high yields and stereoselectivity. The detailed standardized synthesis steps are provided in the guide below.
- Prepare engineered E. coli BL21(DE3) strains containing pET-28a-KRED-Bt and glucose dehydrogenase genes, inducing expression with IPTG at 15-25°C.
- Harvest cells via centrifugation, resuspend in phosphate buffer, and perform ultrasonic disruption to obtain enzyme-rich cell supernatants.
- Mix the KRED-Bt and GDH supernatants with the beta-keto ester substrate, glucose, and NADP+ cofactor in a buffered solvent system at 20-40°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this biocatalytic process offers tangible strategic benefits that extend beyond simple technical metrics. The elimination of transition metal catalysts fundamentally alters the cost structure of the synthesis. By removing the dependency on ruthenium or rhodium, manufacturers avoid the volatility associated with precious metal markets and the significant capital expenditure required for metal scavenging technologies. This leads to substantial cost savings in the overall production budget. Furthermore, the use of glucose as a co-substrate is a major economic driver; glucose is an abundant, renewable, and inexpensive commodity chemical, contrasting sharply with the costly silanes or borohydrides used in traditional chemical reductions. This shift ensures a more stable and predictable cost base for long-term supply contracts.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the high volumetric productivity enabled by the enzyme's tolerance to high substrate concentrations. The patent indicates that the system can operate effectively with substrate loadings up to 300 g/L. This high density means that for a given reactor volume, significantly more product can be generated per batch compared to dilute chemical processes. Higher throughput directly translates to lower fixed costs per kilogram of product, as utilities, labor, and equipment depreciation are spread over a larger output. Additionally, the simplified work-up procedure, which avoids complex metal removal steps, reduces the consumption of auxiliary chemicals and solvents, further driving down the variable costs associated with commercial scale-up of complex intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the biological components. The engineered E. coli strains are stable and can be stored for extended periods, ensuring a consistent supply of the biocatalyst. Unlike chemical catalysts that may suffer from batch-to-batch variability or degradation during shipping, the lyophilized or frozen cell preparations offer reliable performance. Moreover, the broad substrate scope of KRED-Bt means that a single biocatalytic platform can be adapted to produce a variety of structurally related intermediates. This flexibility allows manufacturers to respond quickly to changes in demand for different API precursors without needing to retool entire production lines or source new specialized chemical catalysts, thereby reducing lead time for high-purity intermediates.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is inherently safer and more compliant with modern environmental standards. The reaction operates at mild temperatures (20-40°C) and atmospheric pressure, reducing energy consumption and safety risks associated with high-pressure hydrogenation. The aqueous nature of the reaction medium minimizes the use of volatile organic compounds (VOCs), aligning with green chemistry principles and easing the burden on waste treatment facilities. The biodegradable nature of the enzyme and the non-toxic by-products (gluconolactone) simplify waste disposal. This environmental compatibility is increasingly critical for maintaining social licenses to operate and meeting the sustainability goals of multinational pharmaceutical partners, ensuring uninterrupted supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this KRED-Bt mediated synthesis. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is vital for making informed decisions about process adoption and vendor qualification.
Q: How does the KRED-Bt process eliminate heavy metal contamination risks?
A: Unlike traditional chemical methods relying on ruthenium or rhodium catalysts, this biocatalytic route uses recombinant enzymes (KRED-Bt and GDH). This completely removes the need for transition metals, thereby eliminating the costly and complex downstream purification steps required to meet strict ppm limits for heavy metals in pharmaceutical intermediates.
Q: What is the stereoselectivity performance of this enzymatic method?
A: The process demonstrates exceptional stereocontrol, consistently achieving enantiomeric excess (ee) values greater than 99% and diastereomeric ratios (dr) exceeding 99:1 across a broad range of substrates, including those with methylsulfonyl, nitro, and halogen substituents.
Q: Is this method suitable for high-concentration industrial production?
A: Yes, the patent explicitly supports high substrate loadings ranging from 1.0 g/L up to 300 g/L. This high volumetric productivity, combined with the use of inexpensive glucose for cofactor regeneration, makes the process highly economically viable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Cis-Beta-Aryl-Beta-Hydroxy-Alpha-Amino Ester Supplier
The technological potential of the KRED-Bt catalyzed route represents a significant opportunity for optimizing the supply chain of critical antibiotic and pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities, guaranteeing that every batch meets the exacting standards required for GMP manufacturing. We understand that consistency is key, and our process engineering team is dedicated to maintaining the high stereoselectivity and yield demonstrated in the patent literature.
We invite you to collaborate with us to leverage this advanced biocatalytic technology for your specific project needs. Whether you require a Customized Cost-Saving Analysis to evaluate the economic benefits of switching from chemical to enzymatic synthesis, or you need specific COA data to verify impurity profiles, our technical procurement team is ready to assist. Please contact us today to request route feasibility assessments and discuss how we can support your long-term supply goals with high-quality, sustainably produced chiral intermediates.
