Advanced Racemization Strategy for High-Purity Duloxetine Intermediate Manufacturing
Advanced Racemization Strategy for High-Purity Duloxetine Intermediate Manufacturing
The pharmaceutical industry continuously seeks methods to optimize the production of chiral active pharmaceutical ingredients (APIs), particularly for blockbuster drugs like Duloxetine, widely known under the trade name Cymbalta. A critical bottleneck in the synthesis of such molecules lies in the efficient preparation of their chiral intermediates, specifically S-(-)-3-(dimethylamino)-1-(2-thienyl)-1-propanol. Patent CN102285961B introduces a transformative methodology that addresses the inherent inefficiencies of traditional chiral resolution. By implementing a novel racemization cycle, this technology converts the unwanted R-enantiomer byproduct back into a usable racemic mixture, thereby breaking the 50% yield barrier typical of classical resolution techniques. This insight report analyzes the technical merits and commercial implications of this process for global procurement and R&D teams seeking reliable pharmaceutical intermediate suppliers.
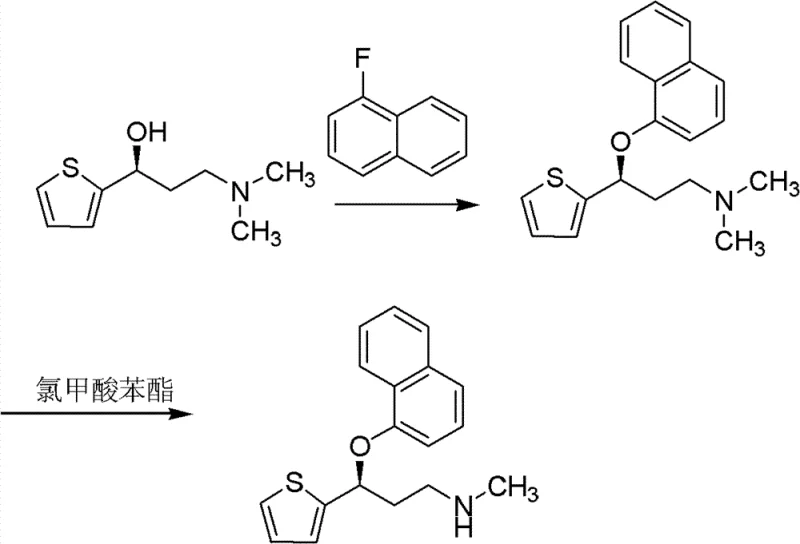
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral amines like the duloxetine intermediate has relied heavily on classical diastereomeric salt formation. While effective at separating enantiomers, this method suffers from a fundamental stoichiometric limitation: the maximum theoretical yield for the desired enantiomer is capped at 50%. The remaining 50%, which consists of the opposite enantiomer (in this case, the R-(+)-isomer), is traditionally treated as waste or sold at a significant discount for less valuable applications. This inefficiency not only doubles the raw material cost per unit of product but also generates substantial chemical waste, creating environmental liabilities and increasing disposal costs. For large-scale manufacturers, discarding half of the synthesized material represents a massive loss of atom economy and operational efficiency, making the final API significantly more expensive to produce.
The Novel Approach
The innovation detailed in the patent data proposes a closed-loop system that fundamentally alters the economics of chiral synthesis. Instead of discarding the R-(+)-3-(dimethylamino)-1-(2-thienyl)-1-propanol byproduct, the process subjects it to a specific acid-catalyzed racemization treatment. This chemical transformation effectively resets the stereochemistry of the unwanted isomer, converting it back into the racemic (±)-mixture. This regenerated racemate is then fed back into the resolution stage. As illustrated in the reaction scheme below, this cyclical utilization ensures that nearly all starting material is eventually converted into the desired S-enantiomer. This approach not only maximizes yield but also aligns with green chemistry principles by minimizing waste generation and reducing the overall consumption of chiral resolving agents.
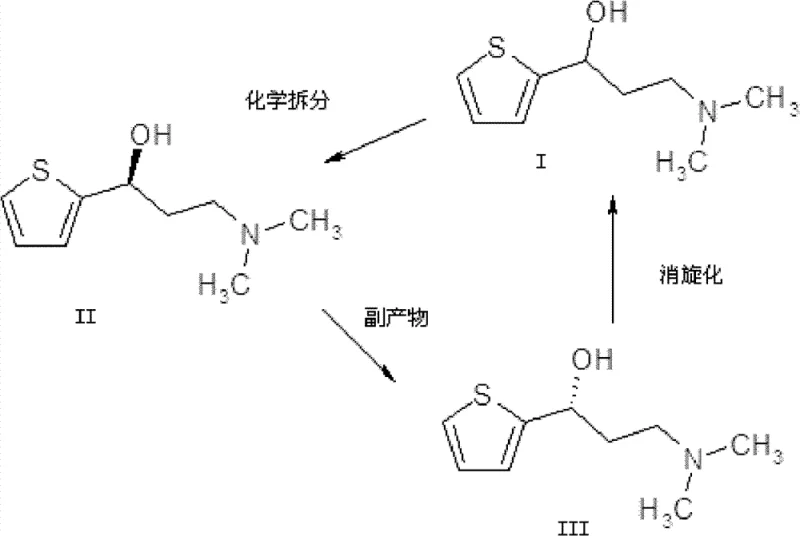
Mechanistic Insights into Acid-Catalyzed Racemization and Resolution
The core of this technology relies on two distinct chemical mechanisms working in tandem: selective crystallization and acid-catalyzed stereoinversion. In the resolution phase, chiral organic acids such as L-tartaric acid, L-2,3-dibenzoyl tartaric acid, or L-camphorsulfonic acid are employed. These acids form diastereomeric salts with the amine intermediate. Due to differences in solubility and crystal lattice energy, the salt of the desired S-enantiomer precipitates out of the solvent system (typically ethyl acetate or ethanol), while the R-enantiomer remains in the mother liquor. The patent highlights that optimizing the molar ratio of the resolving acid to the amine, preferably between 0.9:1 and 1.0:1, is critical for achieving high optical purity, with experimental results demonstrating ee values consistently above 99%.
The racemization mechanism involves heating the R-enantiomer in a mixture of lower aliphatic alcohols and carboxylic acids, such as butyric or propionic acid. Under reflux conditions (80°C to 130°C), the acidic medium facilitates the reversible formation of an achiral intermediate, likely involving an iminium ion or a stabilized carbocation species at the benzylic-like position adjacent to the thiophene ring. This temporary loss of chirality allows the molecule to re-form the C-N bond with equal probability for both spatial configurations upon cooling and workup. The use of simple carboxylic acids as catalysts is particularly advantageous as they are inexpensive, easy to remove via distillation, and do not introduce heavy metal contaminants, ensuring the final product meets stringent regulatory standards for pharmaceutical intermediates.
How to Synthesize S-(-)-3-(dimethylamino)-1-(2-thienyl)-1-propanol Efficiently
The synthesis protocol outlined in the patent provides a robust framework for scaling this process from laboratory to commercial production. The procedure begins with the dissolution of a chiral resolving agent in a suitable solvent, followed by the addition of the racemic amine to induce salt formation. After filtration and recrystallization to ensure high enantiomeric excess, the solid salt is neutralized to release the free base. Crucially, the mother liquor containing the unwanted isomer is not discarded but processed through the racemization reactor. Detailed standard operating procedures regarding temperature controls, reflux times, and pH adjustments during the extraction phases are essential for maintaining reproducibility. For a complete breakdown of the specific reaction parameters and workup instructions, please refer to the technical guide below.
- Perform chiral resolution on the racemic amine using L-tartaric acid or L-camphorsulfonic acid in a solvent system like ethyl acetate or ethanol to isolate the desired S-enantiomer salt.
- Collect the mother liquor containing the unwanted R-enantiomer and subject it to acid-catalyzed racemization using carboxylic acids such as butyric or propionic acid under reflux conditions.
- Recycle the regenerated racemic mixture back into the resolution step to maximize overall yield and minimize chemical waste generation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this racemization-recycling technology offers profound strategic benefits beyond simple chemistry. The primary advantage lies in the drastic reduction of raw material intensity. By循环利用 (recycling) the byproduct, the effective consumption of the starting racemic amine is nearly halved compared to traditional linear processes. This directly translates to significant cost reductions in pharmaceutical intermediate manufacturing, as the cost of goods sold (COGS) is heavily influenced by material inputs. Furthermore, the reliance on expensive chiral resolving agents is optimized, as the same amount of resolving agent can theoretically process double the amount of final product over time, improving the return on investment for these specialized reagents.
- Cost Reduction in Manufacturing: The elimination of waste disposal costs for the R-enantiomer and the reduced purchase volume of starting materials create a leaner cost structure. Since the process avoids the need for asymmetric synthesis which often requires costly chiral catalysts or enzymes, the capital expenditure for catalyst recovery systems is also avoided. This qualitative shift in process efficiency allows suppliers to offer more competitive pricing structures for high-volume contracts without compromising margin integrity.
- Enhanced Supply Chain Reliability: By maximizing the yield from every batch of racemic starting material, manufacturers reduce their dependency on external suppliers for bulk precursors. This self-sufficiency mitigates risks associated with raw material shortages or price volatility in the upstream chemical market. Additionally, the simplified workflow, which integrates resolution and racemization in a continuous loop, reduces the complexity of the supply chain, leading to more predictable lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The reagents used, such as ethanol, ethyl acetate, and simple carboxylic acids, are commodity chemicals with well-established global supply chains, ensuring scalability from pilot plants to multi-ton production. From an environmental perspective, the reduction in chemical waste aligns with increasingly strict global regulations on pharmaceutical effluent. This proactive approach to waste minimization reduces the burden on wastewater treatment facilities and lowers the risk of regulatory non-compliance, securing the long-term viability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral resolution and racemization technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the process feasibility. Understanding these details is crucial for R&D teams assessing the transferability of this method to their existing infrastructure.
Q: How does the racemization process improve the theoretical yield of chiral resolution?
A: Traditional chiral resolution is limited to a maximum theoretical yield of 50% because one enantiomer is discarded as waste. By chemically converting the unwanted R-enantiomer back into a racemic mixture through acid-catalyzed racemization, the material can be re-subjected to the resolution process. This cyclic approach allows the theoretical yield to approach 100% over multiple cycles, drastically improving atom economy.
Q: What specific reagents are recommended for the racemization step in this patent?
A: The patent specifies using a combination of lower aliphatic alcohols (such as ethanol) and aliphatic carboxylic acids (such as butyric acid or propionic acid) as the reaction medium. The mixture is heated to reflux temperatures, typically between 80°C and 130°C, to facilitate the stereochemical inversion required to regenerate the racemic amine.
Q: What level of optical purity can be achieved with this resolution method?
A: According to the experimental data provided in the patent, the resolution process utilizing chiral organic acids like L-2,3-dibenzoyl tartaric acid or L-camphorsulfonic acid consistently achieves optical purities (ee values) exceeding 99%. Recrystallization of the intermediate salts further ensures that the final free base meets stringent pharmaceutical specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-(-)-3-(dimethylamino)-1-(2-thienyl)-1-propanol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the efficient production of chiral intermediates is the cornerstone of modern antidepressant therapy. Our technical team has extensively analyzed the racemization strategies described in patent CN102285961B and possesses the expertise to implement these advanced recycling loops at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and uninterrupted supply of critical materials. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee ee values exceeding 99% for every batch released.
We invite global partners to collaborate with us to leverage these cost-saving technological advancements. By integrating this efficient racemization cycle into our production schedule, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain for duloxetine intermediates is both economically optimized and technically robust.
