Advanced Manufacturing of Neratinib Impurity G via Novel Quaternary Ammonium Route
Advanced Manufacturing of Neratinib Impurity G via Novel Quaternary Ammonium Route
The pharmaceutical industry's relentless pursuit of quality control has placed impurity profiling at the forefront of drug development, particularly for potent kinase inhibitors like Neratinib. As detailed in patent CN111995618B, a groundbreaking preparation method for Neratinib Impurity G has been established, addressing critical bottlenecks in reference standard manufacturing. This technology leverages a strategic four-step synthesis starting from inexpensive N,N-dimethylamino-trans-crotonic acid, effectively bypassing the thermodynamic limitations of previous methods. By meticulously coordinating catalyst selection, solvent systems, and reaction temperatures, this process not only prevents the hydrolysis of the quinoline ring side chain but also achieves a target product yield exceeding 44%. Furthermore, the methodology ensures a final purity of greater than 99.0% without the necessity for resource-intensive column chromatography, marking a significant leap forward in the efficient production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Neratinib Impurity G relied heavily on using Neratinib Impurity B as the primary starting material, a approach fraught with significant chemical and economic inefficiencies. In these legacy processes, the equivalent ratio of Impurity B had to be drastically reduced, often to as low as 0.3 times, to mitigate side reactions, yet the impurity inevitably remained in the reaction solution due to dynamic equilibrium. This equilibrium involves the hydrolysis of the side chain amide in Impurity G, which regenerates Impurity B, thereby capping the maximum theoretical yield and complicating isolation. Consequently, manufacturers were forced to employ column chromatography for separation, a technique that is notoriously difficult to scale, consumes vast quantities of organic solvents, and introduces variability in batch-to-batch consistency. These factors collectively resulted in high production costs and extended lead times, making the reliable supply of this critical impurity standard a persistent challenge for quality control laboratories globally.
The Novel Approach
In stark contrast, the innovative pathway disclosed in the patent constructs the molecule from the ground up using N,N-dimethylamino-trans-crotonic acid, completely circumventing the hydrolysis equilibrium that plagues the traditional route. This de novo synthesis strategy allows for precise control over the formation of the amide bond, ensuring that the side chain remains intact throughout the reaction sequence. The process is designed to be operationally simple, utilizing mild reaction conditions that do not require specialized large-scale equipment, thus enhancing its adaptability for industrial application. By eliminating the need for column purification, the method drastically reduces solvent consumption and processing time, directly translating to a more sustainable and cost-effective manufacturing profile. The ability to achieve high purity (>99.0%) through simple filtration and recrystallization underscores the robustness of this chemical design, offering a scalable solution for the global supply of Neratinib impurities.
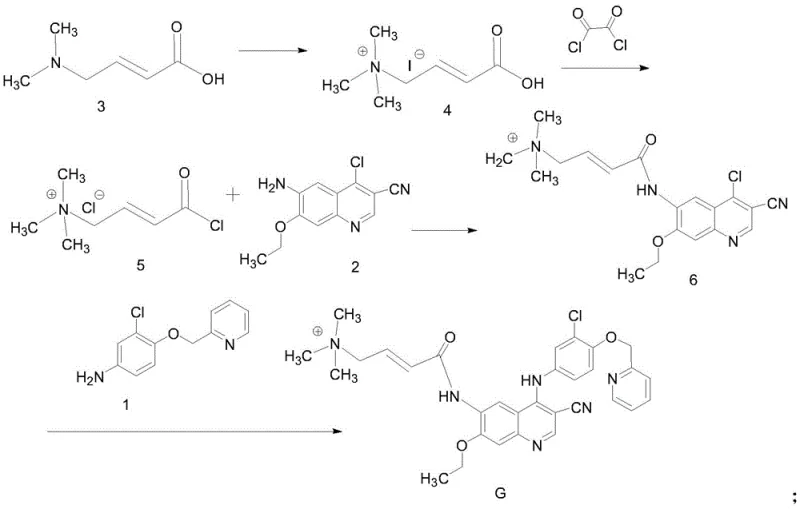
Mechanistic Insights into Quaternary Ammonium Mediated Amidation
The core of this synthetic breakthrough lies in the activation of the crotonic acid derivative through quaternization, which fundamentally alters the reactivity of the carboxylic acid group. The initial step involves the reaction of N,N-dimethylamino-trans-crotonic acid with methyl iodide in anhydrous THF over a period of 36 to 50 hours at room temperature. This prolonged reaction time ensures the complete conversion of the tertiary amine into a quaternary ammonium salt (Compound 4), which serves as a stable precursor for subsequent activation. The presence of the positively charged nitrogen atom exerts a strong electron-withdrawing effect, facilitating the subsequent nucleophilic attack by the chlorinating agent. This mechanistic nuance is critical, as it primes the molecule for efficient conversion into the acid chloride without degrading the sensitive alkene moiety, setting the stage for high-yield amidation in later steps.
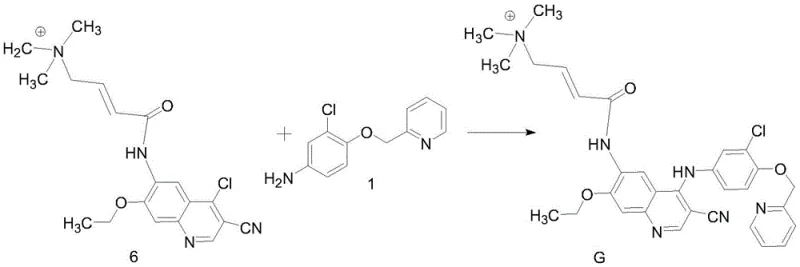
Following activation with oxalyl chloride at low temperatures (-5°C to 0°C), the resulting acid chloride (Compound 5) undergoes coupling with the quinoline amine (Compound 2) in N-methylpyrrolidone (NMP). The final and perhaps most critical transformation is the coupling of the intermediate (Compound 6) with the aniline derivative (Compound 1). This step utilizes zinc nitrate as a catalyst under reflux in acetonitrile for 4 to 6 hours. The zinc ion likely acts as a Lewis acid, coordinating with the carbonyl oxygen or the amine nitrogen to lower the activation energy for the nucleophilic aromatic substitution or condensation. This catalytic intervention is pivotal; it drives the reaction to completion while suppressing potential side reactions that could lead to hydrolysis or polymerization. The result is a clean formation of the final Impurity G structure, which precipitates out of the solution as a yellow solid, allowing for easy isolation via filtration.
How to Synthesize Neratinib Impurity G Efficiently
The execution of this synthesis requires strict adherence to anhydrous conditions and precise temperature control to maximize the yield of the quaternary intermediate and prevent premature hydrolysis. The process is divided into four distinct operational stages, beginning with the quaternization in THF, followed by acid chloride formation, amidation with the quinoline core, and finally the zinc-catalyzed coupling. Each step builds upon the purity of the previous intermediate, emphasizing the importance of thorough washing and filtration protocols, particularly the use of acetone and ethanol to remove residual salts and solvents. For laboratory and pilot-scale teams aiming to replicate this high-purity standard, the detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety.
- Quaternization of N,N-dimethylamino-trans-crotonic acid with methyl iodide in anhydrous THF at room temperature for 36-50 hours to form the quaternary ammonium salt intermediate.
- Activation of the carboxylic acid group using oxalyl chloride at low temperature (-5°C to 0°C) to generate the reactive acid chloride species.
- Coupling the activated acid chloride with the quinoline amine derivative (Compound 2) in NMP solvent to form the amide linkage (Compound 6).
- Final condensation with the aniline derivative (Compound 1) using zinc nitrate as a catalyst under reflux in acetonitrile to yield the target impurity G.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from equilibrium-limited synthesis to this linear, high-yield pathway represents a substantial opportunity for cost optimization and risk mitigation. The elimination of column chromatography is not merely a technical improvement but a major economic driver, as it removes one of the most expensive and time-consuming unit operations in fine chemical manufacturing. This simplification allows for faster batch turnover and significantly reduces the volume of hazardous organic solvents required for purification, aligning with increasingly stringent environmental regulations. Furthermore, the use of commodity chemicals like N,N-dimethylamino-trans-crotonic acid and methyl iodide ensures a stable and predictable raw material supply chain, insulating production from the volatility often associated with complex, custom-synthesized starting materials.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the drastic simplification of the downstream processing workflow. By achieving >99.0% purity through crystallization and filtration rather than chromatography, manufacturers can eliminate the capital expenditure associated with large chromatography columns and the operational expenditure of high-grade silica gel and solvents. Additionally, the avoidance of Impurity B as a starting material removes the cost penalty of purchasing a high-value degradation product to make another degradation product, replacing it with low-cost bulk chemicals. This fundamental shift in raw material strategy, combined with reduced solvent recovery costs, results in a significantly lower cost of goods sold (COGS) for the final impurity standard.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on scarce or difficult-to-source intermediates, but this method utilizes widely available industrial chemicals that are produced at multi-ton scales globally. The robustness of the reaction conditions, which do not require cryogenic temperatures or high-pressure vessels, means that production can be easily transferred between different manufacturing sites without extensive re-validation. This flexibility ensures that supply chains remain resilient against regional disruptions or equipment failures. Moreover, the high yield (>44% overall from the key intermediate) means that less raw material is needed to produce the same amount of final product, further securing supply availability even during periods of raw material tightness.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a greener alternative to traditional methods by minimizing waste generation. The absence of column chromatography significantly reduces the volume of spent silica and solvent mixtures that require disposal or energy-intensive distillation for recycling. The reaction conditions are mild, operating mostly at room temperature or moderate reflux, which lowers the energy footprint of the manufacturing process. These factors make the technology highly scalable, allowing producers to move from kilogram to multi-ton production with minimal environmental impact, thereby meeting the sustainability goals of modern pharmaceutical supply chains while maintaining regulatory compliance.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for quality assurance teams and regulatory affairs professionals who must validate the source of their reference standards. The following questions address common inquiries regarding the stability, purity, and scalability of the Neratinib Impurity G produced via this patented method. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide accurate guidance for procurement and R&D decision-making.
Q: Why is the new synthesis route superior to using Neratinib Impurity B as a starting material?
A: The conventional method using Impurity B suffers from dynamic equilibrium issues where the side chain amide hydrolyzes back to Impurity B, leading to low yields and requiring difficult column chromatography. The new route avoids this hydrolysis entirely by building the side chain from scratch.
Q: What purity levels can be achieved with this patented method?
A: The patented process achieves a purity of greater than 99.0% without the need for column chromatography purification, significantly simplifying the downstream processing and reducing solvent waste.
Q: What is the role of Zinc Nitrate in the final step?
A: Zinc nitrate acts as a Lewis acid catalyst in the final coupling step between Compound 6 and Compound 1. It facilitates the formation of the aniline-quinoline bond under reflux conditions while minimizing side reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Neratinib Impurity G Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your drug development pipeline depends on the quality and availability of critical impurity standards. Our technical team has extensively analyzed the pathway described in CN111995618B and possesses the expertise to implement this advanced synthesis at commercial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs capable of verifying the stringent purity specifications required for regulatory submissions, guaranteeing that every batch of Neratinib Impurity G meets the >99.0% purity benchmark without compromise.
We invite you to collaborate with us to optimize your supply chain for Neratinib-related materials. By leveraging our manufacturing capabilities, you can secure a stable source of high-quality intermediates while benefiting from our process optimization expertise. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our production of Neratinib Impurity G can support your quality control and regulatory objectives efficiently.
