Advanced Enzymatic Synthesis of Gemcitabine Intermediate for Scalable Pharmaceutical Manufacturing
Advanced Enzymatic Synthesis of Gemcitabine Intermediate for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry is currently witnessing a paradigm shift towards greener, more efficient synthetic routes for critical oncology intermediates, driven by the urgent need for cost-effective and sustainable manufacturing processes. A groundbreaking development in this sector is detailed in patent CN113717996B, which discloses a novel biosynthesis method for producing 2-deoxy-2,2-difluoro-3,5-dibenzoyl-D-ribofuranose, a pivotal precursor for the anticancer drug Gemcitabine hydrochloride. This technology leverages a specific CIR enzyme to catalyze the reduction of a ketone precursor, offering a transformative alternative to traditional chemical methods that have long plagued manufacturers with safety hazards and environmental burdens. By replacing hazardous reagents with a biocatalytic system, this innovation not only enhances the purity and yield of the intermediate but also aligns perfectly with modern green chemistry principles, presenting a compelling value proposition for global supply chains seeking reliability and compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-deoxy-2,2-difluoro-3,5-dibenzoyl-D-ribofuranose has relied heavily on chemical reduction strategies that introduce significant operational risks and cost inefficiencies into the production line. The most common prior art methods utilize palladium-carbon catalytic hydrogenation or strong chemical reducing agents like lithium aluminum hydride (LiAlH4) and Red-Al. The palladium-carbon route, while effective, suffers from the high cost of precious metal catalysts and poses severe safety risks associated with high-pressure hydrogenation processes, which require specialized explosion-proof equipment and rigorous safety protocols. Alternatively, the use of lithium aluminum hydride introduces extreme hazards due to its flammability and explosiveness upon contact with moisture, creating a dangerous working environment and generating complex waste streams that are difficult and costly to treat. Furthermore, these chemical methods often struggle with selectivity, leading to lower yields and the formation of impurities that complicate downstream purification, ultimately inflating the cost of goods sold and extending production lead times.
The Novel Approach
In stark contrast to these hazardous chemical pathways, the novel biosynthetic approach described in the patent utilizes a highly specific CIR enzyme to achieve the reduction under exceptionally mild and safe conditions. This enzymatic method operates in a benign alcohol solvent system, such as isopropanol or ethanol, at ambient temperatures ranging from 20°C to 30°C, completely eliminating the need for high-pressure equipment or pyrophoric reagents. The transition from a ketone structure to the desired alcohol configuration is achieved with remarkable precision, as illustrated by the structural comparison below, which highlights the specific conversion of the carbonyl group at the C1 position without disturbing the sensitive fluorine substituents or benzoyl protecting groups.
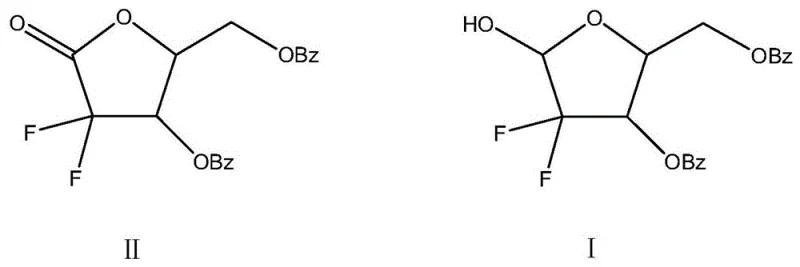
This fundamental shift in reaction mechanism not only mitigates safety risks but also drastically simplifies the post-reaction workup. Instead of complex quenching procedures required for hydride reductions or filtration of heavy metal catalysts, the enzymatic process allows for simple filtration to remove the biocatalyst followed by solvent distillation. This streamlined workflow reduces the number of unit operations, minimizes solvent consumption through efficient recycling, and significantly lowers the environmental footprint of the manufacturing process, making it an ideal candidate for sustainable industrial scale-up.
Mechanistic Insights into CIR Enzyme-Catalyzed Reduction
The core of this technological breakthrough lies in the unique catalytic activity of the CIR enzyme, identified by the protein sequence SEQ ID NO. 1, which acts as a highly efficient biocatalyst for the stereoselective reduction of the ketone moiety. Unlike chemical reducers that rely on electron transfer from reactive metal hydrides, the CIR enzyme facilitates the transfer of hydride equivalents from a cofactor (typically NADPH or similar, regenerated in situ or added) to the carbonyl carbon of the substrate, 2-deoxy-2,2-difluoropentose-1-one-3,5-dibenzoate. This enzymatic pocket provides a chiral environment that ensures the hydride attack occurs from a specific face of the planar carbonyl group, thereby enforcing high stereoselectivity and preventing the formation of unwanted diastereomers that often plague non-enzymatic reductions. The reaction proceeds smoothly in an alcohol solvent, which serves not only as a medium for substrate solubility but also potentially participates in the stabilization of the enzyme-substrate complex, maintaining the structural integrity of the biocatalyst throughout the reaction cycle.
The overall transformation is depicted in the reaction scheme below, which clearly demonstrates the conversion of Compound II into Compound I with the retention of the difluoro motif and benzoyl groups, underscoring the chemoselectivity of the enzyme.
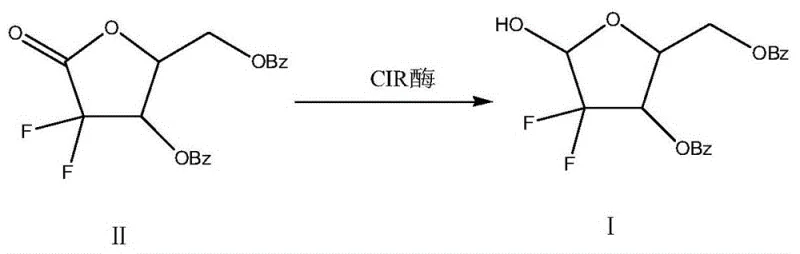
From an impurity control perspective, the mild nature of the enzymatic reaction is a decisive advantage. Chemical reductions at elevated temperatures or with aggressive reagents often trigger side reactions such as dehalogenation of the fluorine atoms or hydrolysis of the ester bonds. However, the CIR enzyme operates at a physiological temperature range of 20°C to 30°C, effectively suppressing these thermal degradation pathways. The result is a crude reaction mixture with a significantly cleaner profile, where the primary impurity is typically unreacted starting material rather than complex structural analogs. This high level of purity, often exceeding 96% directly from the reaction pot and reaching up to 99% with optimization, reduces the burden on crystallization and chromatography steps, ensuring a robust and consistent quality of the final pharmaceutical intermediate.
How to Synthesize 2-deoxy-2,2-difluoro-3,5-dibenzoyl-D-ribofuranose Efficiently
Implementing this biosynthetic route in a laboratory or pilot plant setting requires careful attention to enzyme loading and solvent selection to maximize efficiency. The patent data suggests that an enzyme loading of 10% to 25% by weight relative to the substrate is optimal, with a preferred range of 18% to 20% providing the best balance between reaction rate and cost. The choice of alcohol solvent is also critical; isopropanol and ethanol are preferred due to their low toxicity, ability to dissolve the substrate effectively, and ease of removal via distillation. The reaction is typically allowed to proceed for 4 to 6 hours, after which the enzyme is removed by simple filtration. The following section outlines the standardized procedural steps derived from the patent examples to ensure reproducible high-yield results.
- Prepare the reaction mixture by dissolving 2-deoxy-2,2-difluoropentose-1-one-3,5-dibenzoate in an alcohol solvent such as isopropanol or ethanol.
- Add the specific CIR enzyme (SEQ ID NO. 1) to the solution, ensuring the enzyme loading is between 10% to 25% by weight relative to the substrate.
- Maintain the reaction temperature between 20°C and 30°C for 4 to 6 hours, then filter and distill to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic technology translates into tangible strategic benefits that extend far beyond simple yield improvements. The elimination of expensive palladium catalysts and hazardous hydride reagents fundamentally alters the cost structure of the intermediate, removing volatile raw material costs and the substantial expenses associated with hazardous waste disposal. Furthermore, the use of common, low-cost alcohol solvents like isopropanol allows for straightforward solvent recovery and recycling loops, which significantly reduces the overall solvent purchase volume and waste treatment fees. This process intensification leads to a leaner manufacturing operation with fewer unit operations, directly contributing to a lower cost of goods sold (COGS) and improved margin potential for the final API.
- Cost Reduction in Manufacturing: The transition to a biocatalytic process eliminates the dependency on precious metal catalysts like palladium, which are subject to significant market price fluctuations and supply chain constraints. By removing the need for high-pressure hydrogenation equipment and the associated safety infrastructure, capital expenditure (CAPEX) for new production lines is drastically reduced. Additionally, the mild reaction conditions minimize energy consumption for heating and cooling, while the simplified workup procedure reduces labor hours and processing time, collectively driving down the operational expenditure (OPEX) for the manufacturing of this critical oncology intermediate.
- Enhanced Supply Chain Reliability: Relying on hazardous reagents like lithium aluminum hydride often introduces supply chain fragility due to strict transportation regulations and storage limitations. In contrast, the CIR enzyme and alcohol solvents used in this process are stable, easy to transport, and widely available, ensuring a continuous and uninterrupted supply of raw materials. The robustness of the enzymatic reaction against minor variations in conditions also means higher batch-to-batch consistency, reducing the risk of production failures and ensuring that delivery schedules to downstream API manufacturers are met with greater reliability and predictability.
- Scalability and Environmental Compliance: As regulatory pressures on pharmaceutical manufacturing intensify, the environmental profile of a synthetic route becomes a key differentiator. This enzymatic method generates significantly less hazardous waste compared to traditional chemical reductions, simplifying compliance with increasingly stringent environmental regulations. The absence of heavy metal residues in the product stream removes the need for complex metal scavenging steps, facilitating a smoother regulatory filing process. Moreover, the process is inherently scalable, having been demonstrated to work efficiently from gram to kilogram scales, making it ready for immediate commercial scale-up to meet global demand without the need for extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this CIR enzyme-catalyzed biosynthesis. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, aiming to clarify the operational parameters and benefits for potential partners and licensees. Understanding these details is crucial for R&D teams evaluating the feasibility of technology transfer and for procurement teams assessing the long-term viability of this supply source.
Q: What are the advantages of using CIR enzyme over traditional chemical reduction for this intermediate?
A: The CIR enzyme method eliminates the need for hazardous reagents like lithium aluminum hydride or expensive palladium-carbon catalysts. It operates under mild conditions (20-30°C) in safe alcohol solvents, resulting in significantly higher yields (>98%) and purity (>96%) while reducing environmental waste.
Q: How does this biosynthetic route impact the impurity profile of the final API?
A: Enzymatic catalysis offers superior stereoselectivity compared to chemical reduction. By specifically targeting the carbonyl group without affecting other sensitive functionalities, the CIR enzyme minimizes the formation of side-products and diastereomers, simplifying downstream purification and ensuring a cleaner impurity profile for the final Gemcitabine hydrochloride.
Q: Is this enzymatic process scalable for industrial production?
A: Yes, the process is highly scalable. The use of common alcohol solvents like isopropanol facilitates easy solvent recovery and recycling. Furthermore, the mild reaction conditions reduce energy consumption and equipment corrosion risks, making it ideal for large-scale commercial manufacturing from 100 kgs to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-deoxy-2,2-difluoro-3,5-dibenzoyl-D-ribofuranose Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of oncology intermediates like 2-deoxy-2,2-difluoro-3,5-dibenzoyl-D-ribofuranose for the global pharmaceutical market. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation capabilities, allowing us to implement complex biocatalytic processes with stringent purity specifications that meet or exceed international pharmacopeia standards. We are committed to delivering not just a product, but a comprehensive supply solution that integrates seamlessly into your existing manufacturing workflows.
We invite you to collaborate with us to leverage this advanced enzymatic technology for your Gemcitabine production needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this green synthesis route can optimize your bottom line. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample availability, and let us partner with you to drive efficiency and sustainability in your pharmaceutical supply chain.
