Advanced Synthesis of Substituted Benzotriazole Compounds for High-Performance UV Protection
The development of high-performance ultraviolet (UV) absorbers is critical for enhancing the durability and longevity of polymeric materials and coatings exposed to harsh environmental conditions. Patent CN101717372B introduces a significant technological advancement in the synthesis of substituted benzotriazole compounds, specifically targeting the production of 2-[2'-hydroxy-4'-(2-hydroxy-3-alkoxy(or aryloxy)-propoxy)phenyl]-5(substituted)-2H-benzotriazole derivatives. This intellectual property outlines a novel synthetic pathway that diverges from traditional methodologies by utilizing (2-hydroxy-3-chloro)propyl alkyl or aryl ethers as key alkylating agents instead of the more complex epoxypropyl ethers. By streamlining the reaction sequence and optimizing the choice of solvents and inorganic base catalysts, this invention addresses long-standing challenges related to process complexity and production costs in the fine chemical sector. For R&D directors and procurement specialists seeking reliable sources for advanced UV stabilizer intermediates, understanding the mechanistic nuances and commercial implications of this patent is essential for strategic sourcing and product development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of these specialized benzotriazole UV absorbers has relied heavily on the reaction between 2-(2',4'-dihydroxyphenyl)-5(substituted)-2H-benzotriazole and epoxypropyl alkyl or aryl ethers. As illustrated in the reaction scheme below, this conventional route necessitates the prior synthesis of the epoxide intermediate, which itself involves multiple steps including the ring-opening of epichlorohydrin with alcohols or phenols followed by a dehydrohalogenation elimination reaction. 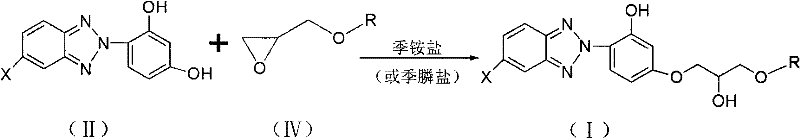 This multi-step precursor synthesis not only increases the overall material cost due to the consumption of additional reagents like boron trifluoride ether complexes but also introduces significant operational inefficiencies. Furthermore, the subsequent coupling reaction typically requires the presence of quaternary ammonium or phosphonium salts as phase transfer catalysts to facilitate the reaction in inert solvents, adding another layer of complexity to the downstream purification process. The reliance on these specific catalytic systems and the inherent instability of epoxide intermediates often lead to variability in yield and purity, creating bottlenecks for manufacturers aiming for consistent commercial scale-up of complex polymer additives.
This multi-step precursor synthesis not only increases the overall material cost due to the consumption of additional reagents like boron trifluoride ether complexes but also introduces significant operational inefficiencies. Furthermore, the subsequent coupling reaction typically requires the presence of quaternary ammonium or phosphonium salts as phase transfer catalysts to facilitate the reaction in inert solvents, adding another layer of complexity to the downstream purification process. The reliance on these specific catalytic systems and the inherent instability of epoxide intermediates often lead to variability in yield and purity, creating bottlenecks for manufacturers aiming for consistent commercial scale-up of complex polymer additives.
The Novel Approach
In stark contrast to the legacy methods, the technology disclosed in CN101717372B proposes a direct alkylation strategy that fundamentally simplifies the manufacturing workflow. The core innovation lies in the substitution of the epoxypropyl ether with a (2-hydroxy-3-chloro)propyl alkyl or aryl ether, which reacts directly with the benzotriazole substrate under the influence of an inorganic base catalyst. 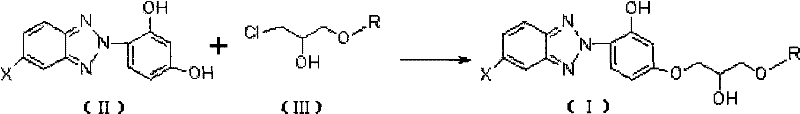 This approach effectively bypasses the need for the elimination step required to form the epoxide, thereby shortening the synthetic route by at least one major chemical transformation. The reaction is conducted in aprotic polar solvents such as n-butanol, DMF, or methyl isobutyl ketone at elevated temperatures ranging from 70°C to 180°C, ensuring robust reaction kinetics without the need for expensive phase transfer catalysts. By eliminating the epoxide intermediate and the associated quaternary salt catalysts, this novel approach not only reduces the raw material inventory requirements but also significantly simplifies the post-reaction workup, leading to a more economically viable and environmentally friendly production process for high-purity OLED material precursors and coating additives.
This approach effectively bypasses the need for the elimination step required to form the epoxide, thereby shortening the synthetic route by at least one major chemical transformation. The reaction is conducted in aprotic polar solvents such as n-butanol, DMF, or methyl isobutyl ketone at elevated temperatures ranging from 70°C to 180°C, ensuring robust reaction kinetics without the need for expensive phase transfer catalysts. By eliminating the epoxide intermediate and the associated quaternary salt catalysts, this novel approach not only reduces the raw material inventory requirements but also significantly simplifies the post-reaction workup, leading to a more economically viable and environmentally friendly production process for high-purity OLED material precursors and coating additives.
Mechanistic Insights into Inorganic Base-Catalyzed Nucleophilic Substitution
The chemical mechanism driving this synthesis is a classic nucleophilic substitution where the phenolic hydroxyl group of the benzotriazole derivative acts as the nucleophile. In the presence of an inorganic base catalyst, such as sodium carbonate, potassium carbonate, or sodium hydroxide, the phenolic proton is abstracted to generate a highly reactive phenoxide anion. This anionic species then attacks the electrophilic carbon atom adjacent to the chlorine leaving group on the (2-hydroxy-3-chloro)propyl ether chain. The use of aprotic polar solvents is critical in this mechanism as they effectively solvate the cation of the inorganic base while leaving the phenoxide anion relatively naked and more nucleophilic, thereby accelerating the reaction rate. The reaction temperature is carefully controlled between 90°C and 150°C to provide sufficient activation energy for the substitution to proceed to completion within a practical timeframe of 2 to 10 hours, ensuring high conversion rates while minimizing potential thermal degradation of the sensitive benzotriazole core.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over the epoxide ring-opening route. In the conventional epoxide method, regioselectivity can sometimes be an issue, potentially leading to mixtures of isomers if the ring opening is not perfectly controlled. However, the direct displacement of the chloride ion in the new method is highly regioselective, occurring primarily at the terminal carbon bearing the chlorine atom. Furthermore, the byproduct of this reaction is simply an inorganic salt (e.g., sodium chloride or potassium chloride), which is insoluble in the organic reaction medium at lower temperatures and can be easily removed by hot filtration. This physical separation method is far superior to chromatographic separations often required to remove organic catalysts, resulting in a final product with exceptional purity profiles, often exceeding 99% as demonstrated in the patent examples, which is crucial for applications requiring stringent purity specifications in electronic chemical manufacturing.
How to Synthesize Substituted Benzotriazole Efficiently
The practical implementation of this synthesis involves a straightforward procedure that is well-suited for standard chemical processing equipment found in most fine chemical facilities. The process begins by charging the reactor with the benzotriazole starting material, the chosen inorganic base, and the solvent, followed by heating to the target temperature before the slow addition of the chlorohydrin ether reagent. Detailed standardized operating procedures regarding specific molar ratios, addition rates, and crystallization parameters are critical for reproducibility and are outlined in the technical documentation below.
- Combine 2-(2',4'-dihydroxyphenyl)-5(substituted)-2H-benzotriazole with an inorganic base catalyst in an aprotic polar solvent.
- Add (2-hydroxy-3-chloro)propyl alkyl or aryl ether dropwise while maintaining the reaction temperature between 70°C and 180°C.
- Filter off inorganic salts while hot, concentrate the solution, and recrystallize the product to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology presents a compelling value proposition centered around cost efficiency and supply reliability. The primary economic driver is the reduction in the number of synthetic steps, which directly correlates to lower labor costs, reduced energy consumption, and decreased waste generation. By removing the need to synthesize the epoxypropyl ether intermediate in-house or source it from external suppliers, manufacturers can significantly reduce their raw material exposure and simplify their supply chain logistics. This consolidation of the supply chain not only mitigates the risk of delays associated with multi-vendor sourcing strategies but also enhances the overall agility of the production schedule, allowing for faster response times to market demands for reliable agrochemical intermediate supplier networks.
- Cost Reduction in Manufacturing: The elimination of the epoxide synthesis step and the removal of expensive quaternary ammonium catalysts result in substantial cost savings across the entire production lifecycle. Without the need for the dehydrohalogenation step to form the epoxide, the consumption of auxiliary reagents like strong bases and Lewis acid catalysts is drastically reduced, leading to a leaner bill of materials. Additionally, the simplified workup procedure involving simple filtration and recrystallization reduces the demand for extensive purification solvents and processing time, further driving down the operational expenditure per kilogram of finished product. These cumulative efficiencies translate into a more competitive pricing structure for the final UV absorber, providing a distinct margin advantage in cost reduction in coating additive manufacturing.
- Enhanced Supply Chain Reliability: Utilizing (2-hydroxy-3-chloro)propyl ethers as starting materials leverages a supply chain that is generally more robust and less prone to volatility compared to specialized epoxide intermediates. These chlorohydrin ethers are chemically stable and can be stored for extended periods without the risk of polymerization or degradation that often plagues epoxide inventories. This stability allows manufacturers to maintain strategic stockpiles of raw materials, ensuring continuous production even during periods of upstream supply disruption. Consequently, this leads to reducing lead time for high-purity polymer additives, enabling suppliers to meet tight delivery windows and maintain high service levels for their downstream customers in the plastics and coatings industries.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on common inorganic bases and standard organic solvents that are readily available in bulk quantities globally. The absence of heavy metal catalysts or complex organometallic reagents simplifies the environmental compliance landscape, as the waste streams are easier to treat and dispose of according to standard industrial regulations. The generation of benign inorganic salt byproducts facilitates easier wastewater treatment compared to processes generating complex organic waste, aligning with modern green chemistry principles. This environmental compatibility ensures that the commercial scale-up of complex UV absorbers can proceed without significant regulatory hurdles, supporting sustainable growth and long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis method, derived directly from the experimental data and claims within the patent literature. Understanding these details is vital for process engineers looking to adapt this technology for large-scale production environments.
Q: What are the optimal reaction conditions for this benzotriazole synthesis?
A: According to patent CN101717372B, the reaction proceeds optimally at temperatures between 90°C and 150°C for a duration of 4 to 6 hours, utilizing solvents such as n-butanol, DMF, or methyl isobutyl ketone.
Q: How does this method improve upon conventional glycidyl ether routes?
A: This method eliminates the need for synthesizing epoxypropyl alkyl ethers as intermediates, thereby reducing the total number of synthetic steps and avoiding the use of expensive phase transfer catalysts required in the traditional epoxide ring-opening process.
Q: What purity levels can be achieved with this synthesis protocol?
A: Experimental data within the patent indicates that following the described workup procedures, including hot filtration and recrystallization, HPLC purity levels consistently exceed 98.4%, with several examples reaching above 99.0%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Benzotriazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality UV absorber intermediates in the formulation of durable polymers and advanced coatings. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of substituted benzotriazole performs consistently in your final applications. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal conditions required by this patented process, ensuring optimal yield and quality.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your supply chain efficiency and product performance.
